Abstract
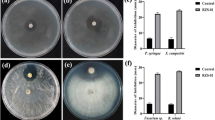
The inhibition effects of ten eugenol derivatives against Fusarium graminearum Q1 strain were determined using the mycelia growth rate process. Our results revealed that the toxicities of various derivatives differed substantially as compared to that of eugenol. Isoeugenol had the highest toxicity, about 4.2 times that of eugenol, with the EC50 value of 279.7 µM. Based on the toxicity ratio (TR) and synergistic coefficient (SR), the optimum mixing mass ratios of isoeugenol to phenamacril and isoeugenol to tebuconazole against F. graminearum were 7:1 and 6:1, respectively. Based on their additive effects, these mixtures could be used as potential alternatives to control F. graminearum resistance against phenamacril and tebuconazole due to different mechanism of action.
Similar content being viewed by others
References
Bai GH, Shaner G (2004) Management and resistance in wheat and barley to Fusarium head blight. Annu Rev Phytopathol 42:135–161
Beyer M, Klix MB, Klink H, Verreet A (2006) Quantifying the effects of previous crop, tillage, cultivar and triazole fungicides on the deoxynivalenol content of wheat grain-a review. J Plant Dis Prot 113(6):241–246
Blandino M, Haidukowski M, Pascale M, Plizzari L, Scudellari D, Reyneri A (2012) Integrated strategies for the control of Fusarium head blight and deoxynivalenol contamination in winter wheat. Field Crop Res 133:139–149
Bottalico A (1998) Fusarium diseases of cereals: species complex and related mycotoxin profiles, in Europe. J Plant Pathol 80:85–103
Brage PC, Sasso MD, Culici M, Alfieri M (2007) Eugenol and thymol, alone or in combination, induce morphological alterations in the envelope of Candida albicans. Fitoterapia 78:396–400
Campaniello D, Corbo MR, Sinigaglia M (2010) Antifungal activity of eugenol against Penicillium, Aspergillus, and Fusarium Species. J Food Prot 73:1124–1128
Carrasco H, Raimondi M, Svetaz L, Di Liberto M, Rodriguez MV, Espinoza L, Madrid A, Zacchino S (2012) Antifungal activity of eugenol analogues. Influence of different substituents and studies on mechanism of action. Molecules 17:1002–1024
Chen Y, Li HK, Chen CJ, Zhou MG (2008) Sensitivity of Fusarium graminearum to fungicide JS399-19: in vitro determination of baseline sensitivity and the risk of developing fungicide resistance. Phytoparasitica 36:326–337
Chen Y, Zhou MG (2009) Characterization of Fusarium graminearum isolates resistant to both carbendazim and a new fungicide JS399-19. Phytopathology 99:441–446
Dweba CC, Figlan S, Shimelis HA, Motaung TE, Sydenham S, Mwadzingeni L, Tsilo TJ (2017) Fusarium head blight of wheat: pathogenesis and control strategies. Crop Prot 91:114–122
FRAC (2020) FRAC Code List ©*2020: Fungal control agents sorted by cross resistance pattern and mode of action (including FRAC Code numbering) https://www.frac.info/docs/default-source/publications/frac-code-list/frac-code-list-2020-finalb16c2b2c512362eb9a1eff00004acf5d.pdf?sfvrsn=54f499a_2
Gill AO, Holley RA (2006) Inhibition of membrane bound ATPase of Escherichia coli and Listeria monocytogenes by plant oil aromatics. Int J Food Microbiol 111:170–174
Goswami RS, Kistler HC (2004) Heading for disaster: Fusarium graminearum on cereal crops. Mol Plant Pathol 5:515–525
Lei CY, Yin JL, Fang ZW, Wang SP, Zhang X, Ma DF (2018) Ihhibitory effects of phenamacril, tebuconazole and their mixtures on Fusarium graminearum in wheat. Plant Diseases and Pests 9(3–4):14–17
Li HK, Diao YM, Wang JX, Chen CJ, Ni JP, Zhou MG (2008) JS399-19, a new fungicide against wheat scab. Crop Prot 27(1):90–95
Liu Y, Chen X, Jiang J, Hamada MS, Yin Y, Ma Z (2014) Detection and dynamics of different carbendazim resistance conferring β-tubulin variants of Gibberella zeae collected from infected wheat heads and rice stubble in China. Pest Manag Sci 70(8):1228–1236
Marchese A, Barbieri R, Coppo E, Orhan IE, Daglia M, Nabavi SF, Izadi M, Abdollahi M, Nabavi SM, Ajami M (2017) Antimicrobial activity of eugenol and essential oils containing eugenol: A mechanistic viewpoint. Crit Rev Microbiol 43(6):668–689
Maurizio D., Richard L., Marisabel M., Rocco R., Vito Antonio R., Licia V., 2020. Fragrance components of gymnadenia conopsea and gymnadenia odoratissima collected at several sites in italy and germany. Natural Product Research, 1-5. http://dx.doi.org/10.1080/14786419.2020.1851227
Mesterhazy A, Bartok T, Lamper C (2003) Influence of wheat cultivar, species of Fusarium, and isolate aggressiveness on the efficiency of fungicides for control of Fusarium head blight. Plant Dis 87:1107–1115
Muellenborn C, Steiner U, Ludwig Mand Oerke E (2008) Effect of fungicides on the complex of Fusarium species and saprophytic fungi colonizing wheat kernels. Plant Pathol 120:157–166
Olea AF, Bravo A, Martínez R, Thomas M, Sedan C, Espinoza L, Carrasco H (2019) Antifungal activity of eugenol derivatives against Botrytis cinerea. Molecules 24(7):1239–1252
Pal C, Bandyopadhyay U (2012) Redox-active antiparasitic drugs. Antioxid Redox Signal 17:555–582
Qian HW, Du J, Chi MY, Sun XM, Liang WX, Huang JG, Li BD (2018) The Y137H mutation in the cytochrome P450 FgCYP51B protein confers reduced sensitivity to tebuconazole in Fusarium graminearum. Pest Manag Sci 74:1472–1477
Qu TL, Meng ZL, Li JQ (2008) Determination of degradation dynamics of fungicide 2-allylphenol in Rhizoctonia cerealis by high performance liquid Chromatography. Chin J Anal Chem 5:637–641 (in Chinese)
Romanazzi G, Lichter A, Gabler FM, Smilanick JL (2012) Recent advances on the use of natural and safe alternatives to conventional methods to control postharvest gray mold of table grapes. Postharvest Biol Technol 63(1):141–147
Sutton JC (1982) Epidemiology of wheat head blight and maize ear rot caused by Fusarium graminearum. Can J Plant Pathol 4:195–209
Varga E, Wiesenberger G, Hametner C, Ward TJ, Dong Y, Schöfbeck D, Krska R (2015) New tricks of an old enemy: isolates of Fusarium graminearum produce a type A trichothecene mycotoxin. Environ Microbiol 17(8):2588–2600
Wang CM, Zhang J, Chen H, Fan YJ, Shi ZQ (2010) Antifungal activity of eugenol against Botrytis cinerea. Tropical Plant Pathology 35(3):137–143
Wu RF, Si SY (2006) The screening of optimum ratio of abamectin and alphacypermethrin against Liriomyza sativae Blanchard. Modern Agrochemicals 5(3):49–51
Yang CJ, Gao Y, Du KY, Luo XY (2020) Screening of 17 Chinese medicine plants against phytopathogenic fungi and active component in Syzygium aromaticum. J Plant Dis Prot 127:237–244
Zaker M (2016) Natural plant products as eco-friendly fungicides for plant diseases control—a review. Agriculturists 14(1):134–141
Zhang YJ, Zhang X, Chen CJ, Zhou MG, Wang HC (2010) Effects of fungicides JS399-19, azoxystrobin, tebuconazloe, and carbendazim on the physiological and biochemical indices and grain yield of winter wheat. Pestic Biochem Physiol 98:151–157
Zheng ZT, Hou YP, Cai YQ, Zhang Y, Li YJ, Zhou MG (2015) Whole-genome sequencing reveals that mutations in myosin-5 confer resistance to the fungicide phenamacril in Fusarium graminearum. Sci Rep 5:8248–8256
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cui, W., Du, KY., Ling, YX. et al. Activity of eugenol derivatives against Fusarium graminearum Q1 strain and screening of isoeugenol mixtures. J Plant Pathol 103, 915–921 (2021). https://doi.org/10.1007/s42161-021-00875-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-021-00875-5