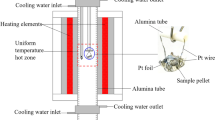
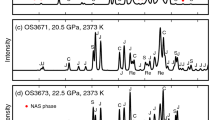
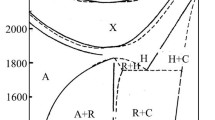
Abstract
Phase relations of the Al2O3–SiO2 system were determined by multi-anvil experiments at pressures of 13–21 GPa and temperatures of 2300–2800 K. We obtained a new high-pressure Al2Si2O7 phase (227-phase), which possesses a triclinic unit cell (Z = 3) with a = 7.0237(12) Å, b = 7.1002(14) Å, c = 6.6729(10) Å, α = 103.466(15)°, β = 99.107(15)°, γ = 60.542(12)°, and V = 281.41(24) Å3, in addition to the two new high-pressure Al2SiO5 phases reported recently, kyanite II and kyanite III. Because of the formation of the three new phases at temperatures above 2400 K, the phase relations are complicated, in contrast to the previously-known phase assemblage of corundum + stishovite stable at the post-kyanite pressures and temperatures below 2400 K. The three new phases and also kyanite are chemically invariant intermediate phases in the binary system and can be formed by the isothermal reactions, Al2O3 (corundum) + SiO2 (stishovite) = Al2SiO5 (kyanite II or III) and Al2SiO5 (kyanite, II, or III) + SiO2 (stishovite) = Al2Si2O7 (227-phase). In comparison to kyanite II and kyanite III, 227-phase is formed at higher temperatures. Crystal structure of 227-phase is based on distorted closest packing of O2− and tetrahedrally- and octahedrally-coordinated cations, similar to those of kyanite and kyanite II. The appearance of 227-phase once again suggests the key effect of temperature for stabilizing dense crystal structures fully made of Al–O and Si–O polyhedra at high pressures.








Similar content being viewed by others
References
Ahmed-Zaïd I, Madon M (1991) A high-pressure form of Al2SiO5 as a possible host of aluminum in the lower mantle. Nature 353(6343):426–428. https://doi.org/10.1038/353426a0
Åsbrink S (1980) The crystal structure of and valency distribution in the low-temperature modification of V3O5. The decisive importance of a few very weak reflexions in a crystal-structure determination. Acta Cryst B36:1332–1339. https://doi.org/10.1107/S0567740880006036
Brown ID (1992) Chemical and steric constraints in inorganic solids. Acta Cryst B48:553–572. https://doi.org/10.1107/S0108768192002453
Farrugia LJ (1999) WinGX suite for small-molecule single-crystal crystallography. J Appl Crystallogr 32(4):837–838. https://doi.org/10.1107/S0021889899006020
Hazen RM (1976) Effects of temperature and pressure on the crystal structure of forsterite. American Mineralogist 61:1280–1293. https://pubs.geoscienceworld.org/msa/ammin/article-abstract/61/11-12/1280/40602/Effects-of-temperature-and-pressure-on-the-crystal?redirectedFrom=fulltext
Horiuchi H, Sawamoto H (1981) β-Mg2SiO4: Single-crystal X-ray diffraction study. American Mineralogist 66:568–575. https://pubs.geoscienceworld.org/msa/ammin/article-abstract/66/5-6/568/41286/Mg2SiO4-Single-crystal-X-ray-diffraction-study?redirectedFrom=fulltext
Horiuchi H, Hirano M, Ito E, Matsui Y (1982) MgSiO3 (ilmenite-type): single crystal X-ray diffraction study. American Mineralogist 67:788–793. https://pubs.geoscienceworld.org/msa/ammin/article-abstract/67/7-8/788/41416/MgSiO3-ilmenite-type-single-crystal-X-ray?redirectedFrom=fulltext
Inoue K, Yamaguchi A (2003) Synthesis of Al4SiC4. J Am Ceram Soc 86(6):1028–1030. https://doi.org/10.1111/j.1151-2916.2003.tb03414.x
Irifune T, Kuroda K, Minagawa T, Unemoto M (1995) Experimental study of the decomposition of kyanite at high pressure and high temperature. In: Yukutake T (ed) The Earth's central part: Its structure and dynamics, Tokyo: Terra Scientific Publishing Company (TERRAPUB), pp 35–44. https://www.terrapub.co.jp/e-library/ecp/pdf/EC0035.PDF
Kirfel A, Eichhorn K (1990) Accurate structure analysis with synchrotron radiation. The electron density in Al2O3 and Cu2O. Acta Cryst A46:271–284. https://doi.org/10.1107/S0108767389012596
Litasov KD, Kagi H, Shatskiy A, Ohtani E, Lakshtanov DL, Bass JD, Ito E (2007) High hydrogen solubility in Al-rich stishovite and water transport in the lower mantle. Earth Planetary Sci Lett 262:620–634. https://doi.org/10.1016/j.epsl.2007.08.015
Oganov AR, Brodholt JP (2000) High-pressure phases in the Al2SiO5 system and the problem of aluminous phase in the Earth’s lower mantle: ab initio calculation. Phys Chem Miner 27:430–439. https://doi.org/10.1007/s002699900081
Oganov AR, Ono S (2005) The high-pressure phase of alumina and implications for Earth’s D’’ layer. Proc Natl Acad Sci USA 102:10828–10831. https://doi.org/10.1073/pnas.0501800102
Oganov AR, Gillan MJ, David Price G (2005) Structural stability of silica at high pressures and temperatures. Phys Rev B 71:064104. https://doi.org/10.1103/PhysRevB.71.064104
Palatinus L, Chapuis G (2007) Superflip—a computer program for the solution of crystal structures by charge flipping in arbitrary dimensions. J Appl Crystallogr 40(4):786–790. https://doi.org/10.1107/S0021889807029238
Ross NL, Shu J, Hazen RM, Gasparik T (1990) High-pressure crystal chemistry of stishovite. American Mineralogist 75:739–747. https://pubs.geoscienceworld.org/msa/ammin/article-abstract/75/7-8/739/42397/High-pressure-crystal-chemistry-of-stishovite?redirectedFrom=fulltext
Sasaki S, Prewitt CT, Sato Y, Ito E (1982) Single-crystal X-ray study of γ-Mg2SiO4. J Geophys Res 87:7829–7832. https://doi.org/10.1029/JB087iB09p07829
Schmidt MW, Poli S, Comodi P, Zanazzi PF (1997) High-pressure behavior of kyanite: decomposition of kyanite into stishovite and corundum. Am Miner 82:460–466. https://doi.org/10.2138/am-1997-5-603
Schmidt MW, Finger LW, Angel RJ, Dinnebier RE (1998) Synthesis, crystal structure, and phase relations of AlSiO3OH, a high-pressure hydrous phase. Am Miner 83:881–888. https://doi.org/10.2138/am-1998-7-820
Sheldrick GM, Schneider TR (1997) SHELXL: High-resolution refinement. Methods Enzymol 277:319–343. https://doi.org/10.1016/S0076-6879(97)77018-6
Tsuchiya T, Caracas R, Tsuchiya J (2004) First principles determination of the phase boundaries of high-pressure polymorphs of silica. Geophys Res Lett 31:L11610. https://doi.org/10.1029/2004GL019649
Tsuchiya J, Tsuchiya T, Wentzcovitch RM (2005) Transition from the Rh2O3(II)-to-CaIrO3 structure and the high-pressure-temperature phase diagram of alumina. Phys Rev B 72:020103(R). https://doi.org/10.1103/PhysRevB.72.020103
Winter JK, Ghose S (1979) Thermal expansion and high-temperature crystal chemistry of the Al2SiO5 polymorphs. American Mineralogist 64:573–586. https://www.minsocam.org/ammin/AM64/AM64_573.pdf
Wunder B, Rubie DC, Ross II CR, Medenbach O, Seifert F, Schreyer W (1993) Synthesis, stability, and properties of Al2SiO4(OH)2: a fully hydrated analogue of topaz. American Mineralogist 78:285–297. https://pubs.geoscienceworld.org/msa/ammin/article-abstract/78/3-4/285/42731/Synthesis-stability-and-properties-of-Al2SiO4-OH-2?redirectedFrom=fulltext
Yamanaka T, Fukuda T, Tsuchiya J (2002) Bonding character of SiO2 stishovite under high pressures up to 30 GPa. Phys Chem Miner 29(9):633–641. https://doi.org/10.1007/s00269-002-0257-3
Zhou Y, Irifune T, Ohfuji H, Kuribayashi T (2018) New high-pressure forms of Al2SiO5. Geophys Res Lett 45:8167–8172. https://doi.org/10.1029/2018GL078960
Acknowledgements
The synchrotron X-ray diffraction experiments were performed at BL-10A, PF, KEK, Japan (PAC. 2018G118; 2020G586).
Funding
The present study was supported by the Grand-in-Aid for Scientific Research on Innovative Areas (No. 15H5829) to Tetsuo Irifune.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, Y., Irifune, T. & Kuribayashi, T. Phase relations of the Al2O3–SiO2 system at 13–21 GPa and 2300–2800 K and a new high-pressure Al2Si2O7 phase. Phys Chem Minerals 48, 26 (2021). https://doi.org/10.1007/s00269-021-01149-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00269-021-01149-3