Abstract
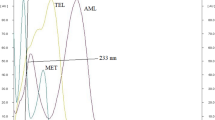
A precise, robust and accurate high-performance thin-layer chromatography (HPTLC) method has been developed for the simultaneous estimation of chlorthalidone and metoprolol succinate using analytical quality by design approach based on principles of quality risk management and design of experiments. The quality risk management was performed by the identification of probable method risk parameters and their assessment by allotting a risk priority number. Design of experiments was performed by Taguchi OA screening design and central composite response surface analysis using resolution between peaks as critical method performance attributes. A total of seven method risk parameters were screened for their main effect on the resolution between peaks using Design-Expert software (trial version). The main effect of three method parameters were found critical and needed to be optimised for method development. Hence, response surface analysis was done by linking the effects of critical method parameters, mobile phase composition, volume of modifier and chamber saturation time with the resolution between peaks by central composite design. Method operable design region was navigated for development of the method as per the analytical target profile. Chromatographic separation was performed using silica gel GF254 as the stationary phase and toluene‒methanol‒triethylamine (8:2:0.5, V/V) as the mobile phase in twin-trough chamber keeping saturation time of 15 min. The developed method was validated as per the International Council for Harmonisation (ICH) Q2 (R1) guideline. The developed method was applied for the assay of combined pharmaceutical dosage forms of chlorthalidone and metoprolol succinate and the results were found in good agreement with their labelled claim.









Similar content being viewed by others
References
Joel GH, Lee EL (2001) Goodman and Gilman’s, The Pharmacological basis of therapeutics 10th International Edn. McGraw Hill Publishers, Medical Publishing Division, New York, NY, pp 829, 859–60
Barar FSK (2004) Essentials of Pharmacotherapeutics. 5th edn. S. Chand & Company Ltd., New Delhi, pp 298–301, 239–49
Page C, Curtis M, Walker M, Hoffman B (2006) Integrated Pharmacology. 3rd edn. Mosby Elsevier; Maryland Heights, MO, pp 324–326
Tripathi KD (2003) Essential of Medical Pharmacology. 5th edn. Jaypee Brothers Medical Publishers, New Delhi, pp 48–52
Indian Pharmacopoeia, Vol. 1, 2 and 3. The Indian Pharmacopoeia Commission, Govt. of India, Ministry of Health and Family Welfare, Ghaziabad, 2010, pp 847–848, 1076–1078
British Pharmacopoeia, Vol. 2. The Department of Health, Medicines and Healthcare Products Regulatory Agency (MHRA), London, 2010, pp 185–186, 484–485
The United States Pharmacopeia-28. Asian edn. The USP Convention, Rockville, MD, 2005, pp 1739–1740, 4753
European Pharmacopoeia, Vol. 2. European Directorate for the Quality of Medicines & HealthCare, Strasbourg, 2011, pp 1424, 1671
British Pharmacopoeia, Vol. 2. The Department of Health, Medicines and Healthcare Products Regulatory Agency, The Stationery Office, London, 2010, pp 3933–3942
The United States Pharmacopeia-30. National Formulary-25. Asian edn. The USP Convention, Rockville, MD, 2007, pp 2647
International Council for Harmonization, guidelines Q2 (R1), Q8 (R2), Q9, Q10, Q14; ICH Harmonised Tripartite Guideline, 2009, Geneva
Niraimathi V, Suresh AJ, Kumar IS (2013) UV spectrophotometric methods for the estimation of chlorthalidone in bulk and oral dosage form. Indo Am J Pharm Res 3(9):7160–7167
Abdullah NS, Hassan MA, Hassan RO (2017) Spectrophotometric determination of chlorthalidone in pharmaceutical formulations using different order derivative methods. Arab J Chem 10(Suppl):S3426–S3433
Bhanushali VM, Hinge MA, Patel SN (2015) Development and validation of an UV spectrophotometric method for simultaneous determination of chlorthalidone and losartan potassium. J Pharm Res 9(1):54–59
Patel SN, Hinge MA, Bhanushali VM (2015) Development and validation of an UV spectrophotometric method for simultaneous determination of cilnidipine and chlorthalidone. J Pharm Res 9(1):41–45
Barot D, Pradhan PK, Patel G, Shah S, Parmar HP, Dey S, Upadhyay UM (2014) Simultaneous UV spectrophotometric estimation of olmesartan medoxomil and chlorthalidone in tablet dosage form. Pharma Innov 3(10):76–80
Singh B, Patel DK, Ghosh SK (2009) A reversed-phase high-performance liquid chromatographic method for determination of chlorthalidone in pharmaceutical formulation. Int J Pharm Pharma Sci 1(2):24–29
Sonawane S, Jadhav S, Rahade P, Chhajed S, Kshirsagar S (2016) Development and validation of stability-indicating method for estimation of chlorthalidone in bulk and tablets with the use of experimental design in forced degradation experiments. Scientifica 2(1):20–29
Quaglia MG, Farina AM, Fanali S (1988) Determination of chlorthalidone and its impurities in bulk and in dosage forms by high-performance thin-layer chromatographic densitometry. J Chromatogr A 456:435–439
Naazneen S, Sridevi A (2014) Stability-indicating RP-HPLC method for the simultaneous estimation of azilsartan medoxomil and chlorthalidone in solid dosage forms. Int J Pharm Pharma Sci 6(6):236–243
Parmar KE, Mehta RS, Patel ND, Parmar KE (2013) Development and validation of HPTLC method for simultaneous determination of telmisartan and chlorthalidone in bulk and pharmaceutical dosage form. Int J Pharm Pharma Sci 5(2):420–425
Kudumula N, Prasad YR (2014) Development and validation of RP-HPLC method for the simultaneous estimation of chlorthalidone and cilnidipine in bulk and combined tablet dosage form. Pharmacophore 5(4):442–450
Youssef RM, Maher HM, El-Kimary EI, Hassan EM, Barary MH (2013) Validated stability-indicating methods for the simultaneous determination of amiloride hydrochloride, atenolol, and chlorthalidone using HPTLC and HPLC with photodiode array detector. J AOAC Int 96(2):313–323
Pagar SA, Shinkar DM, Saudagar RB (2013) Development and validation of spectrophotometric method for determination of metoprolol succinate. Pharm Sci 3(4):224–228
Kulkarni MN, Kshirsagar RV, Sakarkar DM (2009) Development and validation of spectrophotometric method for determination of metoprolol succinate. Int J Chem Tech Res 1(4):1273–1277
Kadia TK, Shah DB, Dilip GM (2014) Development and validation of Q-absorbance ratio spectrophotometric method for simultaneous estimation of cilnidipine and metoprolol succinate in bulk and combined dosage form. Int J Pharm Pharma Sci 6(6):401–407
Jadhav SD, Kumbhar SM, Patel VD, Panchal RM, Bhatia MS (2013) Simultaneous estimation of metoprolol succinate and olmesartan medoxomil from capsule dosage form by first order derivative spectroscopic method. J Pharm Sci 40(1):1–8
Patel K, Patel A, Dave J, Patel C (2012) Absorbance correction method for estimation of telmisartan and metoprolol succinate in combined tablet dosage forms. Pharm Methods 3(2):106–111
Phale MD, Hamrapurkar PD (2009) A validated and simplified RP-HPLC of metoprolol succinate from bulk drugs. Asian J Res Chem 2(2):119–122
Shitole SV, Gurjar M, Shah M, Bal G, Pimple S, Patel R (2014) Stability indicating RP-HPLC method for quantitative estimation of S(‒)metoprolol succinate in tablet dosage form. Am J PharmTech Res 4(5):712–721
Hinge MA (2015) Development and validation of an RP-HPLC method for simultaneous determination of trimetazidine hydrochloride and metoprolol succinate. Int J Chem Pharm Anal 2(2):77–83
Patil VP (2012) Simultaneous HPTLC analysis of hydrochlorothiazide and metoprolol succinate in tablet and bulk dosage form. World Res J Org Chem 1(1):1–5
Bari SB, Patil MS (2013) Validated and stability indicating of RP-HPLC method for simultaneous estimation of S(‒)metoprolol succinate & clopidogrel bisulfate in bulk and tablet dosage form. Int J Innov Res Dev 2(7):484–489
Desai D, Vashi N, Dalvadi H, Desai S, Hinge M (2016) HPTLC method development and validation of cilnidipine and metoprolol succinate in combined dosage form. Pharm Methods 7(1):28–33
Thakker NM, Choudhari VP, Kuchekar BS, Panchal HB, Rakholiya DR, Murugan R (2012) Development and validation of a stability indicating RP-HPLC method for simultaneous estimation of olmesartan medoxomil and metoprolol succinate in pharmaceutical dosage form. Pharm Methods 3(2):84–89
Kakde R, Bawane N (2009) High-performance thin-layer chromatographic method for simultaneous analysis of metoprolol succinate and amlodipine besylate in pharmaceutical preparations. J Planar Chromatogr 22(2):115–119
Patel S, Patel D (2013) Simultaneous determination of metoprolol succinate and chlorthalidone by UV spectrophotometric method. Pharmagene 1(3):39–43
Kashyap R, Srinivasa U (2014) Dual wavelength & Q-absorption ratio spectrophotometry methods development and validation for simultaneous estimation of metoprolol succinate and chlorthalidone in bulk and dosage form. Int J Pharma Res Health Sci 2(6):425–431
Padmane SP, Jain ND, Ittadware AM, Walde SP (2014) A derivative UV–spectrophotometric method for the simultaneous determination of metoprolol succinate and chlorthalidone in combine dose tablet formulation. Int J Anal Bioanal Chem 4(1):33–41
Sheth A, Patel C, Ramlingam B, Shah N (2012) Simultaneous estimation of metoprolol succinate and chlorthalidone in pharmaceutical solid dosage form by using a developed and validated reverse phase high performance liquid chromatographic technique. Scholars Res J 2:17–21
Kashyap R, Srinivasa U (2013) Development and validation of HPLC method for the simultaneous estimation of chlorthalidone and metoprolol succinate in bulk and dosage form. Int J Pharm Drug Anal 1(2):1–14
Jyothi AN, Ali SS, Nalluri BN, Unnisa A (2014) Novel RP-HPLC–PDA method for the simultaneous estimation of metoprolol succinate and chlorthalidone in bulk and pharmaceutical dosage forms. Chem Sci Rev Lett 3(12):941–950
Acknowledgements
Authors would also like to thank the principal, Maliba Pharmacy College and provost, Uka Tarsadia University for providing all the required infrastructure and facilities needed to complete this research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Prajapati, P., Patel, M. & Shah, S. A robust high-performance thin-layer chromatography method for the simultaneous estimation of chlorthalidone and metoprolol succinate using quality risk assessment and design of experiments-based enhanced analytical quality by design approach. JPC-J Planar Chromat 34, 229–242 (2021). https://doi.org/10.1007/s00764-021-00107-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00764-021-00107-7