Abstract
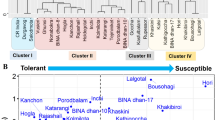
Salinity is a major constraint for plant growth, development and yield worldwide. Evaluation of a large number of germplasms in salt-stressed environments may help identify superior salt-tolerant genotypes. The present study dissects the genetic diversity of 33 pearl millet genotypes (landraces and inbred lines) for salinity tolerance through in vitro screening at the seedling stage. Our results revealed a significant reduction in total biomass and shoot growth of the salt-sensitive genotypes upon exposure to 150 mM NaCl, in contrast to the tolerant genotypes showing better growth characteristics. A significant differential effect of salt treatment on morphological traits was observed by analysis of variance (ANOVA), confirming substantial genetic diversity among all genotypes for salt tolerance. The genotypes were clustered into three groups based on multiple stress indices. The genotypes were also evaluated using principal component analysis (PCA) to identify the key contributing traits for stress tolerance. Based on these results, a total of four contrasting genotypes were selected for further biochemical and molecular analysis. Physiological studies confirmed that salt tolerance might be due to the higher content of osmolytes and the activity of antioxidant enzymes. Similarly, gene expression profiling of catalase (CAT), glutamate dehydrogenase (GDH), glutathione reductase (GR), and nitrate reductase (NR) revealed a profound increase in NR and GDH transcript levels in the tolerant genotypes, suggesting their major role as reactive oxygen species (ROS) scavengers under salinity. The overall findings of this study could be utilized further for candidate gene mining through “omics” approaches, aiming toward development of salinity resilient crop plants.





Similar content being viewed by others
Data Availability
Available to scientific community.
Code Availability
NA
References
Aebi H (1984) Catalase in Vitro Methods Enzymol 105:121–126
Agarwal S, Jha S, Sanyal I, Amla DV (2009) Effect of point mutations in translation initiation context on the expression of recombinant human α1-proteinase inhibitor in transgenic tomato plants. Plant Cell Rep 28:1791–1798
Ahmad Z, Ajmal SU, Munir M, Zubair M, Masood MS (2008) Genetic diversity for morpho-genetic traits in barley germplasm. Pak J Bot 40:1217–1224
Ara N, Nakkanong K, Lv W, Yang J, Hu Z, Zhang M (2013) Antioxidant enzymatic activities and gene expression associated with heat tolerance in the stems and roots of two cucurbit species (“Cucurbita maxima” and “Cucurbita moschata”) and their interspecific inbred line “Maxchata.” Int J Mol Sci 14(12):24008–24028
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Cardamone L, Cuatrín A, Grunberg K, Tomás MA (2018) Variability for salt tolerance in a collection of Panicum coloratum var. makarikariense during early growth stages. Trop Grassl-Forrajes Tropicales 6(3):134–147
Choudhary S, Vadez V, Tom Hash C et al (2019) Pearl millet mapping population parents: performance and selection under salt stress across environments varying in evaporative demand. Proc Natl Acad Sci, India, Sect B Biol Sci 89:201–211
Fariduddin Q, Ahmad BA, Ahmad A (2013) Physiological and biochemical traits as tools to screen sensitive and resistant varieties of tomatoes exposed to salt stress. Braz J Plant Physiol 24(4):281–292
Fernandez GCJ 1992 Effective selection criteria for assessing stress tolerance. In Kuo CG (ed) Proceedings of the international symposium on adaptation of vegetables and other food crops in temperature and water stress, Taiwan, pp. 257–270
Fischer RA, Maurer R (1978) Drought resistance in spring wheat cultivars. I. Grain yield response. Aust J Agric Res 29:897–907
Food and Agriculture Organization of the United Nations 2011 The state of the world’s land and water resources for food and agriculture; managing systems at risk. http://www.fao.org/3/a-i1688e.pdf
Genc Y, McDonald GK, Tester M (2007) Reassessment of tissue Na+ concentration as a criterion for salinity tolerance in bread wheat. Plant Cell Environ 30:1486–1498
Gregorio GB, Senadhira D, Mendoza RD 1997 Screening rice for salinity tolerance. IRRI Discussion Paper Series No. 22:1–30, International Rice research Institute, Manila, Philippines
Guellim A, Catterou M, Chabrerie O, Tetu T, Hirel B, Dubois F, Ahmed HB, Kichey T (2019) Identification of phenotypic and physiological markers of salt stress tolerance in durum wheat (Triticum durum Desf) through integrated analyses. Agronomy 9:e844
Haya S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Signal Behav 7(11):1456–1466
Hoang TML, Tran TN, Nguyen TKT, Williams B, Wurm P, Bellairs S, Mundree S (2016) Improvement of salinity stress tolerance in rice: challenges and opportunities. Agronomy 6:54
Honold GR, Stahmann MA (1968) The oxidation-reduction enzymes of wheat iv qualitative and quantitative investigations of the oxidases. Cereal Chem 45(2):99–108
Hussain K, Ashraf M, Ashraf MY (2008) Relationship between growth and ion relation in pearl millet (Pennisetum glaucum (L) R Br) at different growth stages under salt stress. Afr J Plant Sci 2(3):23–27
Jha S (2018) Proteomics of salinity stress: opportunities and challenges. In: Ramakrishna A, Gill SS (eds) Metabolic adaptations in plants during abiotic stress, CRC Press, Taylor & Francis Group. Boca Raton
Jha S (2019) Transgenic approaches for enhancement of salinity stress tolerance in plants. In: Singh SP et al (eds) Molecular approaches in plant biology and environmental challenges. Springer Nature, Singapore
Jogeswar G, Pallela R, Jakka NM, Reddy PS, Rao JV, Sreenivasulu N, Kavi Kishor PB (2006) Differential response of the antioxidant components of drought susceptible and tolerant varieties of sorghum during seedling growth under short-term salinity stress. Acta Physiol Plant 28:465–475
Krishnamurthy L, Serraj R, Rai KN, Hash CT, Dakhee AJ (2007) Identification of pearl millet [Pennisetum glaucum (L.) R. Br.] lines tolerant to soil salinity. Euphytica 158:179–188
Krishnamurthy L, Zaman-Allah M, Purushothaman R, Irshad Ahmed M, Vadez V (2011). Plant Biomass Productivity under Abiotic Stresses in SAT Agriculture. https://doi.org/10.5772/17279,Pg247-264
Krishnamurthy L, Upadhyaya HD, Purushothaman R, Gowda CLL, Kashiwagi J, Dwivedi SL, Singh S, Vadez V (2014) The extent of variation in salinity tolerance of the minicore collection of finger millet (Eleusine coracana L. Gaertn.) germplasm. Plant Sci 227:51–59
Krishnamurthy SL, Sharma PC, Sharma SK, Batra V, Kumar V, Rao LVS (2016) Effect of salinity and use of stress indices of morphological and physiological traits at the seedling stage in rice. Indian J Exp Bot 54:843–850
Kulkarni VN, Rai KN, Dakheel AJ, Ibrahim M, Hebbara M, Vadez V (2006) Pearl millet germplasm adapted to saline conditions. SAT eJournal 2(1):1–4
Lakra N, Kaur C, Anwar K, Singla-Pareek SL, Pareek A (2018) Proteomics of contrasting rice genotypes: Identification of potential targets for raising crops for saline environment. Plant Cell Environ 41(5):947–969
Lapuimakuni S, Khumaida N, Ardie SW (2018) Evaluation of drought tolerance indices for genotype selection of foxtail millet (Setaria italica). Trop Drylands 2(2):37–40
Luna CM, Pastori GM, Driscoll S, Groten K, Bernard S, Foyer CH (2005) Drought controls on H2O2 accumulation, catalase (CAT) activity and CAT gene expression in wheat. J Exp Bot 56(411):417–423
Maity RK, Satya P (2014) Research advances in major cereal crops for adaptation to abiotic stresses. GM Crops Food 5(4):259–279
Manga VK (2015) Diversity in pearl millet [Pennisetum glaucum (L) R BR] and its management. Indian J Plant Sci 4(1):38–51
Morton MJL, Awlia M, Al-Tamimi N, Saade S, Pailles Y, Negrão S, Tester M (2019) Salt stress under the scalpel – dissecting the genetics of salt Tolerance. The Plant J 97:148–163
Mukhopadhyay R, Hash CT, Bhasker Raj AG, Kavi Kishor PB (2005) Assessment of opportunities to map pearl millet tolerance to salinity during germination and early seedling growth. SAT eJournal 1(1):1–4
Mukhopadhyay R, Reddy PS, Pallela R, Reddy NC, Rao JV, Hash CT, Kavi Kishor PB (2007) Differential responses of proline, ion accumulation and antioxidative enzyme activities in pearl millet [Pennisetum glaucum (L) R Br] lines differing in salt sensitivity. J Plant Biol 34(3):185–192
Negrão S, Schmöckel SM, Tester M (2017) Evaluating physiological responses of plants to salinity stress. Annals Bot 119:1–11
Nguyen HTT, Shim IS, Kobayashi K, Usui K (2005) Regulation of ammonium accumulation during salt stress in rice (Oryza sativa l) seedlings. Plant Prod Sci 8(4):397–404
Ni W, Trelease RN (1991) Post-transcriptional regulation of catalase isozyme expression in cotton seeds. Plant Cell 3(7):737–744
Palma JM, Mateos RM, López-Jaramillo J, Rodríguez-Ruiz M, González-Gordo S, Lechuga-Sancho AM, Corpas FJ (2020) Plant catalases as NO and H2S targets. Redox Biol 34:e101525
Quan R, Wang J, Hui J et al (2018) Improvement of salt tolerance using wild rice genes. Front Plant Sci 8:2269
Radhouane L (2008) Correlation between germination and adult stage under water stress in some Tunisian autochthonous pearl millet ecotypes (Pennisetum glaucum (L) R Br). C R Biol 331(8):623–630
Rajabi Dehnavi A, Zahedi M, Ludwiczak A, Cardenas Perez S, Piernik A (2020) Effect of salinity on seed germination and seedling development of sorghum (Sorghum bicolor (L) Moench) genotypes. Agronomy 10(6):e859
Ribadiya TR, Savalia SG, Vadaliya BM, Davara MA (2018) Effect of salinity on yield, yield attributes and quality of pearl millet (Pennisetum glaucum L) varieties. Int J Chem Studies 6(6):878–882
Roe JH (1955) The determination of sugars in blood and spinal fluid with anthrone reagent. J Biol Chem 212:335–338
Rosielle AA, Hamblin J (1981) Theoretical aspects of selection for yield in stress and non-stress environments. Crop Sci 21:943–946
Rossatto T, do Amaral MN, Benitez LC, Vighi IL, Braga EJB, de Magalhães Júnior AM, Maia MAC, da Silva Pinto L 2017 Gene expression and activity of antioxidant enzymes in rice plants, cv. BRS AG, under saline stress. Physiol Mol Biol Plants 23(4): 865–875
Shinde H, Tanaka K, Dudhate A, Tsugama D, Mine Y, Kamiya T, Gupta SK, Liu S, Takano T (2018) Comparative de novo transcriptomic profiling of the salinity stress responsiveness in contrasting pearl millet lines. Environ Exp Bot 155:619–627
Shivhare R, Lata C (2017) Exploration of genetic and genomic resources for abiotic and biotic stress tolerance in pearl millet. Front Plant Sci 7:2069
Shivhare R, Lata C (2019) Assessment of pearl millet genotypes for drought stress tolerance at early and late seedling stages. Acta Physiol Plantarum 41:39
Singh S, Sengar RS, Kulshreshtha N, Datta D, Tomar RS, Rao VP, Garg D, Ojha A (2015) Assessment of multiple tolerance indices for salinity stress in bread wheat (Triticum aestivum L). J Agri Sci 7(3):49–57
Singh V, Singh AP, Bhadoria J, Giri J, Singh J, Vineeth TV, Sharma PC (2018) Differential expression of salt-responsive genes to salinity stress in salt-tolerant and salt-sensitive rice (Oryza sativa L.) at seedling stage. Protoplasma 255:1667–1681
Tadele Z (2018) African orphan crops under abiotic stresses: challenges and opportunities. Scientifica 2018:1451894. https://doi.org/10.1155/2018/1451894
Toderich K, Shuyskaya E, Rakhmankulova Z, Bukarev R, Khujanazarov T, Zhapaev R, Ismail S, Gupta SK, Yamanaka N, Boboev F (2018) Threshold tolerance of new genotypes of Pennisetum glaucum (L) R Br to salinity and drought. Agronomy 8:e230
Uddin MS, Jahan N, Rahman MZ, Hossain KMW (2017) Growth and yield response of wheat genotypes to salinity at different growth stages. Int J Agron Agri R 11(2):60–67
Vaezi H, Mohammadi-Nejad G, Majidi-Heravan E, Nakhoda B, Darvish-Kajouri F (2020) Effective selection indices for improving tolerance to water stress in millet germplasm. Int J Plant Product 14:93–105
Yadav OP (2010) Drought response of pearl millet landrace-based populations and their crosses with elite composites. Field Crop Res 118(1):51–56
Yadav T, Kumar A, Yadav RK, Yadav G, Kumar R, Kushwaha M (2020) Salicylic acid and thiourea mitigate the salinity and drought stress on physiological traits governing yield in pearl millet- wheat. Saudi J Biol Sci 27:2010–2017
Yakubu H, Ngala AL, Dugje IY (2010) Screening of millet (Pennisetum glaucum L) varieties for salt tolerance in semi-arid soil of northern Nigeria. World J Agri Sci 6(4):374–380
Zeng L (2005) Exploration of relationships between physiological parameters and growth performance of rice (Oryza sativa L.) seedlings under salinity stress using multivariate analysis. Plant Soil 268:51–59
Zhang X, Yin H, Chen S, He J, Guo S 2014 Changes in antioxidant enzyme activity and transcript levels of related genes in Limonium sinense Kuntze seedlings under NaCl stress. J Chem 2014:1–6, Article ID 749047
Zörb C, Geilfus C-M, Dietz K-J (2019) Salinity and crop yield. Plant Biol J 21:31–38
Zulfiqar F, Akram NA, Ashraf M (2020) Osmoprotection in plants under abiotic stresses: new insights into a classical phenomenon. Planta 251(3):1–17
Acknowledgements
The authors gratefully acknowledge the Department of Science and Technology, Government of India for the DST-SERB Young Scientist grant (SB/YS/LS-39/2014) to provide financial support for the research and project-fellowship to JS and CC. The authors also acknowledge the University Grants Commission, Government of India for UGC-BSR Start-up research grant (F.30-50/2014/BSR); UGC-CAS program, and DST-FIST program in Department of Botany, J.N.V. University, Jodhpur (India) for providing basic infrastructural facilities, and CGIAR Research Program on Grain Legumes and Dryland Cereals. We also acknowledge Directors, NBPGR & ICRISAT for providing the valuable germplasms of pearl millet for the study, and Dr. Charu Lata (Sr. Scientist, CSIR-NISCAIR, New Delhi) for helping in data analysis.
Funding
The authors gratefully acknowledge Department of Science and Technology, Government of India for DST-SERB Young Scientist grant (SB/YS/LS-39/2014) to provide financial support for the research and project-fellowship to JS and CC. The authors also acknowledge University Grants Commission, Government of India for UGC-BSR Start-up research grant (F.30–50/2014/BSR); UGC-CAS program and DST-FIST program in Department of Botany, J.N.V. University, Jodhpur, India for providing basic infrastructural facilities.
Author information
Authors and Affiliations
Contributions
SJ conceptualized and designed the experiments. JS performed the screening of genotypes, CC performed physio-biochemical analysis and SJ carried out the molecular analysis. SJ analyzed and interpreted the data, and wrote the entire manuscript. OS and RKS critically reviewed and helped in the revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial or commercial conflict of interest.
Ethics Approval
NA
Consent to Participate
NA
Consent for Publication
NA
Additional information
Handling Editor: Heather Nonhebel.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jha, S., Singh, J., Chouhan, C. et al. Evaluation of Multiple Salinity Tolerance Indices for Screening and Comparative Biochemical and Molecular Analysis of Pearl Millet [Pennisetum glaucum (L.) R. Br.] Genotypes. J Plant Growth Regul 41, 1820–1834 (2022). https://doi.org/10.1007/s00344-021-10424-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-021-10424-0