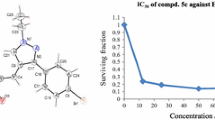
The cytotoxic activity of perfluoroalkyl imidazoindazoles and imidazobenzisoxazoles with respect to MCF-7 (human breast carcinoma) and HepG2 cell lines (human hepatocellular carcinoma) was studied. The imidazoindazoles exhibited significant cytotoxicity against MCF-7 and HepG2 cell lines. 2,6-bis(4-Fluorophenyl)-4,4-dimethyl-8-trifluoromethyl-3,4,5,6-tetrahydroimidazo[4,5-e]indazole displayed the most pronounced cytotoxic action. Regioisomeric imidazobenzisoxazoles inhibited extensively growth of HepG2 cells. The mechanism of cytotoxic action can be related to the induction of apoptosis as a result of disruption of the cell cycle, in particular, arrest of HepG2 cells in the G2 phase and MCF-7 cells in the G1 phase.



Similar content being viewed by others
References
M. Gaba and C. Mohan, Med. Chem. Res., 25(2), 173 – 210 (2016).
A. Kanwal, F. A. Saddique, S. Aslam, et al., Pharm. Chem. J., 51(12), 1068 – 1077 (2017).
I. Denya, S. F. Malan, and J. Joubert, Expert Opin. Ther. Pat., 28(6), 441 – 453 (2018).
D. D. Gaikwad, A. D. Chapolikar, C. G. Devkate, et al., Eur. J. Med. Chem., 90, 707 – 731 (2015).
J. Zhu, J. Mo, H-Z. Lin, et al., Bioorg. Med. Chem., 26(12), 3065 – 3075 (2018).
K. P. Rakesh, C. S. Shantharam, M. B. Sridhara, et al., Med. Chem. Commun., 8(11), 2023 – 2039 (2017).
Yu Zhou, J. Wang, Z. Gu, et al., Chem. Rev., 116(2), 422 – 518 (2016).
V. G. Isakova, T. S. Khlebnikova, and F. A. Lakhvich, Usp. Khim., 79(10), 929 – 960 (2010); Russ. Chem. Rev., 79(10), 849 – 879 (2010).
L. V. Politanskaya, G. A. Selivanova, E. V. Panteleeva, et al., Usp. Khim., 88(5), 425 – 569 (2019); Russ. Chem. Rev., 88(5), 425 – 569 (2019).
T. S. Khlebnikova, Yu. A. Piven’, V. G. Zinovich, et al., Khim.- farm. Zh., 54(7), 21 – 26 (2020); Pharm. Chem. J., 54(7), 700 – 706 (2020).
T. S. Khlebnikova, V. G. Isakova, A. V. Baranovskii, and F. A. Lakhvich, Zh. Obshch. Khim., 78(10), 1718 – 1727 (2008); Russ. J. Gen. Chem., 78(10), 1954 – 1963 (2008).
T. S. Khlebnikova, Yu. A. Piven’, A. V. Baranovskii, and F. A. Lakhvich, Zh. Org. Khim., 49(3), 433 – 439 (2013); Russ. J. Org. Chem., 49(3), 421 – 427 (2013).
T. S. Khlebnicova, Yu. A. Piven’, V. G. Isakova, et al., J. Heterocycl. Chem., 55(7), 1791 – 1797 (2018).
T. S. Khlebnikova, V. G. Isakova, Yu. A. Piven’, et al., Zh. Obshch. Khim., 90(4), 637 – 641 (2020); Russ. J. Gen. Chem., 90(4), 746 – 749 (2020).
J. van Meerloo, G. J. Kaspers, and J. Cloos, Methods Mol. Biol., 731, 237 – 245 (2011).
O. V. Panibrat, V. N. Zhabinskii, and V. A. Khripach, Dokl. Nats. Akad. Nauk Belarusi, 63(4), 437 – 444 (2019).
S. A. Mirmalek, M. A. Azizi, E. Jangholi, et al., Cancer Cell Int., 16, Art. No. 3, 1 – 9 (2016); doi: 10.1186/ s12935-016- 0279-4.
R. U. Khabriev (ed.), Handbook for Experimental (Preclinical) Studies of New Drugs [in Russian], Meditsina, Moscow (2005).
G. H. Williams and K. Stoeber, J. Pathol., 226(6), 352 – 364 (2012).
A. Lombardi Borgia, M. Ciomei, et al., US Pat. 9,597,317 B2, Mar. 21, 2017.
GOST 12.1.007–76, Noxious substances. Classification and general safety requirements.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 55, No. 3, pp. 10 – 14, March, 2021.
Rights and permissions
About this article
Cite this article
Khlebnikova, T.S., Panibrat, O.V., Piven, Y.A. et al. Cytotoxic Activity of Perfluoroalkyl-Substituted Imidazoindazoles and Imidazobenzisoxazoles. Pharm Chem J 55, 219–223 (2021). https://doi.org/10.1007/s11094-021-02401-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-021-02401-4