Abstract
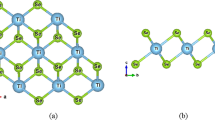
In this study, the electron density functional theory method is used to obtain maps of the electron density distribution for a monolayer TiS3. The adsorption of H2 on the surface of a monolayer TiS3 decorated with Mg cations is studied. The adsorption energy is –0.156 eV/molecule, which is a fairly high value, and allows us to consider this material as promising for creating hydrogen storage devices.






Similar content being viewed by others
REFERENCES
Dunn, S., Hydrogen futures: Toward a sustainable energy system, Int. J. Hydrogen Energy, 2002, vol. 27, no. 3, pp. 235–264.
Putungan, D.B., Lin, S.H., Wei, C.M., and Kuo, J.L., Li adsorption, hydrogen storage and dissociation using monolayer MoS2: An ab initio random structure searching approach, Phys. Chem. Chem. Phys., 2015, vol. 17, no. 17, pp. 11367–11374.
Bhatia, S.K. and Myers, A.L., Optimum conditions for adsorptive storage, Langmuir, 2006, vol. 22, no. 4, pp. 1688–1700.
Park, N., Choi, K., Hwang, J., Kim, D.W., Kim, D.O., and Ihm, J., Progress on first-principles-based materials design for hydrogen storage, Proc. Natl. Acad. Sci., 2012, vol. 109, no. 49, pp. 19893–19899.
Karpova, S.S., Kompan, M.E., and Maksimov, A.I., Osnovy vodorodnoi energetiki (Basics of Hydrogen Energy), 2nd ed., St., Petersburg: SPbGETU LETI, 2011.
Dou, Z.A., Ao, S., Xu, Z., Jiang, Q., and Wang, G., Hydrogen storage in porous graphene with Al decoration, J., Hydrogen Energ., 2014, vol. 39, pp. 16244–1625.
Reardon, H., Hanlon, J.M., Hughes, R.W., Godula-Jopek, A., Mandal, T.K., and Gregory, D.H., Emerging concepts in solid-state hydrogen storage: The role of nanomaterials design, Energy Environ. Sci., 2012, vol. 5, no. 3, pp. 5951–5979.
Van den Berg, A.W.C. and Areán, C.O., Materials for hydrogen storage: Current research trends and perspectives, Chem. Commun., 2008, no. 6, pp. 668–681.
Samanta, A., Furuta, T., and Li, J., Theoretical assessment of the elastic constants and hydrogen storage capacity of some metal-organic framework materials, J. Chem. Phys., 2006, vol. 125, no. 8, p. 084714.
Ataca, C., Aktürk, E., Ciraci, S., and Ustunel, H., High-capacity hydrogen storage by metallized graphene, Appl. Phys. Lett., 2008, vol. 93, no. 4, p. 103109.
Haldar, S., Mukherjee, S., Ahmed, F., and Veer, C., A first principles study of hydrogen storage in lithium decorated defective phosphorene, Int. J. Hydrogen Energy, 2017, pp. 1–10.
Hashmi, A., Farooq, M.U., Khan, I., Son, J., and Hong, J., Ultra-high capacity hydrogen storage in a Li decorated two-dimensional C2N layer, J. Mater. Chem. A, 2017, vol. 5, no. 6, pp. 2821–2828.
Schoen, A.H., Reflections concerning triply-periodic minimal surfaces, Interface Focus, 2012, vol. 2, no. 5, pp. 658–668.
Shevchenko, V.Ya., Sychev, M.M., Lapshin, A.E., Lebedev, L.A., Gruzdkov, A.A., and Glezer, A.M., Polymer structures with the topology of triply periodic minimal surfaces, Glass Phys. Chem., 2017, vol. 43, no. 6, pp. 608–610.
Mohsenizadeh, M., Gasbarri, F., Munther, M., Beheshti, A., and Davami, K., Additively-manufactured lightweight metamaterials for energy absorption, Mater. Des., 2018, vol. 139, pp. 521–530.
Lu, T.J., Stone, H.A., and Ashby, M.F., Heat transfer in open-cell metal foams, Acta Mater., 1998, vol. 46, no. 10, pp. 3619–3635.
Yuan, S., Shen, F., Bai, J., Chua, C.K., Wei, J., and Zhou, K., 3D soft auxetic lattice structures fabricated by selective laser sintering: TPU powder evaluation and process optimization, Mater. Des., 2017, vol. 120, pp. 317–327.
Von Schnering, H.G. and Nesper, R., How nature adapts chemical structures to curved surfaces, Angew. Chem. Int. Ed., 1987, vol. 26, no. 11, pp. 1059–1080.
Perdew, J.P., Burke, K., and Ernzerhof, M., Generalized gradient approximation made simple, Phys. Rev. Lett., 1996, vol. 77, no. 18, pp. 3865–3868.
Giannozzi, P., Baroni, S., Bonini, N., Calandra, M., Car, R., Cavazzoni, C., Ceresoli, D., Chiarotti, G.L., Cococcioni, M., Ismaila Dabo, A., dal Corso, S., de Gironcoli, Fabris, S., Fratesi, G., Gebauer, R., et al., QUANTUM ESPRESSO: a modular and open-source software project for quantum simulations of materials, J. Phys.: Condens. Matter, 2009, vol. 21, no. 39, p. 395502.
Pack, J.D. and Monkhorst, H.J., Special points for Brillouin zone integrations, Phys. Rev. B, 1977, vol. 16, no. 4, pp. 1748–1749.
Momma, K. and Izumi, F., VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data, J. Appl. Crystallogr., 2011 vol. 44, no. 6, pp. 1272–1276.
Ishii, A., Yamamoto, M., Asano, H., and Fujiwara, K., DFT calculation for adatom adsorption on graphene sheet as a prototype of carbon nanotube functionalization, J. Phys.: Conf. Ser., 2008, vol. 100, no. 5, p. 52087.
Arsentev, M.Y., Petrov, A.V., Missyul, A.B., and Hammouri, M., Exfoliation, point defects and hydrogen storage properties of monolayer TiS3: An ab initio study, RSC Adv., 2018, vol. 8, no. 46, pp. 26169–26179.
Zhang, Y., Wu, X., Tao, Y., Mao, C., and Zhu, J., Fabrication and field-emission performance of zirconium disulfide nanobelt arrays, Chem. Commun., 2008, no. 23, pp. 2683–2685.
Pickard, C.J. and Needs, R.J., Ab initio random structure searching, J. Phys.: Condens. Matter, 2011, vol. 23, no. 5, pp. 0953–8984.
Liu, P., Zhang, H., Cheng, X., and Tang, Y., External electric field: An effective way to prevent aggregation of Mg atoms on γ-graphyne for high hydrogen storage capacity, Appl. Surf. Sci., 2016, vol. 371, pp. 44–49.
Arsentev, M., Hammouri, M., Missyul, A., and Petrov, A., Complex interaction of hydrogen with the monolayer TiS2 decorated with Li and Li2O clusters: An ab initio random structure searching approach, Int. J. Hydrogen Energy, 2019, vol. 44, no. 39, pp. 21988–21998.
Funding
The study was supported by a grant from the Russian Science Foundation (project no. 20-73-10171).
CONFLICT OF INTERESTThe authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalinina, M.V., Arsent’ev, M.Y. & Balabanov, S.V. Study of Hydrogen Adsorption on a Monolayer Tis3 Decorated with Mg Cations. Glass Phys Chem 47, 270–275 (2021). https://doi.org/10.1134/S1087659621030044
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1087659621030044