Abstract
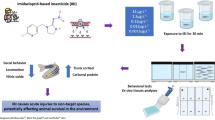
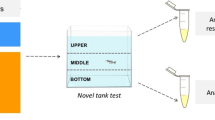
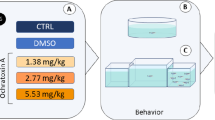
Imidacloprid (IMI) is an insecticide used worldwide, a neonicotinoid that could cause toxicity in non-target organisms. Zebrafish (Danio rerio) is a model organism widely used in different fields of research such as behavioral studies, biochemical parameters as well as neurotoxicity research. Here, we investigate whether the exposure to three concentrations (0.15, 15, and 45 μg/L) of IMI for 96 h alters responses in zebrafish. Oxidative stress parameters and acetylcholinesterase activity (AChE) as well as the behavioral responses of locomotion were measured. IMI exposure decreased distance traveled in fish exposed to the 45 μg/L. In the exploratory activity, time spent and transitions to the top area of the water column decreased in fish exposed to all concentrations of IMI. In addition, exposures to 45 and 15 μg/L of IMI decreased episodes of erratic movement in the zebrafish. Exposures to IMI at a concentration of 45 μg/L decreased the time spent in erratic movements and increased the time spent with no movement (i.e., “freezing”). Glutathione S-transferase (GST) activity was increased in the brain of zebrafish exposed for 96 h to concentrations of 0.15 and 45 μg/L. Brain AChE activity was reduced and the levels of carbonyl protein (CP) increased in brain of zebrafish at concentrations of 15 and 45 μg/L. Lipid peroxidation measured by TBARS and, also non-protein thiols (NPSH) did not show any variation in the brain of zebrafish exposed to IMI. Changes in the activity of cholinergic neurotransmitters in the brain tissues of zebrafish indicate IMI toxicity. Exposures of fish over 96 h to IMI at a nominal concentration of 45 μg/L caused more extensive sublethal responses in zebrafish, but this concentration is well above those expected in the aquatic environment. Studies are warranted to evaluate the effects on behavior and biomarker responses in fish exposed over longer periods to IMI at environmentally relevant concentrations.





Similar content being viewed by others
References
Acayaba RD, Albuquerque AF, Ribessi RL, Umbuzeiro GA, Montagner CC (2020) Occurrence of pesticides in waters from the largest sugar cane plantation region in the world. Environ Sci Pollut R. https://doi.org/10.1007/s11356-020-11428-1
Alvim TT, Martinez CBR (2019) Genotoxic and oxidative damage in the freshwater teleost Prochilodus lineatus exposed to the insecticides lambda-cyhalothrin and imidacloprid alone and in combination. Mut Res 842:85–93. https://doi.org/10.1016/j.mrgentox.2018.11.011
Amaral AMB, Moura LK, Pellegrin J, Guerra LJ, Cerezer FO, Saibt N, Prestes OD, Zanella R, Loro VL, Clasen B (2020) Seasonal factors driving biochemical biomarkers in two fish species from a subtropical reservoir in southern Brazil: an integrated approach. Environ Pollut 266:115168. https://doi.org/10.1016/j.envpol.2020.115168
Anderson JC, Dubetz C, Palace VP (2015) Neonicotinoids in the Canadian aquatic environment: a literature review on current use products with a focus on fate, exposure, and biological effects. Sci Total Environ 505:409–422. https://doi.org/10.1016/j.scitotenv.2014.09.090
Bartlett AJ, Hedges AM, Intini KD, Brown LR, Maisonneuve FJ, Robinson SA, Gillis PL, de Solla SR (2019) Acute and chronic toxicity of neonicotinoid and butenolide insecticides to the freshwater amphipod, Hyalella azteca. Ecotoxicol Environ Saf 175:215–223. https://doi.org/10.1016/j.ecoenv.2019.03.038
Bass C, Denholm I, Williamson MS, Nauen R (2015) The global status of insect resistance to neonicotinoid insecticides. Pestic Biochem Physiol 121:78–87. https://doi.org/10.1016/j.pestbp.2015.04.004
Beauvais SL, Jones SB, Brewer SK, Little EE (2000) Physiological measures of neurotoxicity of diazinon and malathion to larval rainbow trout (Oncorhynchus mykiss) and their correlation with behavioral measures. Environ Toxicol 19:1875–1880. https://doi.org/10.1002/etc.5620190722
Bridi D, Altenhofen S, Gonzalez JB, Reolon GK, Bonan CD (2017) Glyphosate and roundup® alter morphology and behavior in zebrafish. Toxicology 392:32–39. https://doi.org/10.1016/j.tox.2017.10.007
Blacquière T, Smagghe G, Van Gestel CAM, Mommaerts V (2012) Neonicotinoids in bees: a review on concentrations, side-effects and risk assessment. Ecotoxicology 21:1581–1581. https://doi.org/10.1007/s10646-012-0890-7
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Casida JE (2011) Neonicotinoid metabolism: compounds, substituents, pathways, enzymes, organisms and relevance. J Agric Food Chem 59:2923–2931. https://doi.org/10.1021/jf102438c
Castro TFD, Souza JGS, de Carvalho AFS, Assis IL, Palmieri MJ, Vieira LFA, Marcussi S, Machado MRF, Murgas LDS (2018) Anxiety-associated behavior and genotoxicity found in adult Danio rerio exposed to tebuconazole-based commercial product. Environ Toxicol Pharmacol 62:140–146. https://doi.org/10.1016/j.etap.2018.06.011
Champagne DL, Hoefnagels CCM, Kloet RE, Richardson MK (2010) Translating rodent behavioral repertoire to zebrafish (Danio rerio): relevance for stress research. Behav Brain Res 214:332–342. https://doi.org/10.1016/j.bbr.2010.06.001
Cheghib Y, Chouahda S, Soltani N (2020) Side-effects of a neonicotinoid insecticide (Actara®) on a non-target larvivorous fish Gambusia affinis: growth and biomarkers response. Egypt J Aquat Res 46:167–172. https://doi.org/10.1016/j.ejar.2019.12.007
Clasen B, Loro VL, Murussi CR, Tiecher TL, Moraes B, Zanella R (2018) Bioaccumulation and oxidative stress caused by pesticides in Cyprinus carpio reared in a rice-fish system. Sci Total Environ 626:737–743
Crosby EB, Bailey JM, Oliveri AN, Levin ED (2015) Neurobehavioral impairments caused by developmental imidacloprid exposure in zebrafish. Neurotoxicol Teratol 49:81–90. https://doi.org/10.1016/j.ntt.2015.04.006
Donato FF, Martins ML, Munaretto JS, Prestes OD, Adaime MB, Zanella R (2015) Development of a multiresidue method for pesticide analysis in drinking water by solid phase extraction and determination by gas and liquid chromatography with triple quadrupole tandem mass spectrometry. J Braz Chem Soc 26(10):2077–2087. https://doi.org/10.5935/0103-5053.20150192
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol 186:421–431. https://doi.org/10.1016/0076-6879(90)86135-I
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77. https://doi.org/10.1016/0003-9861(59)90090-6
Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Fontana BD, Mezzomo NJ, Kalueff AV, Rosemberg D (2018) The developing utility of zebrafish models of neurological and neuropsychiatric disorders: a critical review. Exp Neurol 299:157–171. https://doi.org/10.1016/j.expneurol.2017.10.004
Ge W, Yan S, Wang J, Zhu L, Chen A, Wang J (2015) Oxidative stress and DNA damage induced by imidacloprid in zebrafish (Danio rerio). J Agric Food Chem 63(6):1856–1862. https://doi.org/10.1021/jf504895h
Gerlai R, Lahav M, Guo S, Rosenthal A (2000) Drinks like a fish: zebra fish (Danio rerio) as a behavior genetic model to study alcohol effects. Pharmacol Biochem Behav 67:773–782. https://doi.org/10.1016/s0091-3057(00)00422-6
Godfray HCJ, Blacquière T, Field LM, Hails RS, Petrokofsky G, Potts SG, Raine NE, Vanbergen AJ, McLean AR (2014) A restatement of the natural science evidence base concerning neonicotinoid insecticides and insect pollinators. Proc R Soc B 281:20140558. https://doi.org/10.1098/rspb.2014.0558
Gonçalves C, Marins AT, do Amaral AMB, Nunes MEM, Müller TE, Severo E, Feijó A, Rodrigues CCR, Zanella R, Prestes OD, Clasen B, Loro VL (2020) Ecological impacts of pesticides on Astyanax jacuhiensis (Characiformes: Characidae) from the Uruguay river, Brazil. Ecotoxicol Environ Saf 205:111314. https://doi.org/10.1016/j.ecoenv.2020.111314
Gonçalves JFS, Souza TM, Vieira LR, Marchi FC, Nascimento AP, Farias DF (2020b) Toxicity testing of pesticides in zebrafish a systematic review on chemicals and associated toxicological endpoints. Environ Sci Pollut Res Int 27(10):10185–10204. https://doi.org/10.1007/s11356-020-07902-5
Habig WH, Pabst MJ, Jacoby WB (1974) Glutathione S-transferase, the first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Howe K, Clark MD, Torroja CF, Torrance J, Berthelot C, Muffato M et al (2013) The zebrafish reference genome sequence and its relationship to the human genome. Nature 496:498–503. https://doi.org/10.1038/nature12111
Husak VV, Mosiichuk NM, Maksymiv IV, Sluchyk IY, Storey JM, Storey KB, Lushchak V (2014) Histopathological and biochemical changes in goldfish kidney due to exposure to the herbicide sencor may be related to induction of oxidative stress. Aquat Toxicol 155:181–189. https://doi.org/10.1016/j.aquatox.2014.06.020
IBAMA (Brazilian Institute of the Environment and Renewable Natural Resources) (2020). Annual pesticide production, import, export and sales bulletins in Brazil. http://www.ibama.gov.br/agrotoxicos/relatorios-de-comercializacao-de- agrotoxicos#historicodecomercializacao
Jabłońska-Trypuć A (2017) Pesticides as inducers of oxidative stress. React Oxyg Species 3:96–110. https://doi.org/10.20455/ros.2017.823
Jemec A, Tisler T, Drobne D, Sepcic K, Fournier D, Trebse P (2007) Comparative toxicity of imidacloprid, of its commercial liquid formulation and of diazinon to a non-target arthropod, the microcrustacean Daphnia magna. Chemosphere 68(8):1408–1418. https://doi.org/10.1016/j.chemosphere.2007.04.015
Jeschke P, Nauen R, Schindled M, Elbert A (2011) Overview of the status and global strategy for neonicotinoids. J Agric Food Chem 59:2897–2908. https://doi.org/10.1021/jf101303g
Kalueff AV, Gebhardt M, Stewart AM, Cachat JM, Brimmer M, Chawla JS et al (2013) Towards a comprehensive catalog of zebrafish behavior 1.0 and beyond. Zebrafish 10:70–86. https://doi.org/10.1089/zeb.2012.0861
Liu H, Song J, Zhang S, Qu L, Zhao Y, Wu Y, Liu H (2005) Analysis of residues of imidacloprid in tobacco by high-performance liquid chromatography with liquid–liquid partition cleanup. Pest Manag Sci 61(5):511–514. https://doi.org/10.1002/ps.987
Lushchak VI (2016) Contaminant-induced oxidative stress in fish: a mechanistic approach. Fish Physiol Biochem 42:711–747. https://doi.org/10.1007/s10695-015-0171-5
Marins AT, Severo ES, Leitemperger JW, Cerezer C, Müller TE, Costa MD, Weimer GH, Bandeira NMG, Prestes OD, Zanella R, Loro VL (2020) Assessment of river water quality in an agricultural region of Brazil using biomarkers in a native neotropical fish, Astyanax spp. (Characidae). Bull Environ Contam Toxicol 104:575–581. https://doi.org/10.1007/s00128-020-02821-0
Müller TE, Nunes SZ, Silveira A, Loro VL, Rosemberg DB (2017) Repeated ethanol exposure alters social behavior and oxidative stress parameters of zebrafish. Mol Neurobiol 79:105–111. https://doi.org/10.1007/s12035-017-0441-6
Nunes ME, Müller TE, Braga MM, Fontana BD, Quadros VA, Marins A, Rodrigues C, Menezes C, Rosemberg DB, Loro VL (2017) Chronic treatment with paraquat induces brain injury, changes in antioxidant defenses system, and modulates behavioral functions in zebrafish. Mol Neurobiol 54:3925–3934. https://doi.org/10.1007/s12035-016-9919-x
Pamanji R, Bethu MS, Yashwanth B, Leelavathi S, Rao JV (2015) Developmental toxic effects of monocrotophos, an organophosphorus pesticide, on zebrafish (Danio rerio) embryos. Environ Sci Pollut Res 22:7744–7753. https://doi.org/10.1007/s11356-015-4120-8
Paredes JF, Cowan M, López-Olmeda JF, Muñoz-Cueto JA, Sánchez-Vázquez FJ (2019) Daily rhythms of expression in reproductive genes along the brain-pituitary-gonad axis and liver of zebrafish. Comp Biochem Physiol Part A Mol Integr Physiol 231:158–169. https://doi.org/10.1016/j.cbpa.2019.02.017
Parlak V (2018) Evaluation of apoptosis, oxidative stress responses, AChE activity and body malformations in zebrafish (Danio rerio) embryos exposed to deltamethrin. Chemosphere 207:397–403. https://doi.org/10.1016/j.chemosphere.2018.05.112
Parvez S, Raisuddin S (2005) Protein carbonyls: novel biomarkers of exposure to oxidative stress-inducing pesticides in freshwater fish Channa punctate (Bloch). Environ Toxicol Pharmacol 20:112–117. https://doi.org/10.1016/j.etap.2004.11.002
Pearson JN, Patel M (2016) The role of oxidative stress in organophosphate and nerve agent toxicity. Ann N Y Acad Sci 1378:17–24. https://doi.org/10.1111/nyas.13115
Pullaguri N, Nema S, Bhargava Y, Bhargava A (2020) Triclosan alters adult zebrafish behavior and targets acetylcholinesterase activity and expression. Environ Toxicol Pharmacol 75:103311. https://doi.org/10.1016/j.etap.2019.103311
Rahal A, Kumar A, Singh V, Yadav B, Tiwari R, Chakraborty S, Dhama K (2014) Oxidative stress, prooxidants, and antioxidants: the interplay. Biomed Res Int. https://doi.org/10.1155/2014/761264
Rodríguez-Fuentes G, Rubio-Escalante FJ, Norena-Barroso E, Escalante-Herrera KS, Schlenk D (2015) Impacts of oxidative stress on acetylcholinesterase transcription, and activity in embryos of zebrafish (Danio rerio) following Chlorpyrifos exposure. Comp Biochem Physiol C Toxicol Pharmacol 172–173:19–25. https://doi.org/10.1016/j.cbpc.2015.04.003
Rosemberg DB, Rico EP, Mussulini BH, Piato AL, Calcagnotto ME, Bonan CD, Dias RD, Blaser RE, Souza DO, Oliveira DL (2011) Differences in spatio-temporal behavior of zebrafish in the open tank paradigma after a short-period confinement into dark and bright environments. PLoS ONE 6(5):e19397. https://doi.org/10.1371/journal.pone.0019397
Schmidel AJ, Assmann KL, Werlang CC, Bertoncello KT, Francescon F, Rambo CL, Beltrame GM, Calegari D, Batista CB, Blaser RE, Roman Júnior WA, Conterato GMM, Piato AL, Zanatta L, Magro JD, Rosemberg DB (2014) Subchronic atrazine exposure changes defensive behaviour profile and disrupts brain acetylcholinesterase activity of zebrafish. Neurotoxicol Teratol 44:62–69. https://doi.org/10.1016/j.ntt.2014.05.006
Severo ES, Marins AT, Cerezer C, Costa D, Nunes M, Prestes OD, Zanella R, Loro VL (2020) Ecological risk of pesticide contamination in a Brazilian river located near a rural area: a study of biomarkers using zebrafish embryos. Ecotox Environ Safe 190:110071. https://doi.org/10.1016/j.ecoenv.2019.110071
Sillapawattana P, Schäffer A (2017) Effects of imidacloprid on detoxifying enzyme glutathione S-transferase on Folsomia candida (Collembola). Environ Sci Pollut Res 24:11111–11119. https://doi.org/10.1007/s11356-016-6686-1
Simon-Delso N, Amaral-Rogers V, Belzunces LP, Bonmatin JM, Chagnon M et al (2015) Systemic insecticides (neonicotinoids and fipronil): trends, uses, mode of action and metabolites. Environ Sci Pollut Res Int 22:5–34. https://doi.org/10.1007/s11356-014-3470-y
Soydan E, Guler A, Bıyık S, Senturk M, Supuran CT, Ekinci D (2017) Carbonic anhydrase from Apis mellifera: purification and inhibition by pesticides. J Enzym Inhib Med Chem 32(1):47–50. https://doi.org/10.1080/14756366.2016.1232255
Sposito JCV, Montagner CC, Casado M, Navarro-Martín L, Solórzano JCJ, Piña B, Grisolia AB (2018) Emerging contaminants in Brazilian rivers: occurrence and effects on gene expression in zebrafish (Danio rerio) embryos. Chemosphere 209:696–704. https://doi.org/10.1016/j.chemosphere.2018.06.046
Tišler T, Jemec A, Mozetic B, Trebse P (2009) Hazard identification of imidacloprid to aquatic environment. Chemosphere 76(7):907–914. https://doi.org/10.1016/j.chemosphere.2009.05.002
Tomizawa M, Casida JE (2005) Neonicotinoid insecticide toxicology: mechanisms of selective action. Annu Rev Pharmacol Toxicol 45:247–268. https://doi.org/10.1146/annurev.pharmtox.45.120403.095930
Tomizawa M, Casida JE (2011) Neonicotinoid insecticides: highlights of a symposium on strategic molecular designs. J Agr Food Chem 59:2883–2886. https://doi.org/10.1021/jf103856c
Topal A, Alak G, Ozkaraca M, Yeltekin AC, Comaklı S, Acil G, Kokturk M, Atamanalp M (2017) Neurotoxic responses in brain tissues of rainbow trout exposed to imidacloprid pesticide: assessment of 8-hydroxy-2-deoxyguanosine activity, oxidative stress and acetylcholinesterase activity. Chemosphere 175:186–191. https://doi.org/10.1016/j.chemosphere.2017.02.047
Ulrich K, Jakob U (2019) The role of thiols in antioxidant systems. Free Radic Biol Med 140:14–27. https://doi.org/10.1016/j.freeradbiomed.2019.05.035
Valadas J, Mocelin R, Sachett A, Marcon M, Zanette RA, Dallegrave E, Herrmann AP, Piato A (2019) Propiconazole induces abnormal behavior and oxidative stress in zebrafish. Environ Sci Pollut R 26:27808–27815. https://doi.org/10.1007/s11356-019-05977-3
Vieira CED, Perez MR, Acayaba RD, Raimundo CCM, Martinez CBR (2018) DNA damage and oxidative stress induced by imidacloprid exposure in different tissues of the neotropical fish Prochilodus lineatus. Chemosphere 195:125–134. https://doi.org/10.1016/j.chemosphere.2017.12.077
Wang X, Anadón A, Wu Q, Qiao F, Ares I, Martínez-Larrañaga MR, Yuan Z, Martínez MA (2018) Mechanism of neonicotinoid toxicity: impact on oxidative stress and metabolism. Annu Rev Pharmacol Toxicol 58:471–507. https://doi.org/10.1146/annurev-pharmtox-010617-052429
Wintermantel D, Odoux JF, Decourtye A, Henry M, Allier F, Bretagnolle V (2020) Neonicotinoid-induced mortality risk for bees foraging on oilseed rape nectar persists despite EU moratorium. Sci Total Environ 704:135400. https://doi.org/10.1016/j.scitotenv.2019.135400
Wu S, Li X, Liu X, Yang G, An X, Wang Q, Wang Y (2018) Joint toxic effects of triazophos and imidacloprid on zebrafish (Danio rerio). Environ Pollut 235:470–481. https://doi.org/10.1016/j.envpol.2017.12.120
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
The current study was approved by the Ethics Commission on Animal Use of the Federal University of Santa Maria (Process Number: 1777051118).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guerra, L.J., do Amaral, A.M.B., de Quadros, V.A. et al. Biochemical and Behavioral Responses in Zebrafish Exposed to Imidacloprid Oxidative Damage and Antioxidant Responses. Arch Environ Contam Toxicol 81, 255–264 (2021). https://doi.org/10.1007/s00244-021-00865-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-021-00865-9