Abstract
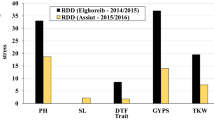
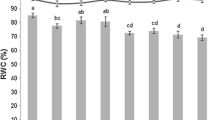
Drought is one of the important abiotic stresses that affects the phenological stages and crop productivity. Phenological stages can seriously influence the crop yield and quality. In this study, phenological traits including days to germination, days to tillering, days to stem elongation, days to head emergence, days to dough development in kernel, days to physiological maturity, number of grains per spike, thousand kernels weight, grain yield were evaluated in 148 commercial barley cultivars in both years under normal and drought stress conditions. In molecular experiment, the molecular profile of barley cultivars was assessed by 14 AFLP primer combinations and 32 SSRs loci. The phenotypic results showed the high level of diversity between studied cultivars. The studied barley cultivars were divided into two subgroups. Linkage disequilibrium analysis revealed that r2 values among all possible markers pairs have an average value of 0.0178. Mixed linear model procedure showed that totally, 137 loci had significant association with nine investigated traits under normal and drought stress conditions. The identified markers potentially can be used in marker-assisted selection in barley drought resistant breeding programs.

Similar content being viewed by others
Availability of data and materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Aghnoum R, Marcel TC, Johrde A, Pecchioni N, Schweizer P, Niks RE (2010) Basal host resistance of barley to powdery mildew: connecting quantitative trait loci and candidate genes. MPMI 23(1):91–102
Bamshad M, Wooding S, Salisbury BA, Stephens JC (2004) Deconstructing the relationship between genetics and race. Nat Rev Genet 5(8):598
Barrett JC, Fry B, Maller J, Daly M (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21(2):263–265
Caldwell KS, Russell J, Langridge P, Powell W (2006) Extreme populationdependent linkage disequilibrium detected in an inbreeding plant species, Hordeum vulgare. Genetics 172(1):557–567
Campoli C, Drosse B, Searle I, Coupland G, von Korff M (2012) Functional characterisation of HvCO1, the barley (Hordeum vulgare) flowering time ortholog of CONSTANS. Plant J 69:868–880
Cattivelli L, Rizza F, Badeck FW, Mazzucotelli E, Mastrangelo AM, Francia E, Marè C, Tondelli A, Stanca AM (2008) Drought tolerance improvement in crop plants: an integrated view from breeding to genomics. Field Crops Res 105:1–14
Chloupek O, Forster BP, Thomas WTB (2006) The effect of semi-dwarf genes on root system size in field-grown barley. Theor Appl Genet 122(5):779–786
Dabbert T, Okagaki RJ, Cho S, Heinen S, Boddu J, Muehlbauer GJ (2010) The genetics of barley low-tillering mutants: low number of tillers-1 (lnt1). Theor Appl Genet 121:705–715
Distelfeld A, Li C, Dubcovsky J (2009) Regulation of flowering in temperate cereals. Curr Opin Plant Biol 12(2):178–184
Ellis RP, Forster BP, Robinson D, Handley LL, Gordon DC, Russell JR, Powell W (2000) Wild barley: a source of genes for crop improvement in the 21st century. J Exp Bot 51:9–17
Fan Y, Zhou G, Shabala S, Chen ZH, Cai S, Li C, Zhou M (2016) Genome-wide association study reveals a new QTL for salinity tolerance in barley (Hordeum vulgare L.). Front Plant Sci 7:946
Flint-Garcia S, Thornsberry J, Buckler E (2003) Structure of linkage disequilibrium in plants. Annu Rev Plant Biol 54:357–374
Guo P, Baum M, Grando S, Ceccarelli S, Bai G, Li R, von Korff M, Varshney RK, Graner A, Valkoun J (2009) Differentially expressed genes between drought-tolerant and drought-sensitive barley genotypes in response to drought stress during the reproductive stage. J Exp Bot 60:3531–3544
Haaning AM, Smith KP, Brown-Guedira GL, Chao S, Tyagi P, Muehlbauer GJ (2020) Natural genetic variation underlying tiller development in barley (Hordeum vulgare L). Genes Genomes Genet 10(4):1197–1212
Haseneyer G, Stracke S, Paul C, Einfeldt C, Broda A, Piepho HP, Graner A, Geiger HH (2010) Population structure and phenotypic variation of a spring barley world collection set up for association studies. Plant Breed 129(3):271–279
Haussmann BIG, Obilana AB, Ayiecho PO, Blum A, Schipprack W, Geiger HH (2000) Yield and yield stability of four population types of grain sorghum in a semi-arid area of Kenya. Crop Sci 40:319–329
Hay RKM, Ellis R (1998) The control of flowering in wheat and barley: what recent advances in molecular genetics can reveal. Ann Bot 82(5):541–554
Jin L, Lu Y, Xiao P, Sun M, Corke H, Bao JS (2010) Genetic diversity and population structure of a diverse set of rice germplasm for association mapping. Theor Appl Genet 121:475–487
Kraakman ATW, Martõ’nez F, Mussiraliev B, van Eeuwijk FA, Niks RE (2006) Linkage disequilibrium mapping of morphological, resistance, and other agronomically relevant traits in modern spring barley cultivars. Mol Breed 17:41–58
Lewis S, Faricelli ME, Appendino ML, Valárik M, Dubcovsky J (2008) The chromosome region including the earliness per se locus Eps-Am 1 affects the duration of early developmental phases and spikelet number in diploid wheat. J Exp Bot 59(13):3595–3607
Martin JM, Frohberg RC, Morris CF, Talbert LE, Giroux MJ (2001) Milling and bread baking traits associated with puroindoline sequence type in hard red spring wheat. Crop Sci 41:228–234
Massman J, Cooper B, Horsley R, Neate S, Dill-Macky R, Chao S, Dong Y, Schwarz P, Muehlbauer GJ, Smith KP (2011) Genome wide association mapping of Fusarium head blight resistance in contemporary barley breeding germplasm. Mol Breed 27:439–454
Mehravaran L, Fakheri B, Sharifi-Rad J (2014) Localization of quantitative trait loci (QTLs) controlling drought tolerance in barley. Int J Biosci 5(7):248–259
Moose SP, Mumm RH (2008) Molecular plant breeding as the foundation for 21st century crop improvement. Plant Physiol 147(3):969–977
Morrel PL (2011) Hordeum. In: Kole C (ed) Wild crop relatives: genomic and breeding resources, cereals. Springer, New York, pp 309–320
Morris CF (2002) Puroindoline: the molecular genetic basis of wheat grain hardness. Plant Mol Biol 48:633–647
Nevo E, Chen G (2011) Drought and salt tolerances in wild relatives for wheat and barley improvement. Plant Cell Environ 33:670–685
Okagaki RJ, Haaning A, Bilgic H, Heinen S, Druka A, Bayer M, Waugh R, Muehlbauer GJ (2018) ELIGULUM-A regulates lateral branch and leaf development in barley. Plant Physiol 176:2750–2760
Pasam RK, Sharma R, Malosetti M, Eeuwijk FAV, Haseneyer G, Kilian B, Graner A (2012) Genome-wide association studies for agronomical traits in a worldwide spring barley collection. BMC Plant Biol 12:16
Payne RW, Murray DA, Harding SA, Baird DB, Soutar DM (2011) An introduction to GenStat for Windows, 14th edn. VSN International, Hemel Hempstead
Pritchard JK, Stephanes M, Rosenberg NA (2000) Association mapping in structured populations. Am J Hum Genet 67:170–181
Ramsay L, Comadran J, Druka A, Marshall DF, Thomas WTB, Macaulay M, MacKenzie K, Simpson C, Fuller J, Bonar N, Hayes PM, Lundqvist U, Franckowiak JD, Close TJ, Muehlbauer GJ, Waugh R (2011) INTERMEDIUM-C, a modifier of lateral spikelet fertility in barley, is an ortholog of the maize domestication gene TEOSINTE BRANCHED 1. Nat Genet 43(2):169–172
Rostoks N, Ramsay L, MacKenzie K, Cardle L, Bhat PR, Roose ML, Svensson JT, Stein N, Varshney RK, Marshall DF, Graner A, Close TJ, Waugh R (2006) Recent history of artificial outcrossing facilitates whole-genome association mapping in elite inbred crop varieties. Proc Natl Acad Sci USA 103(49):18656–18661
Sallam A, Alqudah AM, Dawood MFA, Baenziger PS, Börner A (2019) Drought stress tolerance in wheat and barley: advances in physiology, breeding and genetics Research. Int J Mol Sci 20:3137
Sameri M, Takeda K, Komatsuda T (2006) Quantitative trait loci controlling agro nomic traits in recombinant inbred lines from a cross of oriental- and occidental-type barley cultivars. Breed Sci 56:243–252
Schulze TG, McMahon FJ (2002) Genetic association mapping at the crossroads: which test and why? Overview and practical guidelines. Am J Med Genet 114(1):1–11
Sharafi S, Ghassemi-Golezani K, Mohammadi S, Lak S, Sorkhy B (2014) Evaluation of barley genotypes (Hordeum vulgare L.) by drought tolerance indices and multivariate analysis. Int J Biosci 4:117–129
Stansfield WD (1991) Theory and problems in genetics. McGraw-Hill, New York
Stoskopf NF, Nathaniel RK, Reinbergs E (1974) Comparison of spring wheat and barley with winter wheat: yield components in Ontario. Agron J 66:747–750
Subhani GM, Abdullah M, Ahmad J, Anwar J, Hussain M, Mahmood A (2015) Identification of drought tolerant genotypes of barley (Hordeum vulgare L.) through stress tolerance indices. J Anim Plant Sci 25:686–692
Takeda S, Matsuoka M (2008) Genetic approaches to crop improvement: responding to environmental and population changes. Nat Rev Genet 9:444–457
Tavakol E, Okagaki R, Verderio G, Shariati V, Hussien A (2015) The barley Uniculme4 gene encodes a BLADE-ON-PETIOLE-like protein that controls tillering and leaf patterning. Plant Physiol 168:164–174
Teulat B, Merah O, Souyris I, This D (2001) QTLs for agronomic traits from a Mediterranean barley progeny grown in several environments. Theor Appl Genet 103:774–787
Tsilo TJ, Simsek S, Ohm JB, Hareland GA, Chao S, Anderson JA (2011) Quantitative trait loci influencing endosperm texture, dough-mixing strength, and breed-making properties of the hard red spring wheat breeding lines. Genome 54:460–470
Visioni A, Tondelli A, Francia E, Pswarayi A, Malosetti M, Russell J, Thomas W, Waugh R, Pecchioni N, Romagosa J, Comadran J (2013) Genome-wide association mapping of frost tolerance in barley (Hordeum vulgare L.). BMC Genomics 14:424
Voss-Fels KP, Robinson H, Mudge SR, Richard C, Newman S, Wittkop B et al (2018) VERNALIZATION1 modulates root system architecture in wheat and barley. Mol Plant 11:226–229
Wang J, Yang J, Jia Q, Zhu J, Shang Y, Hua W, Zhou M (2014) A new QTL for plant height in barley (Hordeum vulgare L.) showing no negative effects on grain yield. PLoS ONE 9(2):e90144
Wojciechowski T, Gooding MJ, Ramsay L, Gregory PJ (2009) The effects of dwarfing genes on seedling root growth of wheat. J Exp Bot 60(9):2565–2573
Wójcik-Jagła M, Fiust A, Kościelniak J, Rapacz M (2017) Association mapping of drought tolerance-related traits in barley to complement a traditional biparental QTL mapping study. Theor Appl Genet 131:167–181
Yu J, Pressoir G, Briggs WH, Bi IV, Yamasaki M, Doebley JF, McMullen MD, Gaut BS, Nielsen DM, Holland JB (2006) A unified mixed-model method for association mapping that accounts for multiple levels of relatedness. Nat Genet 38:203–208
Zhu C, Gore M, Buckler ES, Yu J (2008) Status and prospects of association mapping in plants. Plant Genome 1(1):5–20
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization, Methodology, Formal analysis and investigation: [Mitra Jabbari, Barat Ali Fakheri, Nafiseh Mahdi Nezhad, Reza Ataei, Zahra Koochakpour]; Resources: [Reza Aghnoum]; Writing—review and editing: [Reza Darvishzadeh, Mitra Razi].
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Communicated by A. Börner.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jabbari, M., Fakheri, B.A., Aghnoum, R. et al. Identification of DNA markers associated with phenological traits in spring barley (Hordeum vulgare L.) under drought stress conditions. CEREAL RESEARCH COMMUNICATIONS 50, 171–178 (2022). https://doi.org/10.1007/s42976-021-00181-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42976-021-00181-x