Abstract
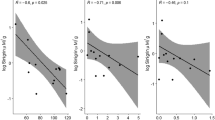
Past work shows a significant negative correlation between foliar oregonin concentration and western tent caterpillar (Malacosoma californicum Packard) feeding on red alder (Alnus rubra Bong.). Above an oregonin threshold of 20% leaf dry weight, little feeding by caterpillars is observed. Concentrations of defensive chemicals are influenced by plant genotype, environmental conditions, insect feeding, and the interactions of these factors. Our objective was to measure the effects of nitrogen (N) availability and wounding on foliar oregonin and condensed tannin concentrations in red alder genotypes. One-year-old seedlings from 100 half-sib red alder families were treated with two levels of ammonium nitrate (NH4NO3) for two growing seasons in a common garden. In the second year, leaves from 50 families from the fertilization experiment were used in a bioassay feeding experiment to determine the effects of N fertilization and genotype on WTC damage, and to identify a subset of 20 families with a range of damage to analyze for phytochemical composition. In separate experiments, wound-induction treatments were conducted outdoors and, in a greenhouse using the N treated trees in their third and fourth year, respectively. Foliar condensed tannin, oregonin and N concentrations were measured and ranked among the plant genotypes, and between the two N treatments and two wounding treatments. Results showed that oregonin and condensed tannin concentrations varied among the alder genotypes. Leaf N concentration was negatively correlated with concentration of oregonin. Neither of the measured phenolic compounds responded to wounding. The results suggest that red alder foliar oregonin and condensed tannin are likely constitutive defenses that are largely determined by genotype, and that the negative correlation of defense compounds with plant internal N status holds in this N-fixing tree.




Similar content being viewed by others
Data Availability
All data are available but not currently deposited in any public repository.
Code Availability
All codes used in this work are available for sharing.
References
Ayres MP (1993) Global change plant defense and herbivory. In: Kingsolver JG, Huey RB (eds) Kareiva PM. Biotic interactions and global change Sinauer Associated, Sunderland, pp 75–94
Ballhorn DJ, Elias JD, Balkan MA, Fordyce RF, Kennedy PG (2017) Colonization by nitrogen-fixing Frankia bacteria causes short-term increases in herbivore susceptibility in red alder (Alnus rubra) seedlings. Oecologia 184:497–506
Barbehenn R, Constabel CP (2011) Tannins in plant-herbivore interactions. Phytochemistry 72:1551–1565
Boateng K, Hawkins BJ, Constabel CP, Yanchuk A, Fellenberg C (2020) Red alder defense mechanisms against western tent caterpillar defoliation. Can J For Res. https://doi.org/10.1139/cjfr-2020-0343
Bruxelles GL, Roberts MR (2001) Signals regulating multiple responses to wounding and herbivores. Crit Rev Plant Sci 20(5):487–521
Bryant JP, Chapin FS III, Reichardt PB, Clausen TP (1987) Response of winter chemical defense in Alaska paper birch and green alder to manipulation of plant carbon/nutrient balance. Oecologia 72:510–514
Coley PD, Bryant JP, Chapin FS (1985) Resource availability and plant antiherbivore defense. Science 230:895–899
Constabel CP, Yip L, Patton JJ, Christopher ME (2000) Polyphenol oxidase from hybrid poplar. Cloning and expression in response to wounding and herbivory. Plant Physiol 124:285–295
Donaldson JR, Lindroth RL (2008) Effects of variable phytochemistry and budbreak phenology on defoliation of aspen during a forest tent caterpillar outbreak. Agric For Entomol 10:399–410
Fabisch T, Gershenzon J, Unsickler SB (2019) Specificity of herbivore defense responses in a woody plant, black poplar (Poplus nigra). J Chem Ecol 45:162–177
Gaulke LS, Henry CL, Brown SL (2006) Nitrogen fixation and growth response of Alnus rubra following fertilization with urea or biosolids. Sci Agric 63:361–369
Glynn C, Herms DA, Egawa M, Hansen R, Mattson WJ (2003) Effects of nutrient availability on biomass allocation as well as constitutive and rapid induced herbivore resistance in poplar. Oikos 101:385–397
González-Hernández MP, Starkey EE, Karchesy J (2000) Seasonal variation in concentration of fiber, crude protein, and phenolic compounds in leaves of red alder (Alnus rubra): Nutritional implication for cervids. J Chem Ecol 26:293–301
Gourlay G, Constabel CP (2019) Condensed tannins are inducible antioxidants and protect hybrid poplar against oxidative stress. Tree Physiol 39:345–355
Hawkins BJ, Jones MD, Kranabetter JM (2015) Ectomycorrhizae and the seedling nitrogen nutrition in forest restoration. New For 46:747–771
Harding SA, Jiang H, Jeong ML, Casado FL, Lin H-W, Tsai C-J (2005) Functional genomics analysis of foliar condensed tannin and phenolic glycoside regulation in natural cottonwood hybrids. Tree Physiol 25:1475–1486
Jackrel SL, Wootton JT (2015) Cascading effects of induced terrestrial plants defence on aquatic and terrestrial ecosystem function. Proc Royal Soc B 282:20142522
Koo C, Molina R, Miller S, Li C (1996) Effects of nitrogen and phosphorus fertilization on ectomycorrhiza development, N-fixation, and growth of red alder seedlings. J Korean For Soc 85:96–106
Koricheva J (2002) Meta-analysis of sources of variation in fitness costs of plant antiherbivore defenses. Ecology 83:176–190
Lavola A, Julkunen-Tiitto R (1994) The effect of elevated carbon dioxide and fertilization on primary and secondary metabolites in birch, Betula pendula (Roth). Oecogia 99:315–321
Lea CS, Bradbury SG, Constabel CP (2021) Anti-herbivore activity of oregonin, a diarylheptanoid found in leaves and bark of red alder (Alnus rubra). J Chem Ecol. https://doi.org/10.1007/s10886-021-01244-3
Lindroth RL, Bloomer MS (1991) Biochemical ecology of the forest tent caterpillar: responses to directory protein and phenolic glycosides. Oecologia. 86: 408-413
Lindroth RL, St. Clair SB (2013) Adaptations of quaking aspen (Populus tremuloides Michx.) for defense against herbivores. For Ecol Manage 299:14–21
Major IT, Constabel CP (2006) Molecular analysis of poplar defense against herbivory. Comparison of wound- and insect elicitor induced gene expression. New Phytol 172:617–635
Major IT, Constabel CP (2007) Shoot–root defense signaling and activation of root defense by leaf damage in poplar. Can J Bot 85:1171–1181
Mansfield JI, Curtis PS, Zak DR, Pregitzer KS (1999) Genotypic variation for condensed tannin production in trembling aspen (Populus tremuloides, salicaceae) under elevated CO2 and in high- and low-fertility soil. Am J Bot 86:1154–1159
Mellway RD, Tran LT, Prouse MB, Campbell MM, Constabel CP (2009) The wound-, pathogen-, and UV-B -responsive MYB134 gene encodes an R2R3 MYB transcription factor that regulates proanthocyanidin synthesis in poplar. Plant Physiol 150:924–941
Mutikainen P, Walls M, Ovaska J, Keina M, Julkunen-Tiitto R, Vapaavuori E (2000) Herbivore resistance in Betula pendula: Effect of fertilization, defoliation and plant genotype. Ecology 81:49–65
Mutikainen P, Walls M, Ovaska J, Keinänen M, Julkunen-Tiitto R, Vapaavuori E (2002) Costs of herbivore resistance in clonal saplings of Betula pendula. Oecologia 133:364–371
Myers JH, Williams KS (1987) Lack of short or long term inducible defenses in the red alder - western tent caterpillar system. Oikos 48:73–78
Oleksyn J, Karolewski P, Giertych MJ, Zytkowiak R, Reich PB, Tjoelker P (1998) Primary and secondary host plants differ in leaf-level photosynthetic response to herbivore: evidence from Alnus and Betula grazed by the alder beetle, Agelastica alni. New Phytol 140:239–249
Osier TL, Lindroth RL (2006) Genotype and environment determine allocation to and cost of resistance in quaking aspen. Oecologia 148:293–303
Porter LJ, Hrstich LN, Chan BG (1986) The conversion of procyanidins and prodelphinidins to cyanidin and delphidin. Phytochemistry 25:223–230
R Core Team (2017) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Ralph S, Oddy C, Cooper D, Yueh H, Jancsik S, Kolosova N, Bohlman J (2006) Genomics of hybrid poplar (Populus trichocarpa x deltoides) interacting with forest tent caterpillars (Malocosoma disstria): normalized and full-length cDNA libraries, expressed sequence tags, and a cDNA microassay for the study of insect-induced defences in poplar. Mol. Ecol 15:1275-1297
Rubert-Nason KF, Couture JJ, Major IT, Constabel CP, Lindroth RL (2015) Influence of genotype, environment, and gypsy moth herbivory on local and systemic chemical defense. J Chem Ecol 41(70):651–661
Sarfraz RM, Cory JS, Myer JH (2013) Life-history consequences and disease resistance of western tent caterpillars in response to localised, herbivore-induced changes in alder leaf quality. Ecol. Entomol. 38: 61-67
Sati SC, Sati N, Sati OP (2011) Bioactive constituents and medicinal importance of genus Alnus. Pharmacogn Rev 5:174–183
Shi Z (2007) Is trypsin inhibitor a plant defensive against the larvae of the forest tent caterpillar (Malocosoma disstria)? MSc. Thesis, Department of Biology, Concordia University. Montreal, Quebec, Canada
Tscharntke T, Thiessena S, Dolch R, Boland W (2001) Herbivory, induced resistance, and interplant signal transfer in Alnus glutinosa. Biochem Syst Ecol 29:1025–1047
Williams KS, Myers JH (1984) Previous herbivore attack on red alder may improve food quality of webworm larvae. Oecologia 63:166–170
Zhang LH, Ye GF, Lin YM, Zhan H, Zeng O (2009) Seasonal changes in tannin and nitrogen contents on Casuarina equisetifolia branchlets. J Zhejiang Univ Sci B 10:103–111
Acknowledgements
The authors gratefully acknowledge funding from the Natural Sciences and Engineering Research Council of Canada (NSERC) CREATE Program in Forests and Climate Change and from NSERC Discovery Grants to BJH and CPC. We thank the staff at the Cowichan Lake Research Station and the UVic Centre for Forest Biology for their technical assistance and support. In particular, thanks go to Samantha Robbins and Brad Binges for their help with lab analyses and greenhouse support. We also thank Steven Kiiskila and staff of the WFP Forestry Centre, Saanichton, for providing greenhouse and technical assistance. The invaluable comments from Dr. Cosmin Filipescu of the Pacific Forestry Centre are gratefully acknowledged.
Funding
This work was supported by NSERC Discovery Grants to BJH and CPC (Grant numbers NSERC DG RGPIN-2018-03763 and RGPIN-2020-06646).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Boateng, K., Hawkins, B.J., Yanchuk, A. et al. Factors Affecting Foliar Oregonin and Condensed Tannin in Red Alder (Alnus rubra Bong.): Phytochemicals Implicated In Defense Against Western Tent Caterpillar (Malacosoma californicum Packard). J Chem Ecol 47, 680–688 (2021). https://doi.org/10.1007/s10886-021-01283-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-021-01283-w