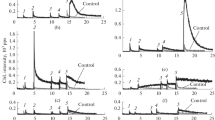
Dibasic acids (DBA) with benzene rings having substituents and connected by a sulfurcontaining bridge exhibited radioprotective effectiveness when administered in non-toxic concentrations to laboratory mice 1 h before γ-irradiation in a lethal dose of 8 Gy. The correlation of protonophore activity on bilayer lipid membranes with radioprotective (in mice) and uncoupling activity (in mitochondria) indicates a molecular protonophore uncoupling radioprotective mechanism of DBA action on mitochondria, which manifests in temporary partial inhibition of energy production. The effectiveness of DBA depends on the position and the degree of dissociation of OH-groups and increased in the presence of a sulfur-containing bridge and substituents in the series Br—>Cl—>NO2—>COOCH3—>COOH—. The higher radioprotective effect was observed for more effective uncouplers of the processes of oxidative phosphorylation and respiration of mitochondria, DBA1 preparations with OH-groups in the 2nd position of the benzene rings (80-100%), than for DBA2 with OH- in the 4th position (40-60%). The radioprotective effect of DBA was related to their antioxidant activity during and after irradiation to a lesser extent than with their uncoupling efficiency. At the cellular level, the radioprotective mechanism of DBA is related to temporary hypoxia and inhibition of metabolism leading to inhibition of generation of ROS, radicals, and LPO products.
Similar content being viewed by others
References
Grebenyuk AN, Gladkikh VD. Modern condition and prospects for development of medicines for prevention and early treatment of radiation injures. Radiats. Biol. Radioekol. 2019;59(2):132-149.
Gudkov SV, Popova NR, Bruskov VI. Radioprotective substances: history, trends and prospects. Biophysics. 2015;60(4):659-667.
Cojocaru AF. Molecular and cell mechanisms of radioprotective action of salicylanilides under their effect before γ-irradiation of laboratory animals in sublethal and fetal doses. Mezhdunarod. Zh. Priklad. Fundament. Issled. 2019;(8):14-24.Russian.
Cojocaru AF. Protonophoric and antioxidant action of natural flavonoids and sulfur-containing dibasic acid synthetic and other phenolic compounds. Mezhdunarod. Zh. Priklad. Fundament. Issled. 2014;(11-1):39-49.
Cojocaru AF, Yurov SS, Dmitrievskiy IM. Effects on ballistospor yeast of secondary biogenic radiation induced by ultraweak gamma radiation and natural background radiation. Mezhdunarod. Zh. Priklad. Fundament. Issled. 2016;(10-3):431-441. Russian.
Kostyuk VA, Potapovich AI. Bioradicals and Bioantioxidants. Minsk, 2004. Russian.
Krasochko PA, Yakubovskii MV, Yatusevich ASI. Diseases of Farm Animals. Minsk, 2005. Russian.
Skulachev VP, Skulachev MV, Zinovkin RA, Severin FF, Antonenko JN, Zorov DB, Plotnikov EJ, Isaev NK, Silachev DN, Knorre DA. Patent RU No. 2527519. Mild cationic mitochondrial uncouplers. Bull. No. 25. Published September 10, 2014.
Popov AM, Krivoshapko ON, Artyukov AA, Klimovich AA, Osipov AN, Korepanova EA. A study of the antioxidant and membranotropic activities of luteolin using different model systems. Biophysics. 2016;61(6):843-850.
Proskurnina EV, Vladimirov YuA. Free radicals as participants in regulatory and pathological processes. Basic Sciences for Medicine: Biophysical Medical Technologies. Grigor’ev AI, Vladimirov YuA, eds. Moscow, 2015. Vol. 1. P. 38-71. Russian.
Amin JK, Xiao L, Pimental DR, Pagano PJ, Singh K, Sawyer DB, Colucci WS. Reactive oxygen species mediate alpha-adrenergic receptor-stimulated hypertrophy in adult rat ventricular myocytes. J. Mol. Cell. Cardiol. 2001;33(1):131-139. doi: https://doi.org/10.1006/jmcc.2000.1285
Dzhatdoeva AA, Proskurnina EV, Nesterova AM, Dubinkin IV, Gaponova TV, Obydenny SI, Vladimirov YuA. Mitochondria as a source of superoxide anion radical in human platelets. Biochem. (Moscow), Suppl. Ser. A: Membrane Cell Biol. 2018;12(1):43-49.
Grivennikova VG, Vinogradov AD. Generation of superoxide by the mitochondrial Complex I. Biochim. Biophys. Acta. 2006;1757(5-6):553-561. doi: https://doi.org/10.1016/j.bbabio.2006.03.013
Grivennikova VG, Vinogradov AD. Partitioning of superoxide and hydrogen peroxide production by mitochondrial respiratory complex I. Biochim. Biophys. Acta. 2013;1827(3):446-454. doi: https://doi.org/10.1016/j.bbabio.2013.01.002
Kushnareva Y, Murphy AN, Andreyev A. Complex I-mediated reactive oxygen species generation: modulation by cytochrome c and NAD(P)+ oxidation-reduction state. Biochem. J. 2002;368(Pt 2):545-553. doi: https://doi.org/10.1042/BJ20021121
Sharapov MG, Novoselov VI, Gudkov SV. Radioprotective Role of Peroxiredoxin 6. Antioxidants (Basel). 2019;8(1):15. doi: https://doi.org/10.3390/antiox8010015
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 1, pp. 62-69, March, 2021
Rights and permissions
About this article
Cite this article
Cojocaru, A.F. Molecular Energotropic and Cellular Mechanism of the Radioprotective Action of Sulfur-Containing Biphenolic Dibasic Acids under Conditions of Lethal γ-Irradiation. Bull Exp Biol Med 171, 164–171 (2021). https://doi.org/10.1007/s10517-021-05189-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-021-05189-1