Abstract
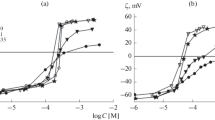
Theoretical analysis of the effect of the ionic strength of a solution on the surface (zeta) potential of liposomes formed by an anionic phospholipid (cardiolipin) with adsorbed polycations has been carried out. The experimental data were previously measured by the electrokinetic method in the presence of polylysine molecules of different molecular weights and a supporting electrolyte, KCl, at concentrations of 10, 40, and 100 mM. To approximate the experimental dependences of the potential on the amount of polylysine in the suspension, we used a theoretical model with parameters, among which the most physically significant are the thickness of the polymer layer, the adsorption constant, and the fraction of the surface of lipid membranes occupied by the polypeptide at the saturation. The found values of the model parameters demonstrate the effect of the length of the polypeptide molecules on the structure of the polymer layer varying from homogeneous to clustered distribution over the surface. A noticeable decrease in the efficiency of adsorption with an increase in the ionic strength of the solution is explained by the conformational rearrangements of the macromolecules on the surface and a decrease in the area of the surface available for their adsorption upon the saturation.




Similar content being viewed by others
REFERENCES
Meier W., 2000. Polymer nanocapsules. Chem. Soc. Rev. 25, 295–303. https://pubs.rsc.org/en/content/articlehtml/2000/cs/a809106d
Xu T., Zhang N., Nichols H.L., Shi D., Wen X. 2007. Modification of nanostructured materials for biomedical applications. Mater. Sci. Eng. C. 27, 579–594. https://doi.org/10.1016/j.msec.2006.05.029
Cho Y.W., Kim J.-D., Park K. 2003. Polycation gene delivery systems: Escape from endosomes to cytosol. J. Pharm. Pharmacol. 55, 721–734. https://doi.org/10.1211/002235703765951311
Hong S., Leroueil P.R., Janus E.K., Peters J.L., Kober M.M., Islam M.T., Orr B.G. 2006. Baker J.R., Banaszak Holl M.M., Interaction of polycationic polymers with supported lipid bilayers and cells: Nanoscale hole formation and enhanced membrane permeability. Bioconjug. Chem. 17, 728–734. https://doi.org/10.1021/bc060077y
Illergård J., Römling U., Wågberg L., Ek M. 2012. Biointeractive antibacterial fibres using polyelectrolyte multilayer modification. Cellulose. 19, 1731–1741. https://doi.org/10.1007/s10570-012-9742-0
McGeachy A.C., Dalchand N., Caudill E.R., Li T., Doǧangün M., Olenick L.L., Chang H., Pedersen J.A., Geiger F.M. 2018. Interfacial electrostatics of poly(vinylamine hydrochloride), poly(diallyldimethylammonium chloride), poly-L-lysine, and poly-L-arginine interacting with lipid bilayers. Phys. Chem. Chem. Phys. 20, 10846–10856. https://doi.org/10.1039/c7cp07353d
May S., Harries D., Ben-Shaul A. 2000. Lipid demixing and protein–protein interactions in the adsorption of charged proteins on mixed membranes. Biophys. J. 79, 1747–1760. https://doi.org/10.1016/S0006-3495(00)76427-7
Schwieger C., Blume A. 2007. Interaction of poly(L-lysines) with negatively charged membranes: An FT-IR and DSC study. Eur. Biophys. J. 36, 437–450. https://doi.org/10.1007/s00249-006-0080-8
Mbamala E.C., Ben-Shaul A., May S. 2005. Domain formation Induced by the adsorption of charged proteins on mixed lipid membranes. Biophys. J. 88, 1702–1714. https://doi.org/10.1529/biophysj.104.048132
Hurwitz G., Guillen G.R., Hoek E.M.V. 2010. Probing polyamide membrane surface charge, zeta potential, wettability, and hydrophilicity with contact angle measurements. J. Memb. Sci. 349, 349–357. https://doi.org/10.1016/j.memsci.2009.11.063
Freire J.M., Domingues M.M., Matos J., Melo M.N., Veiga A.S., Santos N.C., Castanho M.A.R.B. 2011. Using zeta-potential measurements to quantify peptide partition to lipid membranes. Eur. Biophys. J. 40, 481–487. https://doi.org/10.1007/s00249-010-0661-4
Alvares D.S., dos Santos Cabrera M.P., Ruggiero Neto J. 2016. Strategies for exploring electrostatic and nonelectrostatic contributions to the interaction of helical antimicrobial peptides with model membranes. Adv. Biomembr. Lipid Self-Assembly. 24, 43–73. https://doi.org/10.1016/bs.abl.2016.05.001
Dobrynin A.V., Rubinstein M. 2005. Theory of polyelectrolytes in solutions and at surfaces. Prog. Polym. Sci. 30, 1049–1118. https://doi.org/10.1016/j.progpolymsci.2005.07.006
Ohshima H. 1995. Electrophoresis of soft particles. Adv. Colloid Interface Sci. 62, 189–235. https://doi.org/10.1016/0001-8686(95)00279-Y
Finogenova O.A., Filinsky D. V., Ermakov Yu.A. 2008. Electrostatic effects upon adsorption and desorption of polylysines on the surface of lipid membranes of different composition. Biochem. (Moscow) Suppl. Ser. A Membr. Cell Biol. 2, 181–188. https://doi.org/10.1134/s1990747808020128
Marukovich N., McMurray M., Finogenova O., Nesterenko A., Batishchev O., Ermakov Yu. 2013. Interaction of polylysines with the surface of lipid membranes. Adv. Planar Lipid. 17, 139–166. https://doi.org/10.1016/b978-0-12-411516-3.00006-1
Yaroslavov A.A., Efimova A.A., Lobyshev V.I., Ermakov Y.A., Kabanov V.A. 1997. Reversibility of structural rearrangements in lipid membranes induced by adsorption-desorption of a polycation. Membr. Cell. Biol. 10, 683–688. https://europepmc.org/abstract/med/9231366
Heimburg T., Angerstein B., Marsh D. 1999. Binding of peripheral proteins to mixed lipid membranes: Effect of lipid demixing upon binding. Biophys. J. 76, 2575–2586. https://doi.org/10.1016/S0006-3495(99)77410-2
Marukovich N.I., Nesterenko A.M., Ermakov Y.A. 2015. Structural factors of lysine and polylysine interaction with lipid membranes. Biochem. (Moscow) Suppl. Ser. A Membr. Cell Biol. 9, 40–47. https://doi.org/10.1134/S1990747814060038
Kostritskii A.Y., Kondinskaia D.A., Nesterenko A.M., Gurtovenko A.A. 2016. Adsorption of synthetic cationic polymers on model phospholipid membranes: Insight from atomic-scale molecular dynamics simulations. Langmuir. 32, 10402–10414. https://doi.org/10.1021/acs.langmuir.6b02593
Khomich D.A., Nesterenko A.M., Kostritskii A.Y., Kondinskaia D.A., Ermakov Y.A., Gurtovenko A.A. 2017. Independent adsorption of monovalent cations and cationic polymers at PE/PG lipid membranes. J. Phys. Conf. Ser. 794, 012010. https://doi.org/10.1088/1742-6596/794/1/012010
Mcgeachy A.C., Caudill E.R., Liang D., Cui Q., Pedersen J.A., Geiger F.M. 2018. Counting charges on membrane-bound peptides. Chem. Sci. 9, 4285–4298. https://doi.org/10.1039/c8sc00804c
Duan X., Zhang R., Li Y., Shi T., An L., Huang Q. 2013. Monte Carlo study of polyelectrolyte adsorption on mixed lipid membrane. J. Phys. Chem. B. 117, 989–1002. https://doi.org/10.1021/jp310017j
Molotkovsky R.J., Galimzyanov T.R., Ermakov Yu.A. 2019. Polypeptides on the surface of lipid membranes. Theoretical analysis of electrokinetic data. Colloid J. 81, 125–135. https://doi.org/10.1134/S1061933X19020108
Ermakov Yu.A. 2000. Ion equilibrium near lipid membranes: Empirical analysis of the simplest model. Colloid J. 62, 389-400.
Ermakov Y. 1990. The determination of binding site density and assocition constants for monovalent cation adsorption onto liposomes made from mixtures of zwitterionic and charged lipids. Biochim. Biophys. Acta. Biomembr. 1023, 91–97.
Sokolov V.S., Batishchev O. V., Akimov S.A., Galimzyanov T.R., Konstantinova A.N., Malingriaux E., Gorbunova Y.G., Knyazev D.G., Pohl P. 2018. Residence time of singlet oxygen in membranes. Sci. Rep. 8, 1–11. https://doi.org/10.1038/s41598-018-31901-9
Finogenova O.A., Batischev O. V., Indenbom A.V., Zolotarevsky V.I., Ermakov Yu.A. 2009. Molecular distribution and charge of polylysine layers at the surface of lipid membranes and mica. Biochem. (Moscow) Suppl. Ser. A Membr. Cell Biol. 3, 496–503. https://doi.org/10.1134/S1990747809040187
Porcar I., Garcia R., Gómez C., Campos A., Abad C. 1997. Macromolecules in ordered media: 7. Influence of ionic strength and bilayer composition on the association of polyelectrolytes to mixed liposomes. Polymer (Guildf). 38, 5107–5113. https://doi.org/10.1016/S0032-3861(97)00061-X
Shin Y., Roberts J.E., Santore M.M. 2002. The relationship between polymer/substrate charge density and charge overcompensation by adsorbed polyelectrolyte layers. J. Colloid Interface Sci. 247, 220–230. https://doi.org/10.1006/jcis.2001.8100
Murray D., Arbuzova A., Hangyás-Mihályné G., Gambhir A., Ben-Talvvs N., Honig B., McLaughlin S. 1999. Electrostatic properties of membranes containing acidic lipids and adsorbed basic peptides: Theory and experiment. Biophys. J. 77, 3176–3188. https://doi.org/10.1016/S0006-3495(99)77148-1
Ivashkov O. V., Sybachin A. V., Efimova A.A., Pergushov D. V., Orlov V.N., Schmalz H., Yaroslavov A.A. 2015. The influence of the chain length of polycations on their complexation with anionic liposomes. ChemPhysChem. 16, 2849–2853. https://doi.org/10.1002/cphc.201500474
Ermakov Yu.A., Asadchikov V.E., Volkov Y.O., Nuzhdin A.D., Roshchin B.S., Honkimaki V., Tikhonov A.M. 2019. Electrostatic and structural effects at the adsorption of polylysine on the surface of the DMPS monolayer. JETP Lett. 109, 334–339. https://doi.org/10.1134/S0021364019050060
Gregory J., Barany S. 2011. Adsorption and flocculation by polymers and polymer mixtures. Adv. Coll. Int. Sci. 169, 1–12. https://doi.org/10.1016/j.cis.2011.06.004
Feng L., Stuart M. C., Adachi Y. 2015. Dynamics of polyelectrolyte adsorption and colloidal flocculation upon mixing studied using mono-dispersed polystyrene latex particles. Adv. Coll. Int. Sci. 226, 101–114. https://doi.org/10.1016/j.cis.2015.09.004
Yaroslavov A. A., Efimova A. A., Lobyshev V. I., Kabanov V. A. 2002. Reversibility of structural rearrangements in the negative vesicular membrane upon electrostatic adsorption/desorption of the polycation. Biochim. Biophys. Acta. Biomembr. 1560, 14–24. https://doi.org/10.1016/S0005-2736(01)00453-9
ACKNOWLEDGMENTS
This work was supported by the Ministry of Science and Higher Education of the Russian Federation (theme no. AAAA-A19-119010990119-9), as well as by the Russian Foundation for Basic Research (project no. 19-04-00242-a).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by R. Molotkovsky
Rights and permissions
About this article
Cite this article
Molotkovsky, R.J., Galimzyanov, T.R. & Ermakov, Y.A. Influence of Ionic Strength on Adsorption of Polypeptides on Lipid Membranes: Theoretical Analysis. Biochem. Moscow Suppl. Ser. A 15, 175–183 (2021). https://doi.org/10.1134/S1990747821030053
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990747821030053