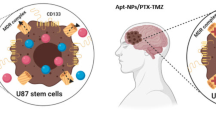
Abstract
Purpose
Glioblastoma (GBM) is a malignant brain tumor with a poor long-term prognosis due to recurrence from highly resistant GBM cancer stem cells (CSCs), for which the current standard of treatment with temozolomide (TMZ) alone will unlikely produce a viable cure. In addition, CSCs regenerate rapidly and overexpress methyl transferase which overrides the DNA-alkylating mechanism of TMZ, leading to resistance. The objective of this research was to apply the concepts of nanotechnology to develop a multi-drug therapy, TMZ and idasanutlin (RG7388, a potent mouse double minute 2 (MDM2) antagonist), loaded in functionalized nanoparticles (NPs) that target the GBM CSC subpopulation, reduce the cell viability and provide possibility of in vivo preclinical imaging.
Methods
Polymer-micellar NPs composed of poly(styrene-b-ethylene oxide) (PS-b-PEO) and poly(lactic-co-glycolic) acid (PLGA) were developed by a double emulsion technique loading TMZ and/or RG7388. The NPs were covalently bound to a 15-nucleotide base-pair CD133 aptamer to target the CD133 antigen expressed on the surfaces of GBM CSCs. For diagnostic functionality, the NPs were labelled with radiotracer Zirconium-89 (89Zr).
Results
NPs maintained size range less than 100 nm, a low negative charge and exhibited the ability to target and kill the CSC subpopulation when TMZ and RG7388 were used in combination. The targeting function of CD133 aptamer promoted killing in GBM CSCs providing impetus for further development of targeted nanosystems for localized therapy in future in vivo models.
Conclusions
This work has provided a potential clinical application for targeting GBM CSCs with simultaneous diagnostic imaging.







Similar content being viewed by others
Data Availability
N/A
Abbreviations
- ACN:
-
Acetonitrile
- BBB:
-
Blood brain barrier
- DCM:
-
Dichloromethane
- DDR:
-
DNA damage response
- DFOM:
-
Deferoxamine mesylate
- EMSA:
-
Electrophoretic mobility shift assay
- FAM:
-
Fluorescein amidite
- GBM:
-
Glioblastoma multiforme
- MDM2:
-
Mouse double minute 2
- NP:
-
Nanoparticles
- PLGA:
-
Poly(lactic-co-glycolic) acid
- PS-b-PEO:
-
Poly(styrene-b-ethylene oxide)
- TMZ:
-
Temozolomide
References
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–96.
Stupp R, Taillibert S, Kanner AA, Kesari S, Steinberg DM, Toms SA, et al. Maintenance therapy with tumor-treating fields plus Temozolomide vs Temozolomide alone for glioblastoma: a randomized clinical trial. Jama. 2015;314(23):2535–43.
Nizamutdinov D, Stock EM, Dandashi JA, Vasquez EA, Mao Y, Dayawansa S, et al. Prognostication of survival outcomes in patients diagnosed with glioblastoma. World Neurosurg. 2018;109:e67–74.
Fabian D, Guillermo Prieto Eibl MDP, Alnahhas I, Sebastian N, Giglio P, Puduvalli V, et al. Treatment of Glioblastoma (GBM) with the Addition of Tumor-Treating Fields (TTF): A Review. Cancers (Basel). 2019;11(2).
Bergmann N, Delbridge C, Gempt J, Feuchtinger A, Walch A, Schirmer L, et al. The Intratumoral heterogeneity reflects the Intertumoral subtypes of glioblastoma Multiforme: a regional immunohistochemistry analysis. Front Oncol. 2020;10:494.
Glaser T, Han I, Wu L, Zeng X. Targeted nanotechnology in glioblastoma Multiforme. Front Pharmacol. 2017;8:166.
Morokoff A, Ng W, Gogos A, Kaye AH. Molecular subtypes, stem cells and heterogeneity: implications for personalised therapy in glioma. J Clin Neurosci. 2015;22(8):1219–26.
Da Ros M, De Gregorio V, Iorio AL, Giunti L, Guidi M, de Martino M, et al. Glioblastoma Chemoresistance: The Double Play by Microenvironment and Blood-Brain Barrier. Int J Mol Sci. 2018;19(10).
Glumac PM, LeBeau AM. The role of CD133 in cancer: a concise review. Clin Transl Med. 2018;7(1):18.
Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, et al. Identification of human brain tumour initiating cells. Nature. 2004;432(7015):396–401.
Bauer M, Strom M, Hammond DS, Shigdar S. Anything You Can Do, I Can Do Better: Can Aptamers Replace Antibodies in Clinical Diagnostic Applications? Molecules. 2019;24(23).
Wang H, Cai S, Bailey BJ, Reza Saadatzadeh M, Ding J, Tonsing-Carter E, et al. Combination therapy in a xenograft model of glioblastoma: enhancement of the antitumor activity of temozolomide by an MDM2 antagonist. J Neurosurg. 2017;126(2):446–59.
Chu L, Wang A, Ni L, Yan X, Song Y, Zhao M, et al. Nose-to-brain delivery of temozolomide-loaded PLGA nanoparticles functionalized with anti-EPHA3 for glioblastoma targeting. Drug Delivery. 2018;25(1):1634–41.
Emamgholizadeh Minaei S, Khoei S, Khoee S, Karimi MR. Tri-block copolymer nanoparticles modified with folic acid for temozolomide delivery in glioblastoma. Int J Biochem Cell Biol. 2019;108:72–83.
Wang H, Cai S, Ernstberger A, Bailey BJ, Wang MZ, Cai W, et al. Temozolomide-mediated DNA methylation in human myeloid precursor cells: differential involvement of intrinsic and extrinsic apoptotic pathways. Clin Cancer Res. 2013;19(10):2699–709.
Thanasupawat T, Glogowska A, Burg M, Krcek J, Beiko J, Pitz M, et al. C1q/TNF-related peptide 8 (CTRP8) promotes temozolomide resistance in human glioblastoma. Mol Oncol. 2018;12(9):1464–79.
Ding Q, Zhang Z, Liu JJ, Jiang N, Zhang J, Ross TM, et al. Discovery of RG7388, a potent and selective p53-MDM2 inhibitor in clinical development. J Med Chem. 2013;56(14):5979–83.
Zhao Y, Aguilar A, Bernard D, Wang S. Small-molecule inhibitors of the MDM2-p53 protein-protein interaction (MDM2 inhibitors) in clinical trials for cancer treatment. J Med Chem. 2015;58(3):1038–52.
Zanjirband M, Edmondson RJ, Lunec J. Pre-clinical efficacy and synergistic potential of the MDM2-p53 antagonists, Nutlin-3 and RG7388, as single agents and in combined treatment with cisplatin in ovarian cancer. Oncotarget. 2016;7(26):40115–34.
Chen L, Rousseau RF, Middleton SA, Nichols GL, Newell DR, Lunec J, et al. Pre-clinical evaluation of the MDM2-p53 antagonist RG7388 alone and in combination with chemotherapy in neuroblastoma. Oncotarget. 2015;6(12):10207–21.
Mohanraj VJ, Chen Y. Nanoparticles - a review. Trop J Pharm Res. 2007;5(1):561–73.
Chen F, Goel S, Valdovinos HF, Luo H, Hernandez R, Barnhart TE, et al. In vivo integrity and biological fate of chelator-free Zirconium-89-labeled mesoporous silica nanoparticles. ACS Nano. 2015;9(8):7950–9.
Pellosi DS, Paula LB, de Melo MT, Tedesco AC. Targeted and synergic glioblastoma treatment: multifunctional nanoparticles delivering Verteporfin as adjuvant therapy for Temozolomide chemotherapy. Mol Pharm. 2019;16(3):1009–24.
Lee CY, Ooi IH. Preparation of Temozolomide-Loaded Nanoparticles for Glioblastoma Multiforme Targeting-Ideal Versus Reality. Pharmaceuticals (Basel). 2016;9(3).
Xu Y, Shen M, Li Y, Sun Y, Teng Y, Wang Y, et al. The synergic antitumor effects of paclitaxel and temozolomide co-loaded in mPEG-PLGA nanoparticles on glioblastoma cells. Oncotarget. 2016;7(15):20890–901.
Nabar GM, Mahajan KD, Calhoun MA, Duong AD, Souva MS, Xu J, et al. Micelle-templated, poly(lactic-co-glycolic acid) nanoparticles for hydrophobic drug delivery. Int J Nanomedicine. 2018;13:351–66.
Fang C, Wang K, Stephen ZR, Mu Q, Kievit FM, Chiu DT, et al. Temozolomide nanoparticles for targeted glioblastoma therapy. ACS Appl Mater Interfaces. 2015;7(12):6674–82.
McCall RL, Sirianni RW. PLGA nanoparticles formed by single- or double-emulsion with vitamin E-TPGS. J Vis Exp. 2013;82:51015.
Di Mauro PP, Cascante A, Brugada Vila P, Gomez-Vallejo V, Llop J, Borros S. Peptide-functionalized and high drug loaded novel nanoparticles as dual-targeting drug delivery system for modulated and controlled release of paclitaxel to brain glioma. Int J Pharm. 2018;553(1–2):169–85.
Portnow J, Badie B, Chen M, Liu A, Blanchard S, Synold TW. The neuropharmacokinetics of temozolomide in patients with resectable brain tumors: potential implications for the current approach to chemoradiation. Clinical cancer research : an official journal of the American Association for Cancer Research. 2009;15(22):7092–8.
Foroozandeh P, Aziz AA. Insight into cellular uptake and intracellular trafficking of nanoparticles. Nanoscale Res Lett. 2018;13(1):1–12.
Mosquera J, García I, Liz-Marzán LM. Cellular uptake of nanoparticles versus small molecules: a matter of size. Acc Chem Res. 2018;51(9):2305–13.
Danaei M, Dehghankhold M, Ataei S, Hasanzadeh Davarani F, Javanmard R, Dokhani A, et al. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics. 2018;10(2).
Veronesi MC, Alhamami M, Miedema SB, Yun Y, Ruiz-Cardozo M, Vannier MW. Imaging of intranasal drug delivery to the brain. Am J Nucl Med Mol Imaging. 2020;10(1):1–31.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors would like to acknowledge Dr. Daniel Minner of IUPUI Integrated Nanosystems Development Institute, Dr. Anne Shanahan of IUPUI Department of Chemistry & Chemical Biology, Ms. Caroline Miller of IU School of Medicine Electron Microscopy Core, and Ms. Barbara Bailey of the IU Simon Cancer Center In Vivo Therapeutics Core.
Funding
This publication was made possible by an award from the Indiana University School of Medicine (Indiana University School of Medicine Biomedical Research Grant). The content is solely the responsibility of the authors and does not necessarily represent the official views of the Indiana University School of Medicine.
Author information
Authors and Affiliations
Contributions
A part of this research constitute the MS thesis of SBS. The PI of the research project is MCV. All coauthors participated in intellectual contribution.
Corresponding author
Ethics declarations
None
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Smiley, S.B., Yun, Y., Ayyagari, P. et al. Development of CD133 Targeting Multi-Drug Polymer Micellar Nanoparticles for Glioblastoma - In Vitro Evaluation in Glioblastoma Stem Cells. Pharm Res 38, 1067–1079 (2021). https://doi.org/10.1007/s11095-021-03050-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-021-03050-8