Abstract
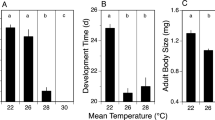
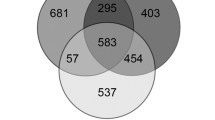
The seabird tick, Ixodes uriae, is a significant ectoparasite of penguins in Antarctica and of other seabirds, mainly in coastal, polar regions of the Northern and Southern hemispheres, but the tick’s distribution extends into more temperate regions as well. The expansive range of this tick suggests that it is exposed to a wide range of abiotic stresses, including dehydration, heat, and cold. To better understand how I. uriae responds to stress exposure, we examined cross-tolerance between dehydration and thermal stress based on survival analyses and used RNA-seq to monitor transcriptional responses to cold, heat, and dehydration. Slight dehydration improved cold, but not heat tolerance, whereas severe dehydration reduced subsequent thermal tolerance. Dehydration exposure prompted transcript-level shifts underlying protein metabolism, recovery from stress, and processes allowing subsequent rehydration by water vapor uptake. Both cold and heat stress yielded expression changes involved in cuticle modification. One gene increased in expression (enzyme P450) and one decreased (transcription factor Hairy) in response to all three stresses. This study provides the groundwork for assessing stress tolerance in this bipolar ectoparasite.




Similar content being viewed by others
Data availability
All datasets used for RNA-seq analyses are available under the NCBI Bioproject PRJNA659796.
References
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA (2000) Gene ontology: tool for the unification of biology. Nature Gen 25:25–29
Azad P, Zhou D, Russo E, Haddad GG (2009) Distinct mechanisms underlying tolerance to intermittent and constant hypoxia in Drosophila melanogaster. PLoS One 4:e5371
Baggerly KA, Deng L, Morris JS, Aldaz CM (2003) Differential expression in SAGE: accounting for normal between-library variation. Bioinformatics 19:1477–1483
Barbosa A, Benzal J, Vidal V et al (2011) Seabird ticks (Ixodes uriae) distribution along the Antarctic Peninsula. Pol Biol 34:1621–1624
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Royal Stat Soc: Series B 57:289–300
Benoit JB (2010) Water management by dormant insects: comparisons between dehydration resistance during summer aestivation and winter diapause. Prog Mol Subcell Biol 49:209–229
Benoit JB, Denlinger DL (2010) Meeting the challenges of on-host and off-host water balance in blood-feeding arthropods. J Insect Physiol 56:1366–1376
Benoit JB, Denlinger DL (2017) Bugs battle stress from hot blood. eLife 6:e33035
Benoit JB, Lazzari CR, Denlinger DL, Lahondère C (2019) Thermoprotective adaptations are critical for arthropods feeding on warm-blooded hosts. Curr Opin Insect Sci 34:7–11
Benoit JB, Lopez-Martinez G, Elnitsky MA et al (2009a) Dehydration-induced cross tolerance of Belgica antarctica larvae to cold and heat is facilitated by trehalose accumulation. Comp Biochem Physiol A 152:518–523
Benoit JB, Lopez-Martinez G, Teets NM, Phillips SA, Denlinger DL (2009b) Responses of the bed bug, Cimex lectularius, to temperature extremes and dehydration: levels of tolerance, rapid cold hardening and expression of heat shock proteins. Med Vet Entomology 23:418–425
Benoit JB, Yoder JA, Lopez-Martinez G et al (2007) Habitat requirements of the seabird tick, Ixodes uriae (Acari: Ixodidae), from the Antarctic Peninsula in relation to water balance characteristics of eggs, nonfed and engorged stages. J Comp Physiol B 177:205–215
Benoit JB, Oyen K, Finch G, Gantz JD, Wendeln K, Arya T, Lee RE Jr (2021) Cold hardening improves larval tick questing under low temperatures at the expense of longevity. Comp Biochem Physiol Part A: Mol Integ Physiol 257:110966
Conesa A, Götz S, García-Gómez JM et al (2005) Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21:3674–3676
Crooks E, Randolph SE (2006) Walking by Ixodes ricinus ticks: intrinsic and extrinsic factors determine the attraction of moisture or host odour. J Exp Biol 209:2138–2142
Dietrich M, Kempf BT, McCoy K (2014) Tracing the colonization and diversification of the worldwide seabird ectoparasite Ixodes uriae. Mol Ecol 23:3292–3305
Dunning LT, Dennis AB, Park D et al (2013) Identification of cold-responsive genes in a New Zealand alpine stick insect using RNA-Seq. Comp Biochem Physiol Part D 8:24–31
Dunning LT, Dennis AB, Sinclair BJ et al (2014) Divergent transcriptional responses to low temperature among populations of alpine and lowland species of New Zealand stick insects (Micrarchus). Mol Ecol 23:2712–2726
Eveleigh ES, Threlfall W (1975) The biology of Ixodes (Ceratixodes) uriae White, 1852 in Newfoundland. Acarologia 16:621–635
Feyereisen R (2012) Insect CYP genes and P450 enzymes. In: Gilbert LI (ed) Insect molecular biology and biochemistry. Academic Press, Cambridge
Feyereisen R (1999) Insect P450 enzymes. Ann Rev Entomol 44:507–533
Feyereisen R (2020) Origin and evolution of the CYP4G subfamily in insects, cytochrome P450 enzymes involved in cuticular hydrocarbon synthesis. Mol Phyl Evol 143:106695
Fieler AM, Rosendale AJ, Farrow DW, Dunlevy MD, Davies B, Oyen K, Xiao Y, Benoit JB (2021) Larval thermal characteristics of multiple ixodid ticks. Comp Biochem Physiol Part A: Mol Integ Physiol 257:110939
Findsen A, Andersen JL, Calderon S, Overgaard J (2013) Rapid cold hardening improves recovery of ion homeostasis and chill coma recovery time in the migratory locust, Locusta migratoria. J Exp Biol 216:1630–1637
Götz S, García-Gómez JM, Terol J et al (2008) High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res 36:3420–3435
Grabherr MG, Haas BJ, Yassour M et al (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652
Gulia-Nuss M, Nuss AB, Meyer JM et al (2016) Genomic insights into the Ixodes scapularis tick vector of Lyme disease. Nat Commun 7:10507
Hallas R, Schiffer M, Hoffmann AA (2002) Clinal variation in Drosophila serrata for stress resistance and body size. Genetics Res 79:141–148
Haas BJ, Papanicolaou A, Yassour M et al (2013) De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat Protoc 8:1494–1512
Hagan RW, Didion EM, Rosselot AE et al (2018) Dehydration prompts increased activity and blood feeding by mosquitoes. Sci Rep 8:6804
Hengherr S, Worland MR, Reuner A et al (2009) High-temperature tolerance in anhydrobiotic tardigrades is limited by glass transition. Physiol Biochem Zool 82:749–755
Herrmann C, Gern L (2013) Survival of Ixodes ricinus (Acari: Ixodidae) nymphs under cold conditions is negatively influenced by frequent temperature variations. Ticks Tick Borne Dis 4:445–451
Holmes CJ, Benoit JB (2019) Biological adaptations associated with dehydration in mosquitoes. Insects 10:375
Holmes CJ, Dobrotka CJ, Farrow DW et al (2018) Low and high thermal tolerance characteristics for unfed larvae of the winter tick Dermacentor albipictus (Acari: Ixodidae) with special reference to moose. Ticks Tick Borne Dis 9:25–30
Hoffmann AA, Anderson A, Hallas R (2002) Opposing clines for high and low temperature resistance in Drosophila melanogaster. Ecol Lett 5:614–618
Hoogstraal H, Eugene Mielcarek J (1954) Ixodes (Ceratixodes) uriae White, 1852, Parasitizing penguins and sea birds in the Falkland Islands (Ixodoidea, Ixodidae). J Parasit 40:232
Huang DW, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4:44–57
Huang Y, Niu B, Gao Y et al (2010) CD-HIT Suite: a web server for clustering and comparing biological sequences. Bioinformatics 26:680–682
Lee RE, Baust JG (1987) Cold-hardiness in the Antarctic tick, Ixodes uriae. Physiol Zool 60:499–506
Li A, Jiao X, Munier FL et al (2004) Bietti crystalline corneoretinal dystrophy is caused by mutations in the novel gene CYP4V2. Am J Hum Genet 74:817–826
Lindsay PJ, Kaufman WR (1986) Potentiation of salivary fluid secretion in ixodid ticks: a new receptor system for γ-aminobutyric acid. Can J Physiol Pharmacol 64:1119–1126
Liu G, Roy J, Johnson EA (2006) Identification and function of hypoxia-response genes in Drosophila melanogaster. Physiol Genomics 25:134–141
Lucien J, Reiffenstein R, Zbitnew G, Kaufman WR (1995) γ-aminobutyric acid (GABA) and other amino acids in tissues of the tick, Amblyomma hebraeum (Acari: Ixodidae) throughout the feeding and reproductive periods. Exp Appl Acarol 19:617–631
MacMillan HA, Knee JM, Dennis AB et al (2016) Cold acclimation wholly reorganizes the Drosophila melanogaster transcriptome and metabolome. Sci Rep 6:28999
MacMillan HA, Williams CM, Staples JF, Sinclair BJ (2012) Reestablishment of ion homeostasis during chill-coma recovery in the cricket Gryllus pennsylvanicus. Proc Natl Acad Sci U S A 109:20750–20755
Marteaux LED, Des Marteaux LE, McKinnon AH et al (2017) Effects of cold-acclimation on gene expression in Fall field cricket (Gryllus pennsylvanicus) ionoregulatory tissues. BMC Genomics 8:357
McCoy KD, Boulinier T, Tirard C, Michalakis Y (2001) Host specificity of a generalist parasite: genetic evidence of sympatric host races in the seabird tick Ixodes uriae. J Evol Biol 14:395–405
McCoy KD, Tirard C, Michalakis Y (2003) Spatial genetic structure of the ectoparasite Ixodes uriae within breeding cliffs of its colonial seabird host. Heredity 91:422–429
Mi H, Muruganujan A, Ebert D et al (2019) PANTHER version 14: more genomes, a new PANTHER GO-slim and improvements in enrichment analysis tools. Nucleic Acids Res 47:D419–D426
Moreira HNS, Barcelos RM, Vidigal PMP et al (2017) A deep insight into the whole transcriptome of midguts, ovaries and salivary glands of the Amblyomma sculptum tick. Parasitol Int 66:64–73
Muñoz-Leal S, González-Acuña D (2015) The tick Ixodes uriae (Acari: Ixodidae): Hosts, geographical distribution, and vector roles. Ticks Tick Borne Dis 6:843–868
Murray MD, Vestjens WJM (1967) Studies on the ectoparasites of seals and penguins. III. The distribution of the tick Ixodes uriae White and the flea Parapsyllus magellanicus heardi de Meillon on Macquarie Island. Aust J Zool 15:715
Needham GR, Teel PD (1991) Off-host physiological ecology of ixodid ticks. Annu Rev Entomol 36:659–681
Nguyen TTA, Michaud D, Cloutier C (2009) A proteomic analysis of the aphid Macrosiphum euphorbiae under heat and radiation stress. Insect Biochem Mol Biol 39:20–30
Ogden NH, Lindsay LR (2016) Effects of climate and climate change on vectors and vector-borne diseases: ticks are different. Trends Parasitol 32:646–656
Overgaard J, Malmendal A, Sørensen JG et al (2007) Metabolomic profiling of rapid cold hardening and cold shock in Drosophila melangaster. J Nsect Physiol 53:1218–1232
Overgaard J, Sørensen JG, Com E, Colinet H (2014) The rapid cold hardening response of Drosophila melanogaster: Complex regulation across different levels of biological organization. J Insect Physiol 62:46–53
Overgaard J, Sørensen JG, Petersen SO et al (2005) Changes in membrane lipid composition following rapid cold hardening in Drosophila melanogaster. J Insect Physiol 51:1173–1182
Parra G, Bradnam K, Korf I (2007) CEGMA: a pipeline to accurately annotate core genes in eukaryotic genomes. Bioinformatics 23:1061–1067
Quan P-Q, Li M-Z, Wang G-R et al (2020) Comparative transcriptome analysis of the rice leaf folder (Cnaphalocrocis medinalis) to heat acclimation. BMC Genomics 21:450
Randolph SE, Storey K (1999) Impact of microclimate on immature tick-rodent host interactions (Acari: Ixodidae): implications for parasite transmission. J Med Entomol 36:741–748
Reimand J, Arak T, Adler P et al (2016) g:Profiler—a web server for functional interpretation of gene lists (2016 update). Nucleic Acids Res 44:W83–W89
Michaud MR, Benoit JB, Lopez-Martinez G et al (2008) Metabolomics reveals unique and shared metabolic changes in response to heat shock, freezing and desiccation in the Antarctic midge, Belgica antarctica. J Insect Physiol 54:645–655
Rosendale AJ, Dunlevy ME, Fieler AM et al (2017) Dehydration and starvation yield energetic consequences that affect survival of the American dog tick. J Insect Physiol 101:39–46
Rosendale AJ, Dunlevy ME, McCue MD, Benoit JB (2019) Progressive behavioural, physiological and transcriptomic shifts over the course of prolonged starvation in ticks. Mol Ecol 28:49–65
Rosendale AJ, Farrow DW, Dunlevy ME et al (2016a) Cold hardiness and influences of hibernaculum conditions on overwintering survival of American dog tick larvae. Ticks Tick Borne Dis 7:1155–1161
Rosendale AJ, Leonard R, Patterson I, Benoit JB (2021) Dual metabolome and transcriptome profiling reveals betaine as a critical factor in tick rapid cold hardening. J Exp Biol Under revision
Rosendale AJ, Romick-Rosendale LE, Watanabe M et al (2016b) Mechanistic underpinnings of dehydration stress in the American dog tick revealed through RNA-Seq and metabolomics. J Exp Biol 219:1808–1819
Rudolph D, Knülle W (1974) Site and mechanism of water vapour uptake from the atmosphere in ixodid ticks. Nature 249:84–85
Schulz MH, Zerbino DR, Vingron M, Birney E (2012) Oases: robust de novo RNA-seq assembly across the dynamic range of expression levels. Bioinformatics 28:1086–1092
Short NJ, Floyd RB, Norval RA, Sutherst RW (1989) Development rates, fecundity and survival of developmental stages of the ticks Rhipicephalus appendiculatus, Boophilus decoloratus and B. microplus under field conditions in Zimbabwe. Exp Appl Acarol 6:123–141
Simão FA, Waterhouse RM, Ioannidis P et al (2015) BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31:3210–3212
Sinclair BJ, Ferguson LV, Salehipour-shirazi G, MacMillan HA (2013) Cross-tolerance and cross-talk in the cold: relating low temperatures to desiccation and immune stress in insects. Integr Comp Biol 53:545–556
Sogame Y, Kikawada T (2017) Current findings on the molecular mechanisms underlying anhydrobiosis in Polypedilum vanderplanki. Cur Opin Insect Sci 19:16–21
Sonenshine DE, Michael Roe R (2013) Biology of ticks. Oxford University Press
Teets NM, Peyton JT, Colinet H, Renault D, Kelley JL, Kawarasaki Y, Lee RE, Denlinger DL (2012) Gene expression changes governing extreme dehydration tolerance in an Antarctic insect. Proc Nat Acad Sci 109:20744–20749
Tian CB, Li YY, Huang J et al (2020) Comparative transcriptome and proteome analysis of heat acclimation in predatory mite Neoseiulus barkeri. Front Physiol 11:426
Vandyk JK, Bartholomew DM, Rowley WA, Platt KB (1996) Survival of Ixodes scapularis (Acari: Ixodidae) exposed to cold. J Med Entomol 33:6–10
Wang T, Yang X, Jia Q et al (2017) Cold tolerance and biochemical response of unfed Dermacentor silvarum ticks to low temperature. Ticks Tick Borne Dis 8:757–763
Wilson N (1970) New distributional records of ticks from Southeast Asia and the Pacific (Metastigmata : Argasidae, Ixodidae). Orient Insects 4:37–46
Yoder JA, Benoit JB, Denlinger DL, Rivers DB (2006) Stress-induced accumulation of glycerol in the flesh fly, Sarcophaga bullata: evidence indicating anti-desiccant and cryoprotectant functions of this polyol and a role for the brain in coordinating the response. J Insect Physiol 52:202–214
Yoder JA, Hedges BZ, Tank JL, Benoit JB (2009) Dermal gland secretion improves the heat tolerance of the brown dog tick, Rhipicephalus sanguineus, allowing for their prolonged exposure to host body temperature. J Therm Biol 34:256–265
Yu Z, Jia Q, Wang T et al (2017) Gene expression profiling of the unfed nymphal Dermacentor silvaum (Acari: Ixodidae) in response to low temperature. Syst Appl Acarol 22:2178
Yu Z-J, Lu Y-L, Yang X-L et al (2014) Cold hardiness and biochemical response to low temperature of the unfed bush tick Haemaphysalis longicornis (Acari: Ixodidae). Parasit Vect 7:346
Yu Z, Pei T, Zhang M et al (2020) Differential transcriptome profiling of the diapause and cold-responsive genes in unfed adult Dermacentor silvarum Olenev (Acari: Ixodidae). Syst Appl Acarol 25:193–213
Zhou D, Xue J, Lai JCK et al (2008) Mechanisms underlying hypoxia tolerance in Drosophila melanogaster: hairy as a metabolic switch. PLoS Genet 4:e1000221
Acknowledgements
This research was supported by the National Science Foundation grant DEB-1654417 (partially), United States Department of Agriculture 2018-67013-28495 (partially), and National Institutes of Health 1R01AI148551-01A1 (partially) to J.B.B. for computational support, National Science Foundation grant OPP-1341393 to D.L.D., and National Science Foundation grant OPP-1341385 to R.E.L. We thank the staff at Palmer Station, Antarctica for assistance in logistics and experiments. We appreciate Karen D. McCoy and Jingze Liu for serving as reviewers and providing helpful comments on the manuscript.
Author information
Authors and Affiliations
Contributions
This study was designed and conceived by J.B.B and A.J.R. The manuscript was written by B.D. and J.B.B. RNA-seq studies were conducted by A.J.R., B. D., and J.B.B. Physiological studies were conducted by J.D.G and J.B.B. Samples were collected by J.D.G., J.B.B., R.E.L., and D.L.D. Figures were prepared by B.D. and J.B.B. All authors were responsible for editing the manuscript and have approved publication.
Corresponding author
Ethics declarations
Conflict of interest
We do not declare any conflict of interests.
Informed consent
Not applicable to current study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
300_2021_2887_MOESM1_ESM.xlsx
Supplementary file1—Online Resource 1 - RNA-seq results for contigs with increased expression following dehydration exposure in Ixodes uriae. Expression values are in transcripts per million. RNA-seq datasets are available under the following NCBI Bioproject PRJNA659796. (XLSX 36 kb)
300_2021_2887_MOESM2_ESM.xlsx
Supplementary file2—Online Resource 2 - RNA-seq results for contigs with decreased expression following dehydration exposure in Ixodes uriae. Expression values are in transcripts per million. RNA-seq datasets are available under the following NCBI Bioproject PRJNA659796. (XLSX 47 kb)
300_2021_2887_MOESM3_ESM.xlsx
Supplementary file3—Online Resource 3 - RNA-seq results for contigs with increased expression following cold exposure in Ixodes uriae. Expression values are in transcripts per million. RNA-seq datasets are available under the following NCBI Bioproject PRJNA659796. (XLSX 15 kb)
300_2021_2887_MOESM4_ESM.xlsx
Supplementary file4—Online Resource 4 - RNA-seq results for contigs with decreased expression following cold exposure in Ixodes uriae. Expression values are in transcripts per million. RNA-seq datasets are available under the following NCBI Bioproject PRJNA659796. (XLSX 16 kb)
300_2021_2887_MOESM5_ESM.xlsx
Supplementary file5—Online Resource 5 - RNA-seq results for contigs with increased expression following heat exposure in Ixodes uriae. Expression values are in transcripts per million. RNA-seq datasets are available under the following NCBI Bioproject PRJNA659796. (XLSX 16 kb)
300_2021_2887_MOESM6_ESM.xlsx
Supplementary file6—Online Resource 6 - RNA-seq results for contigs with decreased expression following heat exposure in Ixodes uriae. Expression values are in transcripts per million. RNA-seq datasets are available under the following NCBI Bioproject PRJNA659796. (XLSX 31 kb)
300_2021_2887_MOESM7_ESM.xlsx
Supplementary file7—Online Resource 7 - Gene ontology categories enriched following cold, heat, and dehydration exposure in Ixodes uriae. (XLSX 16 kb)
Rights and permissions
About this article
Cite this article
Davies, B., Rosendale, A.J., Gantz, J.D. et al. Cross-tolerance and transcriptional shifts underlying abiotic stress in the seabird tick, Ixodes uriae. Polar Biol 44, 1379–1389 (2021). https://doi.org/10.1007/s00300-021-02887-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-021-02887-4