Abstract
Background
Cloning and characterizing the drought-inducible promoters is essential for their use in crop resistance's genetic improvement. Previous studies have shown that the TaNRX1-D gene participates in regulating the response of wheat to drought stress. However, its promoter has not yet been identified.
Objective
In this study, we aimed to characterize the promoter of the TaNRX1-D gene.
Methods
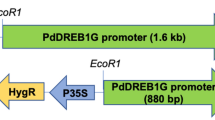
The promoter of TaNRX1-D (named P0, 2081 bp) was isolated from common wheat with several cis-acting elements that regulate in response to abiotic stresses and some core cis-acting elements. Functional verification of the promoter, eight 5′-deletion fragments of TaNRX1-D promoter, was fused to the β-glucuronidase (GUS) gene P0::GUS ~ P7::GUS and transformed into Arabidopsis, respectively. Agrobacterium-mediated GUS transient assay the P6a and P6b promoter regions in tobacco leaves under normal, osmotic or ABA stress.
Results
Activity analysis of the full-length promoter (P0) showed that the intensity of stronger β-glucuronidase (GUS) staining in the roots and leaves was obtained during the growth of transgenic Arabidopsis. P0::GUS displayed the GUS activity was much higher in the roots and leaves than in other parts of the transgenic plant under normal conditions, which was similarly within wheat. Analysis of the 5′-deletion fragments revealed that P0::GUS ~ P6::GUS responded well upon exposure to osmotic (polyethylene glycol-6000, PEG6000) and abscisic acid (ABA) stress treatments and expressed significantly higher GUS activity than the CaMV35S promoter (35S::GUS), while P7::GUS did not. GUS transient assay in tobacco leaves showed that the GUS activities of P6a and P6b were lower than P6 in the PEG6000 and ABA stresses.
Conclusion
The 193 bp (P6) segment was considered the core region of TaNRX1-D responding to PEG6000 or ABA treatment. GUS activity assay in transgenic Arabidopsis showed that this segment was sufficient for the PEG6000 or ABA stress response. The identified 193 bp promoter of TaNRX1-D in this study will help breed osmotic or ABA tolerant crops. The 36 bp segment between P6 and P6b (−193 to −157 bp) was considered the critical sequence for the TaNRX1-D gene responding to PEG6000 or ABA treatment.









Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- PEG 6000:
-
Polyethene glycol-6000
- GUS:
-
β-Glucuronidase
- CaMV35S:
-
Cauliflower mosaic virus 35S
- TRX:
-
Thioredoxin
- NRX:
-
Nucleotoreoxin
- PCR:
-
Polymerase chain reaction
- DMSO:
-
Dimethyl sulfoxide
- X-Gluc:
-
5-Bromo-4-chloro-3-indolyl-β-d-glucuronic acid cyclohexylammonium salt
- 4-MUG:
-
4-Methyl-umbelliferyl-β-d-glucuronide
References
Alomari DZ, Eggert K, Von Wirén N, Alqudah AM, Polley A, Plieske J, Ganal MW, Pillen K (2018) Identifying candidate genes for enhancing grain Zn concentration in wheat. Front Plant Sci 9:1313–1324. https://doi.org/10.3389/fpls.2018.01313
Arnér ES, Holmgren A (2000) Physiological functions of thioredoxin and thioredoxin reductase. Eur J Biochem 267:6102–6109. https://doi.org/10.1046/j.1432-1327.2000.01701.x
Basak P, Sangma S, Mukherjee A, Agarwal T, Sengupta S, Ray S, Majumder AL (2018) Functional characterization of two myo-inositol-1-phosphate synthase (MIPS) gene promoters from the halophytic wild rice (Porteresia coarctata). Planta 248:1121–1141. https://doi.org/10.1007/s00425-018-2957-z
Bihmidine S, Lin J, Stone JM, Awada T, Specht JE, Clemente TE (2013) Activity of the Arabidopsis RD29A and RD29B promoter elements in soybean under water stress. Planta 237:55–64. https://doi.org/10.1007/s00425-012-1740-9
Christensen AH, Sharrock RA, Quail PH (1992) Maize polyubiquitin genes: structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Mol Biol 18:675–689. https://doi.org/10.1007/bf00020010
Cornejo MJ, Luth D, Blankenship KM, Anderson OD, Blechl AE (1993) Activity of a maize ubiquitin promoter in transgenic rice. Plant Mol Biol 23:567–581. https://doi.org/10.1007/bf00019304
Fang Y, Xiong L (2015) General mechanisms of drought response and their application in drought resistance improvement in plants. Cell Mol Life Sci 72:673–689. https://doi.org/10.1007/s00018-014-1767-0
Funato Y, Miki H (2007) Nucleoredoxin, a novel thioredoxin family member involved in cell growth and differentiation. Antioxid Redox Signal 9:1035–1057. https://doi.org/10.1089/ars.2007.1550
Grill E, Himmelbach A (1998) ABA signal transduction. Curr Opin Plant Biol 1:412–418. https://doi.org/10.1016/s1369-5266(98)80265-3
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucleic Acids Res 27:297–300. https://doi.org/10.1093/nar/27.1.297
Hill HD, Straka JG (1988) Protein determination using bicinchoninic acid in the presence of sulfhydryl reagents. Anal Biochem 170:203–208. https://doi.org/10.1016/0003-2697(88)90109-1
Hu H, Xiong L (2014) Genetic engineering and breeding of drought-resistant crops. Annu Rev Plant Biol 65:715–741. https://doi.org/10.1146/annurev-arplant-050213-040000
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907. https://doi.org/10.1002/j.1460-2075.1987.tb02730.x
Kaplan B, Davydov O, Knight H, Galon Y, Knight MR, Fluhr R, Fromm H (2006) Rapid transcriptome changes induced by cytosolic Ca2+ transients reveal ABRE-related sequences as Ca2+-responsive cis-elements in Arabidopsis. Plant Cell 18:2733–2748. https://doi.org/10.1105/tpc.106.042713
Khan S, Anwar S, Yu S, Sun M, Yang Z, Gao ZQ (2019) Development of drought-tolerant transgenic wheat: achievements and limitations. Int J Mol Sci 20:3350–3368. https://doi.org/10.3390/ijms20133350
Koia J, Moyle R, Hendry C, Lim L, Botella JR (2013) Pineapple translation factor SUI1 and ribosomal protein L36 promoters drive constitutive transgene expression patterns in Arabidopsis thaliana. Plant Mol Biol 81:327–336. https://doi.org/10.1007/s11103-012-0002-3
Laughner BJ, Sehnke PC, Ferl RJ (1998) A novel nuclear member of the thioredoxin superfamily. Plant Physiol 118:987–996. https://doi.org/10.1104/pp.118.3.987
Lee BH, Zhu JK (2010) Phenotypic analysis of Arabidopsis mutants: germination rate under salt/hormone-induced stress. Cold Spring Harb Protoc 4:4969–4973. https://doi.org/10.1101/pdb.prot4969
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, Van de Peer Y, Rouzé P, Rombauts S (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327. https://doi.org/10.1093/nar/30.1.325
Li F, Han Y, Feng Y, Xing S, Zhao M, Chen Y, Wang W (2013) Expression of wheat expansion driven by the RD29 promoter in tobacco confers water-stress tolerance without impacting growth and development. J Biotechnol 163:281–291. https://doi.org/10.1016/j.jbiotec.2012.11.008
Li YB, Han LB, Wang HY, Zhang J, Sun ST, Feng DQ, Yang CL, Sun YD (2016) The thioredoxin GbNRX1 plays a crucial role in homeostasis of apoplastic reactive oxygen species in response to verticillium dahliae infection in cotton. Plant Physiol 170:2392–2406. https://doi.org/10.1104/pp.15.01930
Liu W, Mazarei M, Rudis MR, Fethe MH, Stewart CN Jr (2011) Rapid in vivo analysis of synthetic promoters for plant pathogen phytosensing. BMC Biotechnol 11:108–116. https://doi.org/10.1186/1472-6750-11-108
Liu H, Zhu K, Tan C, Zhang J, Zhou J, Jin L, Ma G, Zou Q (2019) Identification and characterization of PsDREB2 promoter involved in tissue-specific expression and abiotic stress response from Paeonia suffruticosa. Peer J 7:7052–7075. https://doi.org/10.7717/peerj.7052
Marchal C, Delorme-Hinoux V, Bariat L, Siala W, Belin C, Saez-Vasquez J, Riondet C, Reichheld JP (2014) NTR/NRX define a new thioredoxin system in the nucleus of Arabidopsis thaliana cells. Mol Plant 7:30–44. https://doi.org/10.1093/mp/sst162
Mata-Perez C, Spoel SH (2019) Thioredoxin-mediated redox signalling in plant immunity. Plant Sci 279:27–33. https://doi.org/10.1016/j.plantsci.2018.05.001
Msanne J, Lin J, Stone JM, Awada T (2011) Characterization of abiotic stress-responsive Arabidopsis thaliana RD29A and RD29B genes and evaluation of transgenes. Planta 234:97–107. https://doi.org/10.1007/s00425-011-1387-y
Mundy J, Yamaguchi-Shinozaki K, Chua NH (1990) Nuclear proteins bind conserved elements in the abscisic acid-responsive promoter of a rice rab gene. Proc Natl Acad Sci USA 87:1406–1410. https://doi.org/10.1073/pnas.87.4.1406
Niu X, Adams CC, Workman JL, Guiltinan MJ (1996) Binding of the wheat basic leucine zipper protein EmBP-1 to nucleosomal binding sites is modulated by nucleosome positioning. Plant Cell 8:1569–1587. https://doi.org/10.1105/tpc.8.9.1569
Odell JT, Nagy F, Chua NH (1985) Identification of DNA sequences required for activity of the cauliflower mosaic virus 35S promoter. Nature 313:810–812. https://doi.org/10.1038/313810a0
Porto MS, Pinheiro MP, Batista VG, dos Santos RC, Filho Pde A, de Lima LM (2014) Plant promoters: an approach of structure and function. Mol Biotechnol 56:38–49. https://doi.org/10.1007/s12033-013-9713-1
Potenza C, Aleman L, Sengupta-Gopalan C (2004) Targeting transgene expression in research, agricultural, and environmental applications: promoters used in plant transformation. In Vitro Cell Dev Biol-Plant 40:1–22. https://doi.org/10.1079/IVP2003477
Schnurr JA, Guerra DJ (2000) The CaMV35S promoter is sensitive to shortened photoperiod in transgenic tobacco. Plant Cell Rep 19:279–282. https://doi.org/10.1007/s002990050012
Simpson SD, Nakashima K, Narusaka Y, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2003) Two different novel cis-acting elements of erd1, a clpA homologous Arabidopsis gene function in induction by dehydration stress and dark-induced senescence. Plant J 33:259–270. https://doi.org/10.1046/j.1365-313x.2003.01624.x
Sun Q, Gao F, Zhao L, Li K, Zhang J (2010) Identification of a new 130 bp cis-acting element in the TsVP1 promoter involved in the salt stress response from Thellungiella halophila. BMC Plant Biol 10:90–102. https://doi.org/10.1186/1471-2229-10-90
Tavakol E, Sardaro ML, Shariati JV, Rossini L, Porceddu E (2014) Isolation, promoter analysis and expression profile of Dreb2 in response to drought stress in wheat ancestors. Gene 549:24–32. https://doi.org/10.1016/j.gene.2014.07.020
Tian S, Cheng J, Yang J, Li R, Zhang Y, Hao X, Guo Y, Zhang X (2019) Interaction analysis of TaNRX1 with TaPDI, TaTRX-h and TaPP2Ac and drought resistance study. JAB 27:1901–1911. https://doi.org/10.3969/j.issn.1674-7968.2019.11.001
Wang J, Li Q, Mao X, Li A, Jing R (2016) Wheat transcription factor taareb3 participates in drought and freezing tolerances in Arabidopsis. Int J Biol Sci 12:257–269. https://doi.org/10.7150/ijbs.13538
Xu LC, Thali M, Schaffner W (1991) Upstream box/TATA box order is the major determinant of the direction of transcription. Nucleic Acids Res 19:6699–6704. https://doi.org/10.1093/nar/19.24.6699
Xu Z, Wang M, Guo Z, Zhu X, Xia Z (2019) Identification of a 119-bp promoter of the maize sulfite oxidase gene (ZmSO) that confers high-level gene expression and aba or drought inducibility in transgenic plants. Int J Mol Sci 20:3326–3340. https://doi.org/10.3390/ijms20133326
Yang Y, Al-Baidhani HHJ, Harris J, Riboni M, Li Y, Mazonka I, Bazanova N, Chirkova L, Sarfraz Hussain S, Hrmova M, Haefele S, Lopato S, Kovalchuk N (2020) DREB/CBF expression in wheat and barley using the stress-inducible promoters of HD-Zip I genes: impact on plant development, stress tolerance and yield. Plant Biotechnol J 18:829–844. https://doi.org/10.1111/pbi.13252
Yuan XB, Chang YN, Wang MM, Fan YD, Wang PT, Wu YJ, Li MY, Zhang L, Meng FR, Li YC (2019) Sequence characteristic and expression patterns of a nucleoredoxin gene TaNRX1 in wheat. J Triticeae Crops 39:1139–1145. https://doi.org/10.7606/j.issn.1009-1041.2019.10.01
Zavallo D, Lopez Bilbao M, Hopp HE, Heinz R (2010) Isolation and functional characterization of two novel seed-specific promoters from sunflower (Helianthus annuus L.). Plant Cell Rep 29:239–248. https://doi.org/10.1007/s00299-010-0816-x
Zhang CJ, Guo Y (2012) OsTRXh1 regulates the redox state of the apoplast and influences stress responses in rice. Plant Signal Behav 7:440–442. https://doi.org/10.4161/psb.19244
Zhang X, Henriques R, Lin SS, Niu QW, Chua NH (2006) Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat Protoc 1:641–646. https://doi.org/10.1038/nprot.2006.97
Zhang F, Jiang L, Ju LP, Jin XF, Wang X, Zhang XK, Wang HL, Fu XJ (2014) Cloning a novel gene TaNRX of trx superfamily and developing its molecular markers related to drought resistance in common wheat. Acta Agron Sin 40:29–36. https://doi.org/10.3724/sp.J.1006.2014.00029
Zhang H, Hou J, Jiang P, Qi S, Xu C, He Q, Ding Z, Wang Z, Zhang K, Li K (2016) Identification of a 467 bp promoter of maize phosphatidylinositol synthase gene (ZmPIS) which confers high-level gene expression and salinity or osmotic stress inducibility in transgenic tobacco. Front Plant Sci 7:42–54. https://doi.org/10.3389/fpls.2016.00042
Zhang H, Jing R, Mao X (2017) Functional characterization of TaSnRK2.8 promoter in response to abiotic stresses by deletion analysis in transgenic Arabidopsis. Front Plant Sci 8:1198–1207. https://doi.org/10.3389/fpls.2017.01198
Acknowledgements
This study was sponsored by the National Natural Science Foundation of China (31671693), Key Research and Development Project of Shaanxi Province (2019ZDLNY04-05), The CAS "Light of West China" Program, The Funds for Local Science and Technology Development Guided by the Central Government (ZY20200089), Cooperation Project of Ningxia Academy of Agriculture and Forestry Science (DWX2019001)
Author information
Authors and Affiliations
Contributions
XZ, JX, YW, and DC provided fund support; XZ conceived the initial screening and research plans; JC and FW performed most of the experiments, analyzed the data, and wrote the initial draft; NL participated the manuscript modification and revisions; TS and MZ helped the data analysis, figures, and tables preparation. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cheng, J., Wei, F., Zhang, M. et al. Identification of a 193 bp promoter region of TaNRX1-D gene from common wheat that contributes to osmotic or ABA stress inducibility in transgenic Arabidopsis. Genes Genom 43, 1035–1048 (2021). https://doi.org/10.1007/s13258-021-01115-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-021-01115-x