Abstract
Objective
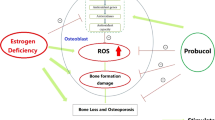
Osteoporosis is affecting the health of postmenopausal women in the world. In case of that, we explored whether FK-506 could ameliorate osteoporosis by inhibiting the activated CaN/NFAT pathway during oxidative stress.
Methods
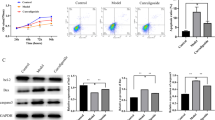
First, the castrated rat model is constructed through the bilateral ovariectomy. Hologic Discovery (S/N 80347) dual-energy X-ray absorptiometry assessed bone mineral density (BMD) implemented at left femur of rats. Next, hematoxylin–eosin (H&E) staining observed and calculated the changes of bone trabecular, mean trabecular plate separation (Tb.Sp), mean trabecular plate thickness (Tb.Th), and bone volume fraction (BV/TV). Then, CCK-8 assay, TUNEL assay, ALP kit and alizarin red staining detected the viability, apoptosis, alkaline phosphatase (ALP) activity, and capacity of mineralization respectively. At last, commercially available kits detected the levels of ROS and SOD in transfected MC3T3-E1 cells and bone tissues, and Western blot analysis detected proteins related to apoptosis and CaN/NFAT pathway.
Results
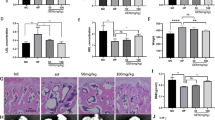
FK-506 increased the BMD and changes of bone trabecular in female castrated rats. FK-506 inhibited the oxidative stress and apoptosis by suppressing the activated CaN/NFAT pathway. Low dose of FK-506 improved the viability, ALP activity, and mineralization capacity. What’s more, it suppressed the apoptosis of H2O2-induced MC3T3-E1 cells, which was deteriorated by the high dose of FK-506. Briefly, low dose of FK-506 inhibited the oxidative stress by suppressing the activated CaN/NFAT pathway, while high dose of that further inhibited the oxidative stress by suppressing the CaN/NFAT pathway.
Conclusion
FK-506 ameliorates osteoporosis resulted from osteoblastic apoptosis which caused by suppressing the activated CaN/NFAT pathway during oxidative stress.





Similar content being viewed by others
References
Okubo R, Sanada LS, Castania VA, Louzada MJ, de Paula FJ, Maffulli N, et al. Jumping exercise preserves bone mineral density and mechanical properties in osteopenic ovariectomized rats even following established osteopenia. Osteoporos Int. 2017;28:1461–71.
Price CT, Koval KJ, Langford JR. Silicon: a review of its potential role in the prevention and treatment of postmenopausal osteoporosis. Int J Endocrinol. 2013;2013:316783.
Kerschan-Schindl K, Mikosch P, Obermayer-Pietsch B, Gasser RW, Dimai HP, Fahrleitner-Pammer A, et al. Current controversies in clinical management of postmenopausal osteoporosis. Exp Clin Endocrinol Diabetes. 2014;122:437–44.
Gambacciani M, Levancini M. Management of postmenopausal osteoporosis and the prevention of fractures. Panminerva Med. 2014;56:115–31.
Griffith JP, Kim JL, Kim EE, Sintchak MD, Thomson JA, Fitzgibbon MJ, et al. X-ray structure of calcineurin inhibited by the immunophilin-immunosuppressant FKBP12-FK506 complex. Cell. 1995;82:507–22.
Liu J, Farmer JD, Lane WS, Friedman J, Weissman I, Schreiber SL. Calcineurin is a common target of cyclophilin-cyclosporin A and FKBP-FK506 complexes. Cell. 1991;66:807–15.
Parson WH, Sigal NH, Wyvratt MJ. FK506-A novel immunosuppressant. Immunomodulating Drugs. 1993;685:22–36.
Crabtree GR, Schreiber SL. SnapShot: Ca2+-calcineurin-NFAT signaling. Cell. 2009;138(210):210.e1.
Ho S, Clipstone N, Timmermann L, Northrop J, Graef I, Fiorentino D, et al. The mechanism of action of Cyclosporin A and FK506. Clin Immunol Immunopathol. 1996;80:S40–5.
Mognol GP, Carneiro FRG, Robbs BK, Faget DV, Viola JPB. Cell cycle and apoptosis regulation by NFAT transcription factors: new roles for an old player. Cell Death Dis. 2016;7:e2199.
Shou J, Jing J, Xie J, You L, Jing Z, Yao J, et al. Nuclear factor of activated T cells in cancer development and treatment. Cancer Lett. 2015;361:174–84.
Sharma S, Findlay G, Bandukwala H, Oberdoerffer S, Baust B, Li Z, et al. Dephosphorylation of the nuclear factor of activated T cells (NFAT) transcription factor is regulated by an RNA-protein scaffold complex. Proc Natl Acad Sci USA. 2011;108:11381–6.
Kaminuma O, Kitamura N, Mori A, Tatsumi H, Nemoto S, Hiroi T. NFAT1 and NFAT2 differentially regulate IL-17A expression in human T cells. Int Arch Allergy Immunol. 2012;158:30–4.
Junkichi K, Nobuo I, Megumi F, Taketoshi S, Noriaki Y, Hideaki ET, et al. Effects of the calcineurin inhibitors cyclosporine and tacrolimus on bone metabolism in rats. Biomed Res. 2018;39:131–9.
Martín-Fernández M, Rubert M, Montero M, De la Piedra C. Effects of cyclosporine, tacrolimus, and rapamycin on osteoblasts. Transplant Proc. 2017;49:2219–24.
Andia DC, Nassar CA, Nassar PO, Guimarães MR, Cerri PS, Spolidorio LC. Treatment with tacrolimus enhances alveolar bone formation and decreases osteoclast number in the maxillae: a histomorphometric and ultrastructural study in rats. Histol Histopathol. 2008;23:1177–84.
Manolagas S. From estrogen-centric to aging and oxidative stress: a revised perspective of the pathogenesis of osteoporosis. Endocr Rev. 2010;31:266–300.
Almeida M, O’Brien C. Basic biology of skeletal aging: role of stress response pathways. J Gerontol A Biol Sci Med Sci. 2013;68:1197–208.
Lijuan G, Xiaomin L, Yuan X, Zhi W, Junyi W. The effect of oxidative stress on intracellular free Ca~(2+) and CaM expression in hair cells. Chin J Ind Med. 2017;30:6–8.
Bing Q, Chun-E L, Xiao-Kang T, Dian-Shuai G, Zhi-An L, Anatomy DO. Expression of Calcium/calmodulin-dependent kinase II in the hippocampus of Alzheimer’s disease-like rats and its relationship with cell apoptosis. J Shandong Univ (Health ences). 2014;52:44–7.
Liu H, Xiong Y, Wang H, Yang L, Wang C, Liu X, et al. Effects of water extract from epimedium on neuropeptide signaling in an ovariectomized osteoporosis rat model. J Ethnopharmacol. 2018;221:126–36.
Yulan L, Hongyan XU, Sen L. Effect of Bushen Juanbi decoction combined with western medicine on bone metabolism and levels of oxidative stress products in postmenopausal osteoporosis. Heb J Trad Chin Med. 2019;41:827–32.
Yin H, Shi ZG, Yu YS, Hu J, Wang R, Luan ZP, et al. Protection against osteoporosis by statins is linked to a reduction of oxidative stress and restoration of nitric oxide formation in aged and ovariectomized rats. Eur J Pharmacol. 2012;674:200–6.
Baek KH, Oh KW, Lee WY, Lee SS, Kim MK, Kwon HS, et al. Association of oxidative stress with postmenopausal osteoporosis and the effects of hydrogen peroxide on osteoclast formation in human bone marrow cell cultures. Calcif Tissue Int. 2010;87:226–35.
Coipeau P, Rosset P, Langonne A, Gaillard J, Delorme B, Rico A, et al. Impaired differentiation potential of human trabecular bone mesenchymal stromal cells from elderly patients. Cytotherapy. 2009;11:584–94.
Almeida M, Ambrogini E, Han L, Manolagas SC, Jilka RL. Increased lipid oxidation causes oxidative stress, increased peroxisome proliferator-activated receptor-gamma expression, and diminished pro-osteogenic Wnt signaling in the skeleton. J Biol Chem. 2009;284:27438–48.
Cervellati C, Bonaccorsi G, Cremonini E, Romani A, Fila E, Castaldini MC, et al. Oxidative stress and bone resorption interplay as a possible trigger for postmenopausal osteoporosis. Biomed Res Int. 2014;2014:569563.
Wu Q, Zhong ZM, Pan Y, Zeng JH, Zheng S, Zhu SY, et al. Advanced oxidation protein products as a novel marker of oxidative stress in postmenopausal osteoporosis. Med Sci Monit. 2015;21:2428–32.
Wilson C. Oxidative stress and osteoporosis. Nat Rev Endocrinol. 2014;10:1–3.
Rachner TD, Khosla S, Hofbauer L. New horizons in osteoporosis. Lancet. 2011;377:1276–8.
Li X, Zhang XL, Shen G, Tang GH. Effects of tensile forces on serum deprivation-induced osteoblast apoptosis: expression analysis of caspases, Bcl-2, and Bax. Chin Med J (Engl). 2012;125:2568–73.
Takayanagi H. Mechanistic insight into osteoclast differentiation in osteoimmunology. J Mol Med (Berl). 2005;83:170–9.
Yeo H, Beck LH, McDonald JM, Zayzafoon M. Cyclosporin A elicits dose-dependent biphasic effects on osteoblast differentiation and bone formation. Bone. 2007;40:1502–16.
Li M, Zhu Z, Liu F, Wang G, Mao Y, Liu M, et al. Effect of blockade of calcineurin/nuclear factor of activated T cells paths by VIVIT peptide on inhibition of osteoprogenitor differentiation by polymethylmethacrylate particles. Chin J Orthop Trauma. 2010;12:156–61.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no competing interests.
Additional information
Communicated by John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jie, J., Li, W., Wang, G. et al. FK506 ameliorates osteoporosis caused by osteoblast apoptosis via suppressing the activated CaN/NFAT pathway during oxidative stress. Inflamm. Res. 70, 789–797 (2021). https://doi.org/10.1007/s00011-021-01452-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-021-01452-3