Abstract
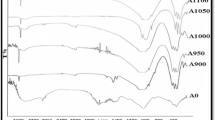
Hydroxyapatite is the most important bio ceramic, due to its structure and chemical composition which is similar to bone and teeth. In this study, hydroxyapatite nanoparticles were prepared from natural teeth in two stages. Initially, the nano HA particles was performed by whole tooth and in the second stage, the tooth was divided into dentine and root and HA nanoparticles were prepared from both portions of teeth. At first, freshly extracted human teeth both (whole tooth and dentine and root) were calcined at 850°C and then, the effects of stirring time, surfactant and different temperatures were investigated. The XRD results confirmed that, the existence of hydroxyapatite phase in all samples. The phosphate bonding groups of \({\text{PO}}_{4}^{{3 - }}\) at 1470 and 669 cm–1 was confirmed by FTIR results. The FESEM results showed that, hydroxyapatite nano particle with the range of 29–46 nm at stirring time of 36 h with the addition of CTAB as a surfactant at 850°C was obtained without agglomeration and good dispersion.













Similar content being viewed by others
REFERENCES
Gunduz, O., Sahin, Y.M., Agathopoulos, S., Ben-Nissan, B., and Oktar, F., A new method for fabrication of nano hydroxyapatite and TCP from the sea snail cerithium vulgatum, J. Nanomater., 2014, vol. 2014, pp. 1–6.
Jamiu, K.O., Danyuo, Y., Abdulazeez Baruwa, D., and Akeem, A.A., Synthesis and characterization of hydroxyapatite from bovine bone for production of dental implants, J. Appl. Biomater. Funct. Mater., 2019, vol. 1, pp. 1–7.
Ivanova, T.I., Frank-Kamenetskaya, O.V., Kol’tsov, A.B., and Ugolkov, V.L., Crystal structure of calcium-deficient carbonated hydroxyapatite, thermal decomposition, J. Solid State Chem., 2001, vol. 160, pp. 340–349.
Kalita, S.J., Bhardwaj, A., and Bhatt, H.A., Nanocrystalline calcium phosphate ceramics in biomedical engineering, Mater. Sci. Eng. C, 2007, vol. 27, pp. 441–449.
Murugan, R. and Ramakrishna, S., Development of nanocomposites for bone grafting, Compos. Sci. Technol., 2005, vol. 65, pp. 2385–2406.
Riman, R.E., Suchanek, W.L., Byrappa, K., Chen, C.W., and Oakes, C., Solution synthesis of hydroxyapatite designer particulates, Solid State Ionics, 2002, vol. 151, pp. 393–402.
Benaqqa, C., Chevalier, J., Saa daoui, M., and Fantozzi, G., Slow crack growth behaviour of hydroxyapatite ceramics, Biomaterials, 2005, vol. 26, pp. 6106–6112.
Orlovskii, V.P., Komlev, V.S., and Barinov, S.M., Hydroxyapatite and hydroxyapatite-based ceramics, Inorg. Mater., 2002, vol. 38, pp. 973–984.
Pasteris, J.D., Lack of oh in nanocrystalline apatite as a function of atomic order: Implications for bone and biomaterials, Biomaterials, 2004, vol. 25, pp. 229–238.
Dorozhkina, E.I., and Dorozhkina, S.V., Mechanism of solid-state transformation of a calcium-deficient hydroxyapatite (CDHA) into biphasic calcium phosphate (BCP) at elevated temperatures, Chem. Mater., 2002, vol.14, pp. 4267–4272.
Vallet-Regi, M., and González-Calbet, J.M., Calcium phosphates as substitution of bone tissues, Progr. Solid State Chem., 2004, vol. 32, pp. 1–31.
Sugiyama, S., Ichii, T., Matsumoto, H., and Hayashi, H., Effect of calcination and sieving of calcium hydroxyapatite on ion-exchangeability with lead cation in the presence and absence of HCl, Adv. Environ. Res., 2002, vol. 62, pp.285–289.
Zhang, H.G., Zhu, Q., and Wang, Y., Morphologically controlled synthesis of hydroxyapatite with partial substitution of fluorine, Chem. Mater., 2005, vol. 17, pp. 5824–5830.
Kannan, S., Rebelo, A., and Ferreira, J.M.F., Novel synthesis and structural characterization of fluorine and chlorine co-substituted hydroxyapatites, J. Inorg. Biochem., 2006, vol. 100, pp. 1692–1697.
Medvecky, L., Stulajterová, R., Parilák L., Trpčevska, J., and Barinov, S.M., Influence of Manganese on Stability and Particle Growth of Hydroxyapatite in Simulated Body Fluid, Colloids Surf. A: Physicochem. Eng. Aspects, 2006, vol. 281, pp. 221–229.
16 Dong, S.S, Jong,K.K., Dissolution of human teeth-derived hydroxyapatite, Annals Biomed. Eng., 2008, vol. 36, no. 1, pp. 132–140.
Ola Saleh, M., Preparation of hydroxyapitate from natural resources literature review, GSJ, 2018, vol. 6, pp. 46–52.
Kim, T.G. and Park, B., Synthesis and growth mechanisms of one-dimensional strontium hydroxyapatite nanostructures, Inorg. Chem., 2005, vol. 44, pp. 9895–9901.
Bhatnagar, V.M., Refinement of the synthetic hydroxyapatite cell parameters, Contr. Mineral. Petrol., 1969, vol. 22, pp. 375–378.
Aoki, H., Kato, K., Ogiso, M., and Tabata, T., Studies on the application of apatite to dental materials, J. Dent. Eng., 1977, vol. 18, pp. 151–156.
Monroe, A.E., Ward, V., Ward, D.B., and McMullen, J., New calcium phosphate ceramic material for bone and tooth implants, J. Dent. Res., 1971, vol. 50, pp. 860–865.
Manafi, S.A., Mirjalili, F., and Reshadi, R., Synthesis and evaluation of the bioactivity of fluorapatite-45s5 bioactive glass nanocomposite, Progr. Biomater., 2019, vol. 8, pp. 77–89.
Zhang, H.G. and Zhu, Q., Surfactant-assited preparation of fluoride-substituted hydroxyapatite nanorods, Mater. Lett., 2005, vol. 59, pp. 3054–3058.
Jones, F.H., Teeth and bones: Application of surface science to dental materials and related biomaterials, Surf. Sci. Rep., 2001, vol. 42, pp.75–205.
Jarcho, M., Kay, J.F., Gumaer, K.I., Doremus, R.H., and Drobeck, H.P., Tissue, cellular, and subcellular events at a bone-ceramic hydroxyapatite interface, J. Bioeng., 1977, vol. 1, pp. 79–92.
Wang, Y., Zhang, S., Wei, K., Zhao, N., Chen, J., and Wang, X., Hydrothermal synthesis of hydroxyapatite nanopowders using cationic surfactant as a template, Mater. Lett., 2006, vol. 60, pp. 1486–1487.
Sadasivan, S., Khushalani, D., and Mann, S., Synthesis of calcium phosphate nanofilaments in reverse micelles, Chem. Mater., 2005, vol. 17, pp. 2765–2770.
Prelot, B., and Zemb, T., Calcium phosphate precipitation in catanionic templates, Mater. Sci. Eng. C, 2005, vol. 25, pp. 553–559.
Wei, K., Wang, Y., Lai, C., Ning, C., Wu, D., Wu, G., Zhao, N., Chen, X., and Ye, J., Synthesis and characterization of hydroxyapatite nanobelts and nanoparticles, Mater. Lett., 2005, vol. 59, pp. 220– 225.
Zhang, Y., Zhou, L., Li, D., Xue, N., Xu, X., and Li, J., Oriented nano-structured hydroxyapatite from the template, Chem. Phys. Lett., 2003, vol. 376, pp. 493–497.
Zhang, F., Zhou, Z., Yang, S., Mao, L., Chen, H., and Yu, X., Hydrothermal synthesis of hydroxyapatite nanorods in the presence of anionic starburst dendrimer, Mater. Lett., 2005, vol. 59, pp. 1422–1425.
Lin, K., Chang, J., Cheng, R., and Ruan, M., Hydrothermal microemulsion synthesis of stoichiometric single crystal hydroxyapatite nanorods with mono-dispersion and narrow-size distribution, Mater. Lett., 2007, vol. 61, pp. 1683–1687.
Liu, Y., Hou, D., and Wang, G., A simple wet chemical synthesis and characterization of hydroxyapatite nanorods, Mater. Chem. Phys., 2004, vol. 86, pp. 69–73.
Cao, M., Wang, Y., Guo, C., Qi, Y., and Hu, C., Preparation of ultrahigh-aspect-ratio hydroxyapatite nanofibers in reverse micelles under hydrothermal conditions, Langmuir, 2004, vol. 20, pp. 4784–4786.
Pang, Y.X. and Bao, X., Influence of temperature, ripening time and calcination on the morphology and crystallinity of hydroxyapatite nanoparticles, J. Eur. Ceram. Soc., 2003, vol. 23, pp.1697–1704.
Ye, W. and Wang, X.X., Ribbon-like and rod-like hydroxyapatite crystals deposited on titanium surface with electrochemical method, Mater. Lett., 2007, vol. 17, pp. 405–409.
Nilesh, R., Rutika, B., Sudhindra, B., and Nilima, T., Comparative evaluation of nano-hydroxyapatite preparation and calcium sucrose phosphate on microhardness of deciduous teeth after iron drop exposure—an in-vitro study, J. Clin. Exp. Dent., 2017, vol. 9, no. 4, pp. 579–583.
Suat, O. and Mehmet, Y., Studies on characterization of bovine hydroxyapatite/CaTiO3 Biocomposites, Adv. Mater. Sci. Eng., 2016, vol. 2016, pp. 1–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Fatemeh Mirjalili, Navabazam, A. & Samanizadeh, N. Preparation of Hydroxyapatite Nanoparticles from Natural Teeth. Russ J Nondestruct Test 57, 152–162 (2021). https://doi.org/10.1134/S1061830921020091
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061830921020091