Abstract
Introduction
Candida species is the most common cause of invasive fungal infection. With the wide variation in species distribution and antifungal susceptibility of causative agents, local epidemiological profiles are needed to provide effective guidelines for the treatment of invasive candidiasis.
Objective
To find out the species distribution and antifungal susceptibilities of Candida strains isolated from patients in an intensive care unit (ICU) of Vietnam.
Methods
All patients in ICU of Vietnam National Hospital of Burn with Candida isolation reported from January 2017 to December 2019 were retrospectively studied. Species identification and antifungal susceptibility testing were performed using VITEK 2 Compact. The identification was reconfirmed by sequencing of the internal transcribed spacer regions when needed.
Results
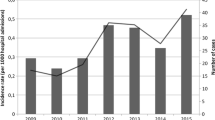
A total of 186 yeasts belonging to ten species were collected. The most common agent was C. tropicalis (45.7%), followed by C. albicans (42.4%), and C. parapsilosis (7.53%). The isolated yeasts showed less susceptibility to fluconazole (resistant rate R 10.7%) than to micafungin, caspofungin, flucytosine and amphotericin B (R 0%, 0.6%, 2.3% and 3.4%, respectively, p < 0.05). C. albicans isolates were more susceptible to fluconazole (R 5.2%) than C. tropicalis (R 15.7%). Resistance to voriconazole was seen only among C. albicans (3.9%) and C. tropicalis isolates (9.9%).
Conclusion
Non-albicans species (especially C. tropicalis) is the predominant species, and there is a significant proportion of isolates with reduced susceptibility to azole but not to echinocandin.


Similar content being viewed by others
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Kullberg BJ, Arendrup MC. Invasive candidiasis. N Engl J Med. 2015;373(15):1445–56.
Enoch DA, Yang H, Aliyu SH, Micallef C. The changing epidemiology of invasive fungal infections. Methods Mol Biol. 2017;1508:17–65.
Struck MF, Gille J. Fungal infections in burns: a comprehensivereview. Ann Burns Fire Disasters. 2013;XXVI(3):147–53.
Cochran A, Morris S, Edelman L, Saffle J. Systemic Candida infection in burn patients: a case-control study of management patterns and outcomes. Surg Infect. 2002;3(4):367–74.
Ballard J, Edelman L, Saffle J, et al. Positive fungal cultures in burn patients: a multicenter review. J Burn Care Res. 2008;29(1):213–21.
Gupta N, Haque A, Lattif AA, et al. Epidemiology and molecular typing of Candida isolates from burn patients. Mycopathologia. 2004;158(4):397–405.
Vrioni G, Matsiota-Bernard P. Molecular typing of Candida isolates from patients hospitalized in an intensive care unit. J Infect. 2001;42(1):50–6.
Guery BTP, Arendrup MC, Auzinger G, et al. Management of invasive candidiasis and candidemia in adult non-neutropenic intensive care unit patients: Part I. Epidemiology and diagnosis. Intensive Care Med. 2009;35(1):55–62.
Brown G, Denning D, Gow N, et al. Hidden killers: human fungal infections. Sci Transl Med. 2012;4(165):165rv13-165rv13.
Hsueh P, Graybill J, Playford E, et al. Consensus statement on the management of invasive candidiasis in intensive care units in the Asia-Pacific Region. Int J Antimicrob Agents. 2009;34(3):205–9.
Tan TY, Hsu LY, Alejandria MM, et al. Antifungal susceptibility of invasive Candida bloodstream isolates from the Asia-Pacific region. Med Mycol. 2016;54(5):471–7.
Bac ND, Anh LT, Quang LB, et al. Prevalence of Candida bloodstream isolates from patients in two hospitals in Vietnam. Iran J Microbiol. 2019;11(2):108–13.
Leaw SN, Chang HC, Sun HF, et al. Identification of medically important yeast species by sequence analysis of the internal transcribed spacer regions. J Clin Microbiol. 2006;44(3):693–9.
CLSI. Reference method for broth dilution antifungal susceptibility testing of yeasts; fourth international supplement. CLSI document M27–4., Wayne, PA: 2012.
CLSI. Reference method for broth dilution antifungal susceptibility testing of yeasts. Approved Standard M27- A3, 3rd edition. Wayne, PA: 2008.
Fothergill AW, Sutton DA, McCarthy DI, Wiederhold NP. Impact of new antifungal breakpoints on antifungal resistance in Candida species. J Clin Microbiol. 2014;52(3):994–7.
Pfaller MA, Diekema DJ. Progress in antifungal susceptibility testing of Candida spp. by use of clinical and laboratory standards institute broth microdilution methods, 2010 to 2012. J Clin Microbiol. 2012;50(9):2846–56.
Pfaller M, Diekema DJ, Gibbs DL, et al. Results from the artemis disk global antifungal surveillance study, 1997 to 2007: A 10.5-year analysis of susceptibilities of candida species to fluconazole and voriconazole as determined by CLSI standardized disk diffusion. J Clin Microbiol. 2010;48(4):1366–77.
Mccarty TP, Pappas PG. Invasive candidiasis. Infect Dis Clin N Am. 2016;30(1):103–24.
Kim SH, Shin JH, Kim E-C, et al. The relationship between antifungal usage and antifungal susceptibility in clinical isolates of Candida: a multicenter Korean study. Med Mycol. 2009;47(3):296–304.
Pfaller MA, Diekema DJ, Procop GW, Rinaldi MG. Multicenter comparison of the VITEK 2 yeast susceptibility test with the CLSI broth microdilution reference method for testing fluconazole against Candida spp. J Clin Microbiol. 2007;45(3):796–802.
Sanguinetti M, Porta R, Sali M, et al. Evaluation of VITEK 2 and RapID yeast plus systems for yeast species identification: experience at a large clinical microbiology laboratory. J Clin Microbiol. 2007;45(4):1343–6.
Pfaller M, Diekema D, Procop G, Rinaldi M. Comparison of the Vitek 2 yeast susceptibility system with CLSI microdilution for antifungal susceptibility testing of fluconazole and voriconazole against Candida spp., using new clinical breakpoints and epidemiological cutoff values. Diagn Microbiol Infect Dis. 2013;77(1):37–40.
Lamoth F, Lockhart SR, Berkow EL, Calandra T. Changes in the epidemiological landscape of invasive candidiasis. J Antimicrob Chemother. 2018;73(suppl_1):i4-13.
Pfaller MA, Diekema DJ, Turnidge JD, et al. Twenty years of the SENTRY antifungal surveillance program: results for candida species from 1997–2016. Open forum Infect Dis. 2019;6(Suppl 1):S79-94.
Sarabahi S, Tiwari VK, Arora S, et al. Changing pattern of fungal infection in burn patients. Burns. 2011;38(4):520–8.
Vallabhaneni S, Mody R, Walker T, Chiller T. The global burden of fungal diseases. Infect Dis Clin North Am. 2016;30(1):1–11.
Yang YL, Cheng HH, Lo HJ, Hospitals Thet. Distribution and antifungal susceptibility of Candida species isolated from different age populations in Taiwan. Med Mycol. 2006;44(3):237–42.
Xiao M, Fan X, Chen SC, et al. Antifungal susceptibilities of Candida glabrata species complex, Candida krusei, Candida parapsilosis species complex and Candida tropicalis causing invasive candidiasis in China: 3 year national surveillance. J Antimicrob Chemother. 2015;70(3):802–10.
Guinea J. Global trends in the distribution of Candida species causing candidemia. Clin Microbiol Infect. 2014;20(Suppl 6):5–10.
Quindós G. Epidemiology of candidaemia and invasive candidiasis A changing face. Rev Iberoam Micol. 2014;31(1):42–8.
Pfaller M, Diekema D, Gibbs D, et al. Results from the ARTEMIS DISK global antifungal surveillance study, 1997 to 2005: an 8.5-year analysis of susceptibilities of Candida species and other yeast species to fluconazole and voriconazole determined by CLSI standardized disk diffusion testing. J Clin Microbiol. 2007;45(6):1735.
Chakrabarti A, Rudramurthy S, Kale P, et al. Epidemiological study of a large cluster of fungaemia cases due to Kodamaea ohmeri in an Indian tertiary care centre. Clin Microbiol Infect. 2014;20(2):O83–9.
Shang S, Lin J, Ho S, et al. The emerging life-threatening opportunistic fungal pathogen Kodamaea ohmeri: optimal treatment and literature review. J Microbiol Immunol Infect. 2010;43(3):200–6.
Tzar M, Shamim A. Candidaemia and antifungal susceptibility testing in a teaching hospital. Med J Malays. 2009;64(1):61–4.
Pappas PG, Kauffman CA, Andes DR, et al. Clinical practice guideline for the management of candidiasis: 2016 update by the infectious diseases society of America. Clin Infect Dis. 2016;62(4):e1-50.
Cleveland A, Farley M, Harrison L, et al. Changes in incidence and antifungal drug resistance in candidemia: results from population-based laboratory surveillance in Atlanta and Baltimore, 2008–2011. Clin Infect Dis. 2012;55(10):1352.
Caggiano G, Coretti C, Bartolomeo N et al. Candida bloodstream infections in Italy: changing epidemiology during 16 years of surveillance. BioMed Res Int. 2015:1–9.
Teo JQ, Candra SR, Lee SJ, et al. Candidemia in a major regional tertiary referral hospital – epidemiology, practice patterns and outcomes. Antimicrob Resist Infect Control. 2017. https://doi.org/10.1186/s13756-017-0184-1.
Zhang L, Zhou S, Pan A, et al. Surveillance of antifungal susceptibilities in clinical isolates of Candida species at 36 hospitals in China from 2009 to 2013. Int J Infect Dis. 2015;33:1–4.
Trouvé C, Blot S, Hayette M, et al. Epidemiology and reporting of candidaemia in Belgium: a multi-centre study. Eur J Clin Microbiol Infect Dis. 2016;36(4):649–55.
Chang T-P, Ho M-W, Yang Y-L, et al. Distribution and drug susceptibilities of Candida species causing candidemia from a medical center in central Taiwan. J Infect Chemother. 2013;19(6):1065–71.
Sanguinetti M, Posteraro B, Lass-Flörl C. Antifungal drug resistance among Candida species: mechanisms and clinical impact. Mycoses. 2015;58(S2):2–13.
Acknowledgements
Not applicable.
Author information
Authors and Affiliations
Contributions
CTS, CBL have contributed equally to this work and acted as joint first authors. CTS, CBL and TAL designed the study, analyzed, interpreted the data and drafted the manuscript. DXQ, NTNM and NNL collected clinical data. DNA, HXS, TAL and TTTH collected the microbiological data and identified species. DQ revised the manuscript critically. All authors read and approved the final manuscript.
Disclosure
This research is part of thesis work for the fulfillment of Doctor of Literature and Philosophy in Health at National Institute of Malaria, Parasitology and Entomology of Vietnam.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for Publication
All of the authors agree to submit the manuscript for possible publication in “Mycopathologia.”
Ethical Approval
The study was approved by the Ethics Committee of Vietnam National Institute of Malariology, Parasitology and Entomology. Written patient consent was not required because of the observational nature of this study.
Additional information
Handling Editor: Damien Costa.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sinh, C.T., Loi, C.B., Minh, N.T.N. et al. Species Distribution and Antifungal Susceptibility Pattern of Candida Recovered from Intensive Care Unit Patients, Vietnam National Hospital of Burn (2017–2019). Mycopathologia 186, 543–551 (2021). https://doi.org/10.1007/s11046-021-00569-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-021-00569-7