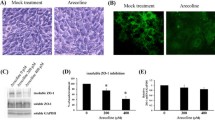
Abstract
Zearalenone (ZEA) and Deoxynivalenol (DON) are two mycotoxins highly detected in agricultural products and feed. Both mycotoxins produce reproductive toxicity and pose a serious threat to human and animal health, among which pigs are the most sensitive animals. Sertoli cells (SCs) play an important role in spermatogenesis; however, the combined toxicity of ZEA and DON and the screening of effective protective agents remains to be determined. By studying the effects of N-acetylcysteine (NAC) on the cells exposed to 20 μM of ZEA and 0.6 μM of DON, we explored the protective mechanism of NAC (4 mM) on the cytotoxic injury of piglets SCs induced by both mycotoxins. The results showed that the combination of ZEA and DON destroy organelles and SCs structures, NAC significantly alleviates the damage caused by ZEA and DON. NAC also significantly increased the expression and distribution of zonula occludens 1 (ZO-1), decreased the relative mRNA and protein expression levels of Bax, Bid, caspase-3, and caspase-9, and increased Bcl-2 expression level and inhibited the decrease of mitochondrial membrane potential. Further, NAC also eases the cell cycle arrest and oxidative stress caused by ZEA and DON. In summary, our results show that NAC could alleviate SCs injury via reducing the oxidative damage and apoptosis caused by ZEA and DON.
Graphical abstract







Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the study are available from the corresponding author on reasonable request.
Abbreviations
- BCA:
-
bicinchoninic acid
- CAT:
-
catalase
- DCFH-DA:
-
2,7-Dichlorodi-hydrofluorescein diacetate
- DON:
-
deoxynivalenol
- ELISA:
-
enzyme-linked immunosorbent assay
- FBS:
-
fetal bovine serum
- GSH-Px:
-
glutathione peroxidase
- MDA:
-
malondialdehyde
- MMP:
-
mitochondrial membrane potential
- NAC:
-
N-acetylcysteine
- PVDF:
-
polyvinylidene fluoride
- ROS:
-
reactive oxygen species
- SDS-PAGE:
-
sodium dodecyl sulfate polyacrylamide gel electrophoresis
- SOD:
-
superoxide dismutase
- ZEA:
-
zearalenone
- ZO-1:
-
zonula occludens 1.
References
Borutova R, Faix S, Placha I, Gresakova L, Cobanova K, Leng L (2008) Effects of deoxynivalenol and zearalenone on oxidative stress and blood phagocytic activity in broilers. Arch Anim Nutr 62:303–312. https://doi.org/10.1080/17450390802190292
Cao L, Zhao J, Chen L et al (2020a) Effects of combined exposure of zearalenone and deoxynivalenol on reproductive toxicity of male mice and alleviation of tea polyphenols. Chinese J Anim Nutr 32(04):1946–1956. https://doi.org/10.3969/j.issn.1006-267x.2020.04.055
Cao Z, Huang W, Sun Y, Li Y (2020b) Deoxynivalenol induced spermatogenesis disorder by blood-testis barrier disruption associated with testosterone deficiency and inflammation in mice. Environ Pollut 264:114748. https://doi.org/10.1016/j.envpol.2020.114748
Chang SP, Shen SC, Lee WR, Yang LL, Chen YC (2011) Imatinib mesylate induction of ROS-dependent apoptosis in melanoma B16F0 cells. J Dermatol Sci 62:183–191. https://doi.org/10.1016/j.jdermsci.2011.03.001
Chen S, Yang S, Wang M, Chen J, Huang S, Wei Z, Cheng Z, Wang H, Long M, Li P (2020) Curcumin inhibits zearalenone-induced apoptosis and oxidative stress in Leydig cells via modulation of the PTEN/Nrf2/Bip signaling pathway. Food Chem Toxicol 141:111385. https://doi.org/10.1016/j.fct.2020.111385
Cheng CY, Mruk DD (2002) Cell junction dynamics in the testis: sertoli-germ cell interactions and male contraceptive development. Physiol Rev 82:825–874. https://doi.org/10.1152/physrev.00009.2002
Cortinovis C, Caloni F, Schreiber NB, Spicer LJ (2014) Effects of fumonisin B1 alone and combined with deoxynivalenol or zearalenone on porcine granulosa cell proliferation and steroid production. Theriogenology 81:1042–1049. https://doi.org/10.1016/j.theriogenology.2014.01.027
Döll S, Dänicke S (2011) The Fusarium toxins deoxynivalenol (DON) and zearalenone (ZON) in animal feeding. Prev Vet Med 102:132–145. https://doi.org/10.1016/j.prevetmed.2011.04.008
Ezraty B, Gennaris A, Barras F, Collet JF (2017) Oxidative stress, protein damage and repair in bacteria. Nat Rev Microbiol 15:385–396. https://doi.org/10.1038/nrmicro.2017.26
Fanning AS, Jameson BJ, Jesaitis LA, Anderson JM (1998) The tight junction protein ZO-1 establishes a link between the transmembrane protein occludin and the actin cytoskeleton. J Biol Chem 273:29745–29753. https://doi.org/10.1074/jbc.273.45.29745
Habrowska-Gne protein occludin and the actin cytos al (2019) Deoxynivalenol modulates the viability, ROS production and apoptosis in prostate cancer cells. Toxins (Basel) 11. doi:https://doi.org/10.3390/toxins11050265
Jia X, Xu Y, Wu W, Fan Y, Wang G, Zhang T, Su W (2017) Aroclor1254 disrupts the blood-testis barrier by promoting endocytosis and degradation of junction proteins via p38 MAPK pathway. Cell Death Dis 8:e2823. https://doi.org/10.1038/cddis.2017.224
Jiang YP, Ye RJ, Yang JM, Liu N, Zhang WJ, Ma L, Sun T, Niu JG, Zheng P, Yu JQ (2020) Protective effects of Salidroside on spermatogenesis in streptozotocin induced type-1 diabetic male mice by inhibiting oxidative stress mediated blood-testis barrier damage. Chem Biol Interact 315:108869. https://doi.org/10.1016/j.cbi.2019.108869
Kiang DT, Kennedy BJ, Pathre SV, Mirocha CJ (1978) Binding characteristics of zearalenone analogs to estrogen receptors. Cancer Res 38:3611–3615
Lee ES, Kim HM, Kang JS, Lee EY, Yadav D, Kwon MH, Kim YM, Kim HS, Chung CH (2016) Oleanolic acid and N-acetylcysteine ameliorate diabetic nephropathy through reduction of oxidative stress and endoplasmic reticulum stress in a type 2 diabetic rat model. Nephrol Dial Transplant 31:391–400. https://doi.org/10.1093/ndt/gfv377
Li D, Ye Y, Lin S, Deng L, Fan X, Zhang Y, Deng X, Li Y, Yan H, Ma Y (2014) Evaluation of deoxynivalenol-induced toxic effects on DF-1 cells in vitro: cell-cycle arrest, oxidative stress, and apoptosis. Environ Toxicol Pharmacol 37:141–149. https://doi.org/10.1016/j.etap.2013.11.015
Liu M, Gao R, Meng Q, Zhang Y, Bi C, Shan A (2014a) Toxic effects of maternal zearalenone exposure on intestinal oxidative stress, barrier function, immunological and morphological changes in rats. PLoS One 9:e106412. https://doi.org/10.1371/journal.pone.0106412
Liu X, Nie S, Huang D, Xie M (2014b) Effects of nonylphenol exposure on expression of cell receptors and secretory function in mouse Sertoli TM4 cells. Environ Toxicol Pharmacol 37:608–616. https://doi.org/10.1016/j.etap.2014.01.017
Long M, Yang S, Dong S, Chen X, Zhang Y, He J (2017) Characterization of semen quality, testicular marker enzyme activities and gene expression changes in the blood testis barrier of Kunming mice following acute exposure to zearalenone. Environ Sci Pollut Res Int 24:27235–27243. https://doi.org/10.1007/s11356-017-0299-1
Ma Y, Zhang A, Shi Z, He C, Ding J, Wang X, Ma J, Zhang H (2012) A mitochondria-mediated apoptotic pathway induced by deoxynivalenol in human colon cancer cells. Toxicol in Vitro 26:414–420. https://doi.org/10.1016/j.tiv.2012.01.010
Mailloux RJ (2015) Teaching the fundamentals of electron transfer reactions in mitochondria and the production and detection of reactive oxygen species. Redox Biol 4:381–398. https://doi.org/10.1016/j.redox.2015.02.001
Park CJ, Lee JE, Oh YS, Shim S, Kim DM, Park NC, Park HJ, Gye MC (2011) Postnatal changes in the expression of claudin-11 in the testes and excurrent ducts of the domestic rabbit (Oryctolagus cuniculus domesticus). J Androl 32:295–306. https://doi.org/10.2164/jandrol.110.010611
Ren Z, Deng H, Deng Y, Liang Z, Deng J, Zuo Z, Hu Y, Shen L, Yu S, Cao S (2017) Combined effects of deoxynivalenol and zearalenone on oxidative injury and apoptosis in porcine splenic lymphocytes in vitro. Exp Toxicol Pathol 69:612–617. https://doi.org/10.1016/j.etp.2017.05.008
Shi B, Su Y, Chang S, Sun Y, Meng X, Shan A (2017) Vitamin C protects piglet liver against zearalenone-induced oxidative stress by modulating expression of nuclear receptors PXR and CAR and their target genes. Food Funct 8:3675–3687. https://doi.org/10.1039/c7fo01301a
Su C, Lee K, Chen M et al (2016) Cantharidin induced oral squamous cell carcinoma cell apoptosis via the JNK-regulated mitochondria and endoplasmic Reticulum stress-related signaling pathways. PLoS One 11(12):e0168095. https://doi.org/10.1371/journal.pone.0168095
Syed V, Hecht N (2002) Disruption of germ cell-sertoli-cell interactions leads to spermatofenic defects. Mol Cell Endocrinol 186(2):155–157. https://doi.org/10.1016/s0303-7207(01)00656-6
Taranu I, Braicu C, Marin DE, Pistol GC, Motiu M, Balacescu L, Beridan Neagoe I, Burlacu R (2015) Exposure to zearalenone mycotoxin alters in vitro porcine intestinal epithelial cells by differential gene expression. Toxicol Lett 232:310–325. https://doi.org/10.1016/j.toxlet.2014.10.022
Tatay E, Espaicu C, Marin DE et al (2015) Exposure to zearalenone mycotoxin alterbance of antioxidant capacity by zearalenone and its metabolites in human cells. Toxicol in Vitro 45:334–339. https://doi.org/10.1016/j.tiv.2017.04.026
Tiemann U, Viergutz T, Jonas L, Schneider F (2003) Influence of the mycotoxins alpha- and beta-zearalenol and deoxynivalenol on the cell cycle of cultured porcine endometrial cells. Reprod Toxicol 17:209–218. https://doi.org/10.1016/s0890-6238(02)00141-7
Venkataramana M, Chandra Nayaka S, Anand T, Rajesh R, Aiyaz M, Divakara ST, Murali HS, Prakash HS, Lakshmana Rao PV (2014) Zearalenone induced toxicity in SHSY-5Y cells: the role of oxidative stress evidenced by N-acetyl cysteine. Food Chem Toxicol 65:335–342. https://doi.org/10.1016/j.fct.2013.12.042
Wang X, Zhang Y, Zhao J, Cao L, Zhu L, Huang Y, Chen X, Rahman SU, Feng S, Li Y, Wu J (2019a) Deoxynivalenol induces inflammatory injury in IPEC-J2 cells via NF-C-Jsignaling pathway. Toxins (Basel) 11. https://doi.org/10.3390/toxins11120733
Wang X, Yu H, Fang H, Zhao Y, Jin Y, Shen J, Zhou C, Zhou Y, Fu Y, Wang J, Zhang J (2019b) Transcriptional profiling of zearalenone-induced inhibition of IPEC-J2 cell proliferation. Toxicon 172:8–14. https://doi.org/10.1016/j.toxicon.2019.10.004
Wang X, Chu X, Cao L, Zhao J, Zhu L, Rahman SU, Hu Z, Zhang Y, Feng S, Li Y, Wu J (2020) The role and regulatory mechanism of autophagy in hippocampal nerve cells of piglet damaged by deoxynivalenol. Toxicol in Vitro 66:104837. https://doi.org/10.1016/j.tiv.2020.104837
Wen Q, Tang EI, Xiao X, Gao Y, Chu DS, Mruk DD, Silvestrini B, Cheng CY (2016) Transport of germ cells across the seminiferous epithelium during spermatogenesis-the involvement of both actin- and microtubule-based cytoskeletons. Tissue Barriers 4:e1265042. https://doi.org/10.1080/21688370.2016.1265042
Xiao Y, Xu S, Zhao S, Liu K, Lu Z, Hou Z (2019) Protective effects of selenium against zearalenone-induced apoptosis in chicken spleen lymphocyte via an endoplasmic reticulum stress signaling pathway. Cell Stress Chaperones 24:77–89. https://doi.org/10.1007/s12192-018-0943-9
Xu X, Yan G, Chang J, Wang P, Yin Q, Liu C, Liu S, Zhu Q, Lu F (2020) Astilbin ameliorates deoxynivalenol-induced oxidative stress and apoptosis in intestinal porcine epithelial cells (IPEC-J2). J Appl Toxicol 40:1362–1372. https://doi.org/10.1002/jat.3989
Yang M, Wu X, Zhang W, Ye P, Wang Y, Zhu W, Tao Q, Xu Y, Shang J, Zhao D, Ding Y, Yin Z, Zhang X (2020) Transcriptional analysis of deoxynivalenol-induced apoptosis of sow ovarian granulosa cell. Reprod Domest Anim 55:217–228. https://doi.org/10.1111/rda.13610
Yu JY, Zheng ZH, Son YO, Shi X, Jang YO, Lee JC (2011) Mycotoxin zearalenone induces AIF- and ROS-mediated cell death through p53- and MAPK-dependent signaling pathways in RAW264.7 macrophages. Toxicol in Vitro 25:1654–1663. https://doi.org/10.1016/j.tiv.2011.07.002
Zheng W, Pan S, Wang G, Wang YJ, Liu Q, Gu JH, Yuan Y, Liu XZ, Liu ZP, Bian JC (2016) Zearalenone impairs the male reproductive system functions via inducing structural and functional alterations of sertoli cells. Environ Toxicol Pharmacol 42:146–155. https://doi.org/10.1016/j.etap.2016.01.013
Zheng W, Wang B, Si M, Zou H, Song R, Gu J, Yuan Y, Liu X, Zhu G, Bai J, Bian J, Liu ZP (2018) Zearalenone altered the cytoskeletal structure via ER stress- autophagy- oxidative stress pathway in mouse TM4 Sertoli cells. Sci Rep 8(1):3320. https://doi.org/10.1038/s41598-018-21567-8
Zheng W, Feng N, Wang Y, Noll L, Xu S, Liu X, Lu N, Zou H, Gu J, Yuan Y, Liu X, Zhu G, Bian J, Bai J, Liu Z (2019) Effects of zearalenone and its derivatives on the synthesis and secretion of mammalian sex steroid hormones: a review. Food Chem Toxicol 126:262–276. https://doi.org/10.1016/j.fct.2019.02.031
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (grant No. 31472250), the Project of Modern Agricultural Industry and Technology System of Anhui Province (grant No. AHCYJSTX-05-07), and the Graduate student innovation fund of Anhui Agricultural University (grant No. 2021yjs-16). We wish to thank anonymous reviewers for their kind advice.
Author information
Authors and Affiliations
Contributions
Li Cao: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Resources, Software, Visualization, Writing-original Draft, Writing-Review and editing. Jie Zhao: Data Curation, Formal Analysis, Investigation, Resources, Validation, Visualization, Writing-Review, and Editing. Jingru Xu: Data Curation, Formal Analysis, Resources, Writing-Review, and Editing. Lei Zhu: Data Curation, Resources. Sajid ur Rahman: Writing-Review and Editing, Data Curation, Resources. Shibin Feng: Project Administration, Resources. Yu Li: Project Administration, Resources. Jinjie Wu: Project Administration, Resources. Xichun Wang*: Conceptualization, Formal Analysis, Funding Acquisition, Investigation, Methodology, Project Administration, Resources, Software, Validation, Visualization, Writing-Review, and Editing.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare that they have no conflicts of interest.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cao, L., Zhao, J., Xu, J. et al. N-acetylcysteine ameliorate cytotoxic injury in piglets sertoli cells induced by zearalenone and deoxynivalenol. Environ Sci Pollut Res 28, 60276–60289 (2021). https://doi.org/10.1007/s11356-021-14052-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-14052-9