Abstract
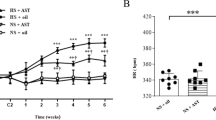
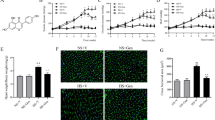
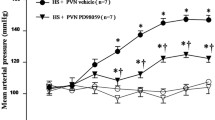
Apigenin, identified as 4′, 5, 7-trihydroxyflavone, is a natural flavonoid compound that has many interesting pharmacological activities and nutraceutical potential including anti-inflammatory and antioxidant functions. Chronic, low-grade inflammation and oxidative stress are involved in both the initiation and progression of hypertension and hypertension-induced cardiac hypertrophy. However, whether or not apigenin improves hypertension and cardiac hypertrophy through modulating NADPH oxidase-dependent reactive oxygen species (ROS) generation and inflammation in hypothalamic paraventricular nucleus (PVN) has not been reported. This study aimed to investigate the effects of apigenin on hypertension in spontaneously hypertensive rats (SHRs) and its possible central mechanism of action. SHRs and Wistar-Kyoto (WKY) rats were randomly assigned and treated with bilateral PVN infusion of apigenin or vehicle (artificial cerebrospinal fluid) via osmotic minipumps (20 μg/h) for 4 weeks. The results showed that after PVN infusion of apigenin, the mean arterial pressure (MAP), heart rate, plasma norepinephrine (NE), Beta 1 receptor in kidneys, level of phosphorylation of PKA in the ventricular tissue and cardiac hypertrophy, perivascular fibrosis, heart level of oxidative stress, PVN levels of oxidative stress, interleukin 1β (IL-1β), interleukin 6 (IL-6), iNOS, monocyte chemotactic protein 1 (MCP-1), tyrosine hydroxylase (TH), NOX2 and NOX4 were attenuated and PVN levels of interleukin 10 (IL-10), superoxide dismutase 1 (Cu/Zn-SOD) and the 67-kDa isoform of glutamate decarboxylase (GAD67) were increased. These results revealed that apigenin improves hypertension and cardiac hypertrophy in SHRs which are associated with the down-regulation of NADPH oxidase-dependent ROS generation and inflammation in the PVN.











Similar content being viewed by others
References
GBDRF Collaborators. (2018). Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet, 392, 1923–1994.
Li, Y., Ibrahim, R. M. S., Chi, H. L., Xiao, T., Xia, W. J., Li, H. B., & Kang, Y. M. (2021). Altered gut microbiota is involved in the anti-hypertensive effects of vitamin c in spontaneously hypertensive rat. Molecular Nutrition & Food Research. https://doi.org/10.1002/mnfr.202000885
Jiang, E., Chapp, A. D., Fan, Y. Y., Larson, R. A., Hahka, T., Huber, M. J., Yan, J. Q., Chen, Q. H., & Shan, Z. Y. (2018). Expression of proinflammatory cytokines is upregulated in the hypothalamic paraventricular nucleus of dahl salt-sensitive hypertensive rats. Frontiers in Physiology. https://doi.org/10.3389/fphys.2018.00104
Zheng, H., & Patel, K. P. (2017). Integration of renal sensory afferents at the level of the paraventricular nucleus dictating sympathetic outflow. Autonomic Neuroscience: Basic and Clinical, 204, 57–64.
Dampney, R. A., Michelini, L. C., Li, D. P., & Pan, H. L. (2018). Regulation of sympathetic vasomotor activity by the hypothalamic paraventricular nucleus in normotensive and hypertensive states. The American Journal of Physiology-Heart and Circulatory Physiology, 315, H1200–H1214.
Crowley, S. D. (2014). The cooperative roles of inflammation and oxidative stress in the pathogenesis of hypertension. Antioxidants & Redox Signaling, 20, 102–120.
Xu, M. L., Yu, X. J., Zhao, J. Q., Du, Y., Xia, W. J., Su, Q., Du, M. M., Yang, Q., Qi, J., Li, Y., Zhou, S. W., Zhu, G. Q., Li, H. B., & Kang, Y. M. (2020). Calcitriol ameliorated autonomic dysfunction and hypertension by down-regulating inflammation and oxidative stress in the paraventricular nucleus of SHR. Toxicology and Applied Pharmacology, 394, 114950.
Gao, H. L., Yu, X. J., Qi, J., Yi, Q. Y., Jing, W. H., Sun, W. Y., Cui, W., Mu, J. J., Yuan, Z. Y., Zhao, X. F., Liu, K. L., Zhu, G. Q., Shi, X. L., Liu, J. J., & Kang, Y. M. (2016). Oral CoQ10 attenuates high salt-induced hypertension by restoring neurotransmitters and cytokines in the hypothalamic paraventricular nucleus. Scientific Reports-UK. https://doi.org/10.1038/srep30301
Su, Q., Liu, J. J., Cui, W., Shi, X. L., Guo, J., Li, H. B., Huo, C. J., Miao, Y. W., Zhang, M., Yang, Q., & Kang, Y. M. (2016). Alpha lipoic acid supplementation attenuates reactive oxygen species in hypothalamic paraventricular nucleus and sympathoexcitation in high salt-induced hypertension. Toxicology Letters, 241, 152–158.
Kang, Y. M., Ma, Y., Zheng, J. P., Elks, C., Sriramula, S., Yang, Z. M., & Francis, J. (2009). Brain nuclear factor-kappa B activation contributes to neurohumoral excitation in angiotensin II-induced hypertension. Cardiovascular Research, 82, 503–512.
Shi, P., Diez-Freire, C., Jun, J. Y., Qi, Y. F., Katovich, M. J., Li, Q. H., Sriramula, S., Francis, J., Sumners, C., & Raizada, M. K. (2010). Brain microglial cytokines in neurogenic hypertension. Hypertension, 56, 297–303.
Qi, J., Yu, X. J., Fu, L. Y., Liu, K. L., Gao, T. T., Tu, J. W., Kang, K. B., Shi, X. L., Li, H. B., Li, Y., & Kang, Y. M. (2019). Exercise training attenuates hypertension through TLR4/MyD88/NF-kappa B signaling in the hypothalamic paraventricular nucleus. Frontiers in Neuroscience-Switzerland. https://doi.org/10.3389/fnins.2019.01138
Li, T. J., Chen, Y. L., Gua, C. J., & Wu, B. G. (2018). Elevated oxidative stress and inflammation in hypothalamic paraventricular nucleus are associated with sympathetic excitation and hypertension in rats exposed to chronic intermittent hypoxia. Frontiers in Physiology. https://doi.org/10.3389/fphys.2018.00840
Wang, M. L., Kang, Y. M., Li, X. G., Su, Q., Li, H. B., Liu, K. L., Fu, L. Y., Saahene, R. O., Li, Y., Tan, H., & Yu, X. J. (2018). Central blockade of NLRP3 reduces blood pressure via regulating inflammation microenvironment and neurohormonal excitation in salt-induced prehypertensive rats. Journal of Neuroinflammation. https://doi.org/10.1186/s12974-018-1131-7
Haspula, D., & Clark, M. A. (2018). Neuroinflammation and sympathetic overactivity: mechanisms and implications in hypertension. Autonomic Neuroscience, 210, 10–17.
Kang, Y., Ding, L., Dai, H. B., Wang, F. Z., Zhou, H., Gao, Q., Xiong, X. Q., Zhang, F., Song, T. R., Yuan, Y., Zhu, G. Q., & Zhou, Y. B. (2019). Intermedin in paraventricular nucleus attenuates Ang II-induced sympathoexcitation through the inhibition of NADPH oxidase-dependent ROS generation in obese rats with hypertension. International Journal of Molecular Sciences. https://doi.org/10.3390/ijms20174217
Santisteban, M. M., Qi, Y., Zubcevic, J., Kim, S., Yang, T., Shenoy, V., Cole-Jeffrey, C. T., Lobaton, G. O., Stewart, D. C., Rubiano, A., Simmons, C. S., Garcia-Pereira, F., Johnson, R. D., Pepine, C. J., & Raizada, M. K. (2017). Hypertension-linked pathophysiological alterations in the gut. Circulation Research, 120, 312–323.
Hostetler, G. L., Ralston, R. A., & Schwartz, S. J. (2017). Flavones: food sources, bioavailability, metabolism, and bioactivity. Advances in Nutrition, 8, 423–435.
Zhang, J. J., Liu, D. P., Huang, Y. T., Gao, Y., & Qian, S. A. (2012). Biopharmaceutics classification and intestinal absorption study of apigenin. International Journal of Pharmaceutics, 436, 311–317.
Venigalla, M., Sonego, S., Gyengesi, E., Sharman, M. J., & Munch, G. (2016). Novel promising therapeutics against chronic neuroinflammation and neurodegeneration in Alzheimer’s disease. Neurochemistry International, 95, 63–74.
Ren, B., Qin, W. W., Wu, F. H., Wang, S. S., Pan, C., Wang, L. Y., Zeng, B., Ma, S. P., & Liang, J. Y. (2016). Apigenin and naringenin regulate glucose and lipid metabolism, and ameliorate vascular dysfunction in type 2 diabetic rats. European Journal of Pharmacology, 773, 13–23.
Huang, C. S., Lii, C. K., Lin, A. H., Yeh, Y. W., Yao, H. T., Li, C. C., Wang, T. S., & Chen, H. W. (2013). Protection by chrysin, apigenin, and luteolin against oxidative stress is mediated by the Nrf2-dependent up-regulation of heme oxygenase 1 and glutamate cysteine ligase in rat primary hepatocytes. Archives of Toxicology, 87, 167–178.
Telange, D. R., Patil, A. T., Pethe, A. M., Fegade, H., Anand, S., & Dave, V. S. (2017). Formulation and characterization of an apigenin-phospholipid phytosome (APLC) for improved solubility, in vivo bioavailability, and antioxidant potential. European Journal of Pharmaceutical Sciences, 108, 36–49.
Li, K. P., He, Z. R., Wang, X. Q. Y., Pineda, M., Chen, R. B., Liu, H. Q., Ma, K. T., Shen, H. J., Wu, C. H., Huang, N. T., Pan, T. L., Liu, Y., & Guo, J. (2018). Apigenin c-glycosides of microcos paniculata protects lipopolysaccharide induced apoptosis and inflammation in acute lung injury through TLR4 signaling pathway. Free Radical Biology and Medicine, 124, 163–175.
Lee, J. H., Zhou, H. Y., Cho, S. Y., Kim, Y. S., Lee, Y. S., & Jeong, C. S. (2007). Anti-inflammatory mechanisms of apigenin: inhibition of cyclooxygenase-2 expression, adhesion of monocytes to human umbilical vein endothelial cells, and expression of cellular adhesion molecules. Archives of Pharmacal Research, 30, 1318–1327.
Lapchak, P. A., & Boitano, P. D. (2014). Effect of the pleiotropic drug CNB-001 on tissue plasminogen activator (tPA) protease activity in vitro: support for combination therapy to treat acute ischemic stroke. Journal of Neurology and Neurophysiology., 5(4), 214.
Huang, C. H., Kuo, P. L., Hsu, Y. L., Chang, T. T., Tseng, H. I., Chu, Y. T., Kuo, C. H., Chen, H. N., & Hung, C. H. (2010). The natural flavonoid apigenin suppresses Th1-and Th2-related chemokine production by human monocyte THP-1 cells through mitogen-activated protein kinase pathways. Journal of Medicinal Food, 13, 391–398.
Tian, H., Kang, Y. M., Gao, H. L., Shi, X. L., Fu, L. Y., Li, Y., Jia, X. Y., Liu, K. L., Qi, J., Li, H. O., Chen, Y. I., Chen, W. S., Cui, W., Zhu, G. Q., & Yu, X. J. (2019). Chronic infusion of berberine into the hypothalamic paraventricular nucleus attenuates hypertension and sympathoexcitation via the ROS/Erk1/2/iNOS pathway. Phytomedicine, 52, 216–224.
Su, Q., Qin, D. N., Wang, F. X., Ren, J., Li, H. B., Zhang, M., Yang, Q., Miao, Y. W., Yu, X. J., Qi, J., Zhu, Z. M., Zhu, G. Q., & Kang, Y. M. (2014). Inhibition of reactive oxygen species in hypothalamic paraventricular nucleus attenuates the renin-angiotensin system and proinflammatory cytokines in hypertension. Toxicology and Applied Pharmacology, 276, 115–120.
Lu, Q. B., Sun, J., Kang, Y., Sun, H. J., Wang, H. S., Wang, Y., Zhu, G. Q., & Zhou, Y. B. (2017). Superoxide anions and NO in the paraventricular nucleus modulate the cardiac sympathetic afferent reflex in obese rats. International Journal of Molecular Sciences. https://doi.org/10.3390/ijms19010059
Chen, Y. M., Yu, X. J., Liu, K. L., Gao, H. L., Li, Y., Sun, T. Z., Shi, X. L., Li, H. B., Zhu, G. Q., Qi, J., & Kang, Y. M. (2019). Inhibition of hypothalamic inhibitor kappa B kinase beta/nuclear transcription factor kappa B pathway attenuates metabolism and cardiac dysfunction in type 2 diabetic rats. Neuroendocrinology. https://doi.org/10.1016/j.molmet.2013.09.005
Li, H. B., Yang, T., Richards, E. M., Pepine, C. J., & Raizada, M. K. (2020). Maternal treatment with captopril persistently alters gut-brain communication and attenuates hypertension of male offspring. Hypertension, 75, 1315–1324.
Li, H. B., Qin, D. N., Cheng, K., Su, Q., Miao, Y. W., Guo, J., Zhang, M., Zhu, G. Q., & Kang, Y. M. (2015). Central blockade of salusin beta attenuates hypertension and hypothalamic inflammation in spontaneously hypertensive rats. Scientific Reports, 5, 11162.
Li, H. B., Li, X., Huo, C. J., Su, Q., Guo, J., Yuan, Z. Y., Zhu, G. Q., Shi, X. L., Liu, J. J., & Kang, Y. M. (2016). TLR4/MyD88/NF-kappa B signaling and PPAR-gamma within the paraventricular nucleus are involved in the effects of telmisartan in hypertension. Toxicology and Applied Pharmacology, 305, 93–102.
Yang, Q., Yu, X. J., Su, Q., Yi, Q. Y., Song, X. A., Shi, X. L., Li, H. B., Qi, J., Zhu, G. Q., & Kang, Y. M. (2020). Blockade of c-Src within the paraventricular nucleus attenuates inflammatory cytokines and oxidative stress in the mechanism of the TLR4 signal pathway in salt-induced hypertension. Neuroscience Bulletin, 36, 385–395.
Buwa, C. C., Mahajan, U. B., Patil, C. R., & Goyal, S. N. (2016). Apigenin attenuates beta-receptor-stimulated myocardial injury via safeguarding cardiac functions and escalation of antioxidant defence system. Cardiovascular Toxicology, 16, 286–297.
Hu, J., Li, Z., Xu, L. T., Sun, A. J., Fu, X. Y., Zhang, L., Jing, L. L., Lu, A. D., Dong, Y. F., & Jia, Z. P. (2015). Protective effect of apigenin on ischemia/reperfusion injury of the isolated rat heart. Cardiovascular Toxicology, 15, 241–249.
Ruoppolo, M., Orru, S., Talamo, F., Ljung, J., Pirneskoski, A., Kivirikko, K. I., Marino, G., & Koivunen, P. (2003). Mutations in domain a’ of protein disulfide isomerase affect the folding pathway of bovine pancreatic ribonuclease A. Protein Science, 12, 939–952.
Lee, Y. M., Lee, G., Oh, T. I., Kim, B. M., Shim, D. W., Lee, K. H., Kim, Y. J., Lim, B. O., & Lim, J. H. (2016). Inhibition of glutamine utilization sensitizes lung cancer cells to apigenin-induced apoptosis resulting from metabolic and oxidative stress. International Journal of Oncology, 48, 399–408.
Sabbatini, A. R., & Kararigas, G. (2020). Estrogen-related mechanisms in sex differences of hypertension and target organ damage. Biology of Sex Differences, 11, 31.
Hinojosa-Laborde, C., Craig, T., Zheng, W., Ji, H., Haywood, J. R., & Sandberg, K. (2004). Ovariectomy augments hypertension in aging female Dahl salt-sensitive rats. Hypertension, 44, 405–409.
Sampson, A. K., Hilliard, L. M., Moritz, K. M., Thomas, M. C., Tikellis, C., Widdop, R. E., & Denton, K. M. (2012). The arterial depressor response to chronic low-dose angiotensin II infusion in female rats is estrogen dependent. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 302, R159-165.
Moreira, J. D., Chaudhary, P., Frame, A. A., Puleo, F., Nist, K. M., Abkin, E. A., Moore, T. L., George, J. C., & Wainford, R. D. (2019). Inhibition of microglial activation in rats attenuates paraventricular nucleus inflammation in G alpha i(2) protein-dependent, salt-sensitive hypertension. Experimental Physiology, 104, 1892–1910.
Xu, M. L., Yu, X. J., Zhao, J. Q., Du, Y., Xia, W. J., Su, Q., Du, M. M., Yang, Q., Qi, J., Li, Y., Zhou, S. W., Zhu, G. Q., Li, H. B., & Kang, Y. M. (2020). Calcitriol ameliorated autonomic dysfunction and hypertension by down-regulating inflammation and oxidative stress in the paraventricular nucleus of SHR. Toxicology and Applied Pharmacology. https://doi.org/10.1016/j.taap.2020.114950
Song, X. A., Jia, L. L., Cui, W., Zhang, M., Chen, W., Yuan, Z. Y., Guo, J., Li, H. H., Zhu, G. Q., Liu, H., & Kang, Y. M. (2014). Inhibition of TNF-alpha in hypothalamic paraventricular nucleus attenuates hypertension and cardiac hypertrophy by inhibiting neurohormonal excitation in spontaneously hypertensive rats. Toxicology and Applied Pharmacology, 281, 101–108.
Papay, Z. E., Kosa, A., Boddi, B., Merchant, Z., Saleem, I. Y., Zariwala, M. G., Klebovich, I., Somavarapu, S., & Antal, I. (2017). Study on the pulmonary delivery system of apigenin-loaded albumin nanocarriers with antioxidant activity. Journal of Aerosol Medicine and Pulmonary Drug Delivery, 30, 274–288.
Wang, Y. C., & Huang, K. M. (2013). In vitro anti-inflammatory effect of apigenin in the Helicobacter pylori-infected gastric adenocarcinoma cells. Food and Chemical Toxicology, 53, 376–383.
Karamese, M., Erol, H. S., Albayrak, M., Findik Guvendi, G., Aydin, E., & Aksak Karamese, S. (2016). Anti-oxidant and anti-inflammatory effects of apigenin in a rat model of sepsis: an immunological, biochemical, and histopathological study. Immunopharmacology and Immunotoxicology, 38, 228–237.
Chen, P. F., Huo, X. H., Liu, W. Q., Li, K., Sun, Z. P., & Tian, J. H. (2020). Apigenin exhibits anti-inflammatory effects in LPS-stimulated BV2 microglia through activating GSK3 beta/Nrf2 signaling pathway. Immunopharmacology and Immunotoxicology, 42, 9–16.
Paredes-Gonzalez, X., Fuentes, F., Jeffery, S., Saw, C. L. L., Shu, L. M., Su, Z. Y., & Kong, A. N. T. (2015). Induction of NRF2-mediated gene expression by dietary phytochemical flavones apigenin and luteolin. Biopharmaceutics & Drug Disposition, 36, 440–451.
Darabi, P., Khazali, H., & Natanzi, M. M. (2020). Therapeutic potentials of the natural plant flavonoid apigenin in polycystic ovary syndrome in rat model: via modulation of pro-inflammatory cytokines and antioxidant activity. Gynecological Endocrinology, 36, 582–587.
Li, F., Lang, F. F., Zhang, H. L., Xu, L. D., Wang, Y. D., Zhai, C. X., & Hao, E. K. (2017). Apigenin alleviates endotoxin-induced myocardial toxicity by modulating inflammation, oxidative stress, and autophagy. Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2017/2302896
Zhu, Z. Y., Gao, T., Huang, Y., Xue, J., & Xie, M. L. (2016). Apigenin ameliorates hypertension-induced cardiac hypertrophy and down-regulates cardiac hypoxia inducible factor-lalpha in rats. Food and Function, 7, 1992–1998.
Acknowledgements
This work was funded by National Natural Science Foundation of China (Nos. 81770426, 82070439, 82070440), the Fundamental Research Funds for the Central Universities (Nos. PY3A044, xjh012019059, xzd012019035), China Postdoctoral Science Foundation (No. 2019M663750), and Natural Science Basic Research Program of Shaanxi (Nos. 2021JQ-068, 2020JM-079, 2019JQ-605).
Author information
Authors and Affiliations
Contributions
YM-K, J-Q and HL-G designed the study. HL-G, J-Q, HB-H, QW-Y, KL-L and YM-C performed all experiments. HL-G, Y–Z and DD-Z performed the data analysis and drafted the manuscript. YM-K, GQ-Z and H-T revised the manuscript. All authors reviewed the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Y. James Kang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hong-Li Gao and Xiao-Jing Yu contributed equally to this study.
Rights and permissions
About this article
Cite this article
Gao, HL., Yu, XJ., Hu, HB. et al. Apigenin Improves Hypertension and Cardiac Hypertrophy Through Modulating NADPH Oxidase-Dependent ROS Generation and Cytokines in Hypothalamic Paraventricular Nucleus. Cardiovasc Toxicol 21, 721–736 (2021). https://doi.org/10.1007/s12012-021-09662-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-021-09662-1