Abstract
Introduction
African Animal Trypanosomiasis (AAT) or nagana in animals, is caused by the blood-borne parasitic protozoa called trypanosomes, and is potentially fatal. It is estimated that Africa loses $4‒5 billion annually due to the death of livestock to nagana in the tsetse belt.
Purpose
Although The Gambia lies within this belt, there is scanty data regarding the epizootiology of nagana in The Gambia. Here, records of reported cases of nagana for the period 2010–2019 at the International Trypanotolerance Centre (ITC) in The Gambia were analyzed retrospectively.
Methods
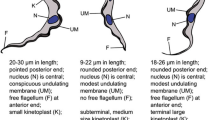
For insights into the current prevalence of AAT, blood samples of 384 cattle, 42 goats, and 59 sheep from the Central River Region (CRR) and Lower River Region (LRR) were analyzed microscopically for parasite identification. Furthermore, trypanosomes were characterized by polymerase chain reaction (PCR) using a panel of primers that identify trypanosomes to the level of the species and subspecies by targeting a portion of the internally transcribed spacer-one (ITS-1) of the ribosomal RNA.
Results
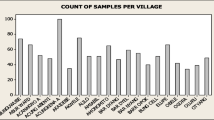
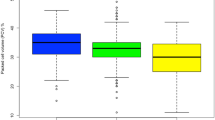
The retrospective study indicates that Trypanosoma vivax (66%) and T. congolense (33.4%) were the predominant species. Based on the archive records of ITC, the villages Touba, Misera, and Sambel Kunda all in the CRR of the Gambia are the most burdened with AAT. Microscopic examination of blood samples from cattle showed a prevalence of 1.56%, whereas the PCR-based analysis gave a higher prevalence of 12.5%. The molecular analysis revealed the presence of T. vivax (3.65%), T. congolense kilifi (2.6%), T. b. brucei (1.3%), T. congolense savannah/forest (0.52%), T. b. gambiense (0.52%). Interestingly, 4.43% of mixed infections i.e. multiple trypanosome species in individual animals were recorded. In 18% of the mixed infection cases, T. godfreyi, T. simiae were coinfecting cattle alongside T. congolense. The molecular identification including the phylogenetic analysis implicated T. congolense as the most predominant trypanosome species infecting animals in The Gambia.
Conclusion
The incidence of nagana in The Gambia is documented and the prevalent trypanosomes identified to be T. vivax, different types of T. congolense, and T. brucei including the gambiense subspecie. Finally, nagana is less profound in sheep and goats compared to cattle, with seasonal and regional variations playing a significant role in the disease dynamics.







Similar content being viewed by others
References
Black SJ, Mansfield JM (2016) Prospects for vaccination against pathogenic African trypanosomes. Parasite Immunol 38(12):735–743. https://doi.org/10.1111/pim.12387
Abebe G (2005) Trypanosomosis in ethiopia. Ethiop J Biol Sci 4(1):75–121
Samdi SM, Fajinmi AO, Kalejaye JO, Wayo B, Haruna MK, Yarnap JE, Mshelia WP, Usman AO, Hamra SM, Jijitar A, Ogunwole R (2011) Prevalence of trypanosomosis in cattle at slaughter in Kaduna central abattoir. Asian J Animal Sci 5(2):162–165
Ng’ayo MO, Njiru ZK, Kenya EU, Muluvi GM, Osir EO, Masiga DK (2005) Detection of trypanosomes in small ruminants and pigs in western Kenya: important reservoirs in the epidemiology of sleeping sickness? Kinetoplastid Biol Dis 4(1):1–7. https://doi.org/10.1186/1475-9292-4-5
Aksoy S (2002) Tsetse Vector Based Strategies for Control of African Trypanosomiasis. In The African Trypanosomes, Springer, Boston, MA, pp 39-49
Solomon NK, Idris LA, Oluseyi OO (2016) Silent human Trypanosoma brucei gambiense infections around the old Gboko sleeping sickness focus in Nigeria. J Parasitol Res. https://doi.org/10.1155/2016/2656121
Swallow BM (1999) Impact of trypanosomiasis on African agriculture. PAAT Technical and Scientific Series 2. PAAT Information Service. Food and Agriculture Organisation, United Nations, Rome, 2000, 1–46. https://hdl.handle.net/10568/50693. http://www.fao.org/3/X4755EN/x4755en.pdf
Technical Centre for Agricultural and Rural Cooperation, CTA (1990) Animal traction for agricultural development. In: Starkey P, Faye A (Eds.), Proceedings of the Third Workshop of the West Africa Animal Traction Network, 7–12 July 1988, Saly, Senegal. CTA, Wageningen, pp 1-495. https://hdl.handle.net/10568/45402
Snow WF, Wacher TJ, Rawlings P (1996) Observations on the prevalence of trypanosomosis in small ruminants, equines and cattle, in relation to tsetse challenge. Gambia Vet Parasitol 66(1–2):1–11. https://doi.org/10.1016/S0304-4017(96)01003-5
Snow W, Boreham PF (1979) The feeding habits and ecology of the tsetse fly Glossina morsitans submorsitans Newstead in relation to nagana transmission in The Gambia. Acta Trop 36(1):47–51. https://doi.org/10.5169/seals-312507
Rawlings P, Ceesay ML, Wacher TJ, Snow WF (1993) The distribution of the tsetse flies Glossina morsitans submorsitans and G palpalis gambiensis (Diptera: Glossinidae) in The Gambia and the application of survey results to tsetse and trypanosomiasis control. Bull Entomol Res 83(4):625–632
Dhollander S, Jallow A, Mbodge K, Kora S, Sanneh M, Gaye M, Bos JFFP, Leak S, Berkvens D, Geerts S (2006) Equine trypanosomosis in the Central River Division of The Gambia: a study of veterinary gate-clinic consultation records. Prev Vet Med 75(3–4):152–162. https://doi.org/10.1016/j.prevetmed.2005.11.009
Pinchbeck GL, Morrison LJ, Tait A, Langford J, Meehan L, Jallow S, Jallow J, Jallow A, Christley RM (2008) Trypanosomosis in The Gambia: prevalence in working horses and donkeys detected by whole genome amplification and PCR, and evidence for interactions between trypanosome species. BMC Vet Res 4(1):7. https://doi.org/10.1186/1746-6148-4-7
McNamara JJ, Snow WF (1990) Improved identification of Nannomonas infections in tsetse flies from The Gambia. Acta Trop 48(2):127–136. https://doi.org/10.1016/0001-706X(90)90052-2
Osaer S, Goossens B, Jeffcoate I, Holmes P (1998) Effects of Trypanosoma congolense and nutritional supplements in Djallonke ewes on live weight during pregnancy, post-partum weight, haematology parameters and lamb performance. Res Vet Sci 65(1):65–69. https://doi.org/10.1016/S0034-5288(98)90029-7
Brown T (2015) The N’Dama dilemma: ethnogenetics and small ruminant breed dynamics in the tsetse zone. Gambia Outlook Agric 44(3):173–178. https://doi.org/10.5367/oa.2015.0210
Kargbo A, Kuye RA (2020) Epidemiology of tsetse flies in the transmission of trypanosomiasis: technical review of The Gambia experience. Int J Biol Chem Sci 14(3):1093–1102. https://doi.org/10.4314/ijbcs.v14i3.35
Adams ER, Malele II, Msangi AR, Gibson WC (2006) Trypanosome identification in wild tsetse populations in Tanzania using generic primers to amplify the ribosomal RNA ITS-1 region. Acta Trop 100(1–2):103–109. https://doi.org/10.1016/j.actatropica.2006.10.002
Majekodunmi AO, Fajinmi A, Dongkum C, Picozzi K, Thrusfield MV, Welburn SC (2013) A longitudinal survey of African animal trypanosomiasis in domestic cattle on the Jos Plateau, Nigeria: prevalence, distribution and risk factors. Parasit Vectors 6(1):239. https://doi.org/10.1186/1756-3305-6-239
Murray M, Murray PK, McIntyre WIM (1977) An improved parasitological technique for the diagnosis of African trypanosomiasis. Trans R Soc Trop Med Hyg 71(4):325–326. https://doi.org/10.1016/0035-9203(77)90110-9
Chagas CR, Binkienė R, Ilgūnas M, Iezhova T, Valkiūnas G (2020) The buffy coat method: a tool for detection of blood parasites without staining procedures. Parasit Vectors 13(1):1–12. https://doi.org/10.1186/s13071-020-3984-8
Picozzi K, Tilley A, Fevre EM, Coleman PG, Magona JW, Odiit M, Eisler MC, Welburn SC (2002) The diagnosis of trypanosome infections: applications of novel technology for reducing disease risk. Afr J Biotech 1(2):39–45. https://doi.org/10.5897/AJB2002.000-007
Enwezor FNC, Samdi SM, Ijabor O, Abenga JN, (2012) The prevalence of bovine trypanosomes in parts of Benue state, north-central Nigeria. J Vector Borne dis 49(3):188. http://mrcindia.org/journal/issues/493188.pdf
Claxton JR, Leperre P, Rawlings P, Snow WF, Dwinger RH (1992) Trypanosomiasis in cattle in Gambia: Incidence, prevalence and tsetse challenge. Acta Trop 50(3):219–225. https://doi.org/10.1016/0001-706X(92)90078-C
Takeet MI, Fagbemi BO, De Donato M, Yakubu A, Rodulfo HE, Peters SO, Wheto M, Imumorin IG (2013) Molecular survey of pathogenic trypanosomes in naturally infected Nigerian cattle. Res Vet Sci 94(3):555–561. https://doi.org/10.1016/j.rvsc.2012.10.018
Faye D, de Almeida PJP, Goossens B, Osaer S, Ndao M, Berkvens D, Speybroeck N, Nieberding F, Geerts S (2001) Prevalence and incidence of trypanosomosis in horses and donkeys in The Gambia. Vet Parasitol 101(2):101–114. https://doi.org/10.1016/S0304-4017(01)00503-9
Traub RJ, Monis PT, Robertson ID (2005) Molecular epidemiology: a multidisciplinary approach to understanding parasitic zoonoses. Int J Parasitol 35(11–12):1295–1307. https://doi.org/10.1016/j.ijpara.2005.06.008
Abdullahi AL, Balogun EO, Yusuf AB, Adepoju OA, Ibrahim B, Gouegni F, Habila AJ, Atawodi SE, Shuaibu MN, Mamman M, Nok AJ (2020) Blood of African Hedgehog Atelerix albiventris Contains 115-kDa Trypanolytic Protein that Kills Trypanosoma congolense. Acta Parasitol 65:733–742
Tiberti N, Hainard A, Sanchez JC (2013) Translation of human African trypanosomiasis biomarkers towards field application. Transl Proteomic 1(1):12–24. https://doi.org/10.1016/j.trprot.2013.04.001
Simo G, Mbida JAM, Eyenga VEO, Asonganyi T, Njiokou F, Grébaut P (2014) Challenges towards the elimination of Human African Trypanosomiasis in the sleeping sickness focus of Campo in southern Cameroon. Parasit Vectors 7(1):374. https://doi.org/10.1186/1756-3305-7-374
Neau P, Hänel H, Lameyre V, Strub-Wourgaft N, Kuykens L (2020) Innovative partnerships for the elimination of Human African Trypanosomiasis and the development of fexinidazole. Tropic Med Infect Dis 5(1):17. https://doi.org/10.3390/tropicalmed5010017
Njiokou F, Nimpaye H, Simo G, Njitchouang GR, Asonganyi T, Cuny G, Herder S (2010) Domestic animals as potential reservoir hosts of Trypanosoma brucei gambiense in sleeping sickness foci in Cameroon. Parasite 17(1):61–66. https://doi.org/10.1051/parasite/2010171061
Weny G, Okwee-Acai J, Okech SG, Tumwine G, Ndyanabo S, Abigaba S, Goldberg TL (2017) Prevalence and risk factors associated with hemoparasites in cattle and goats at the edge of Kibale National Park, western Uganda. J Parasitol 103(1):69–74. https://doi.org/10.1645/16-33
Akazue PI, Ebiloma GU, Ajibola O, Isaac C, Onyekwelu K, Ezeh CO, Eze AA (2019) Sustainable Elimination (Zero Cases) of Sleeping Sickness: How Far Are We from Achieving This Goal? Pathogens 8(3):135. https://doi.org/10.3390/pathogens8030135
Muturi CN, Ouma JO, Malele II, Ngure RM, Rutto JJ, Mithöfer KM, Enyaru J, Masiga DK (2011) Tracking the feeding patterns of tsetse flies (Glossina genus) by analysis of bloodmeals using mitochondrial cytochromes genes. PLoS ONE 6(2):e17284
Kagbadouno MS, Camara M, Rouamba J, Rayaisse JB, Traore IS, Camara O, Onikoyamou MF, Courtin F, Ravel S, De Meeûs T, Bucheton B (2012) Epidemiology of sleeping sickness in Boffa (Guinea): where are the trypanosomes? PLoS Negl Trop Dis 6(12):e1949
McNamara JJ, Mohammed G, Gibson WC (1994) Trypanosoma (Nannomonas) godfreyi sp. nov. from tsetse flies in The Gambia: biological and biochemical characterization. Parasitology 109(4):497–509. https://doi.org/10.1017/S0031182000080756
Yaro M, Munyard KA, Stear MJ, Groth DM (2016) Combatting African animal Trypanosomiasis (AAT) in livestock: the potential role of trypanotolerance. Vet Parasitol 225:43–52. https://doi.org/10.1016/j.vetpar.2016.05.003
Balogun EO, Nok AJ, Kita K (2016) Global warming and the possible globalization of vector-borne diseases: a call for increased awareness and action. Trop med health 44(1):38. https://doi.org/10.1186/s41182-016-0039-0
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 70 for bigger datasets. Mol biol evol 33(7):1870–1874. https://doi.org/10.1093/molbev/msw054
Acknowledgements
We want to appreciate the effort of The Gambia Bureau of Statistics, the Department of Livestock Services, and the International Trypanotolerant Center in The Gambia for their invaluable contribution to this work. Finally, we thank Dr. Idowu Aimola and the Late Dr. T.T Gbem both from Ahmadu Bello University, Zaria, Nigeria for their helpful comments and assistance in the course of this work.
Funding
This work was supported with funding through the Africa Center of Excellence for Development (ACE Impacts) project to Africa Center of Excellence for Neglected Tropical disease and forensic Biotechnology (ACENTDFB), Ahmadu Bello University, Nigeria, and in part, by the Africa Center of Excellence project under the ministry of higher education science and technology, The Gambia.
Author information
Authors and Affiliations
Contributions
AK, EOB performed the experiments; MJ help in the data collection; AK, GUE, and EOB performed data analysis and wrote the manuscript; YKE, EOB, and GDC directed the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Ethical statement
Ethical approval was given by Department of Livestock Services, Ministry of Agriculture of the Republic of The Gambia, and also from Ahmadu Bello University, Zaria, Ethical Committee on Animal Use and care (ABUCAUC). All animals used in this study were handled Veterinarians based on ethical guidelines on the use of animals for research purpose as stipulated by the Ministry of Agriculture, The Gambia and ABUCAUC.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kargbo, A., Ebiloma, G.U., Ibrahim, Y.K.E. et al. Epizootiology and Molecular Identification of Trypanosome Species in Livestock Ruminants in the Gambia. Acta Parasit. 67, 130–142 (2022). https://doi.org/10.1007/s11686-021-00442-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11686-021-00442-z