Abstract
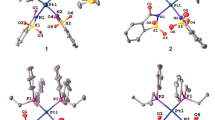
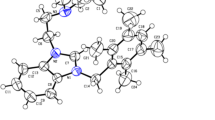
A series of 1,4-disubstituted digold–alkynyl complexes, [(PPh3)Au]2(μ-C≡C–Ar–CH≡C)], in which the 1,4-diethenylbenzene bridge contains two alkyl or oligo(ethylene glycol)methylether side chains at different positions (2,5- and 2,3-positions), have been synthesized and structurally characterized by spectroscopic techniques. The structure of [(AuPPh3)2{µ-C≡C–C6H2(OC6H13)2-2,5-C≡C}] (2c) has been established by X-ray crystallography. Their antitumor activities in vitro were investigated by MTT assay against four tumor cell lines. Compared to alkyl-functionalized complexes, the oligo(ethylene glycol)methylether-functionalized complexes showed significant antitumor activity, which may be attributed to the improvement of the hydrophilicity of the complex to achieve the appropriate lipophilicity. In particular, complex [(AuPPh3)2{µ-C≡C–C6H2(OCH2CH2OCH2CH2OCH3)2-2,3-C≡C}] (2j) displayed high activity against the A549 cell line and low cytotoxicity toward normal cell lines. Notably, further investigations are required if complex 2j is to undergo trials as a new potential antitumor agent.


Similar content being viewed by others
References
Wai-Yin Sun R, Che CM (2009) Coord Chem Rev 253:1682–1691
Ott I (2009) Coord Chem Rev 253:1670–1681
Che CM, Sun RWY (2011) Chem Commun 47:9554–9560
Zou T, Lum CT, Chui SS, Che CM (2013) Angew Chem Int Ed 52:2930–2933
Zou T, Lum CT, Lok C, To W, Low K, Che CM (2014) Angew Chem Int Ed 53:5810–5814
Bindoli A, Rigobello MP, Scutari G, Gabbiani C, Casini A, Messori L (2009) Coord Chem Rev 253:1692–1707
Meyer A, Bagowski CP, Kokoschka M, Stefanopoulou M, Alborzinia H, Can S, Vlecken DH, Sheldrick WS, Wöfl S, Ott I (2012) Angew Chem Int Ed 51:8895–8899
Ma DL, Shum TYT, Zhang F, Che CM, Yang M (2005) Chem Commun 37:4675–4677
Zhang JJ, Abuel Maaty MA, Hoffmeister H, Schmidt C, Muenzner JK, Schobert R, Wçlfl S, Ott I (2020) Angew Chem Int Ed 59:16795–16800
Andermark V, Goke K, Kokoschka M, Abuel Maaty MA, Lum CT, Zou T, Sun RWY, Aguiló E, Oehninger L, Rodríguez L, Bunjes H, Wölfl S, Che CM, Ott I (2016) J Inorg Biochem 160:140–148
Meyer A, Gutiérrez A, Ott I, Rodríguez L (2013) Inorg Chim Acta 398:72–76
Arcau J, Andermark V, Aguiló E, Gandioso A, Moro A, Cetina M, Lima JC, Rissanen K, Ott I, Rodríguez L (2014) Dalton Trans 43:4426–4436
Zhang D, Xu Z, Yuan J, Zhao YX, Qiao ZY, Gao YJ, Yu GA, Li J, Wang H (2014) J Med Chem 57:8132–8139
Nisi AD, Bergamini C, Leonzio M, Sartor G, Fato R, Naldi M, Monari M, Calonghi N, Bandini M (2016) Dalton Trans 45:1546–1553
Balasingham RG, Williams CF, Mottram HJ, Coogan MP, Pope SJA (2012) Organometallics 31:5835–5843
Vanesa FM, Marzob I, Gimeno MC (2014) Chem Sci 5:4434–4446
Cottrill SM, Sharma HL, Dyson DB, Parish RV, Auliffe CAM (1989) J Chem Soc Perkin Trans 2:53–56
Chui CH, Wong RSM, Gambari R, Cheng GYM, Yuen MCW, Chan KW, Tong SW, Lau FY, Lai PBS, Lam KH, Ho CL, Kan CW, Leung KSY, Wong WY (2009) Bioorg Med Chem 17:7872–7877
Vergara E, Cerrada E, Casini A, Zava O, Laguna M, Dyson PJ (2010) Organometallics 29:2596–2603
Rubbiani R, Schuh E, Meyer A, Lemke J, Wimberg J, Metzler-Nolte N, Meyer F, Mohr F, Ott I (2013) Med Chem Commun 4:942–948
Hickey JL, Ruhayel RA, Barnard PJ, Baker MV, Berners-Price SJ, Filipovska A (2008) J Am Chem Soc 130:12570–12571
Baron M, Bellemin-Laponnaz S, Tubaro C, Basato M, Bogialli S, Dolmella A (2014) J Inorg Biochem 141:94–102
Bruce MI, Nicholson BK, Bin Shawkataly O (1989) Inorg Synth 26:325–326
Khan MS, Al-Mandhary MRA, Al-Suti MK, Corcoran TC, Al-Mahrooqi Y, Attfield JP, Feeder N, David WIF, Shankland K, Friend RH, Köhler AEAM, Tedesco E, Tang CC, Raithby PR, Collings JC, Roscoe KP, Batsanov AS, Stimson LM, Marder TB (2003) New J Chem 27:140–149
Pelter A, Jones DE (2000) J Chem Soc Perkin Trans 1(14):2289–2294
Zhou CZ, Liu T, Xu JM, Chen ZK (2003) Macromolecules 36:1457–1464
Tian LY, Liu YM, Tian GX, Wu XH, Li Z, Kou JF, Ou YP, Liu SH, Fu WF (2014) Dalton Trans 43:4093–4101
Zhu Z, Swager TM (2001) Org Lett 3:3471–3474
Sheldrick GM SHELXS–97, A Program for Crystal Structure Solution, Götingen, Germany, 1997.
Sheldrick GM SHELXL–97, A Program for Crystal Structure Refinement, Götingen, Germany, 1997.
Mosmann T (1983) J Immunol Methods 65:55–63
Acknowledgements
The authors acknowledge financial support from Natural Science Foundation of Yunnan Province (No. 202001AT070068).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
CCDC 1,019,402 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Rights and permissions
About this article
Cite this article
Chen, Q.M., Chen, Y. & Wu, X.H. Synthesis and antitumor activity of novel alkynyl (phosphine) digold(I) complexes. Transit Met Chem 46, 495–501 (2021). https://doi.org/10.1007/s11243-021-00467-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-021-00467-3