Abstract
Aims
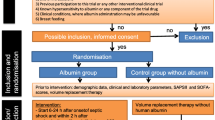
Sepsis and septic shock are common causes of hospitalization and mortality in patients with cirrhosis. There is no data on the choice of fluid and resuscitation protocols in sepsis-induced hypotension in cirrhosis.
Methods
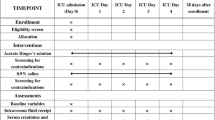
In this open-label trial conducted at a single center, we enrolled 308 cirrhotics with sepsis-induced hypotension and randomized them to receive either 5% albumin or normal saline. The primary endpoint was a reversal of hypotension [mean arterial pressure, MAP, ≥ 65 mmHg] at 3 h. Secondary endpoints included serial effects on heart rate, arterial lactate and urine output.
Results
154 patients each received 5% albumin (males, 79.8%, mean MAP 52.9 ± 7.0 mm Hg) or 0.9% saline (85.1%, 53.4 ± 6.3 mm Hg) with comparable baseline parameters and liver disease severity. Reversal of hypotension was higher in patients receiving 5% albumin than saline at the end of one hour [25.3% and 11.7%, p = 0.03, Odds ratio (95% CI)—1.9 (1.08–3.42)] and at the end of three hours [11.7% and 3.2%, p = 0.008, 3.9 (1.42–10.9)]. Sustained reduction in heart rate and hyperlactatemia (p < 0.001) was better in the albumin group. At one week, the proportion of patients surviving was higher in the albumin group than those receiving saline (43.5% vs 38.3%, p = 0.03). Female gender and SOFA ≥ 11 were predictors of non-response to fluid.
Conclusions
5% human albumin is safe and beneficial in reversing sepsis-induced hypotension compared to normal saline in patients with cirrhosis improving clinically assessable parameters of systemic hemodynamics, tissue perfusion and in-hospital short-term survival of cirrhosis patients with sepsis.
Graphic abstract



Similar content being viewed by others
Abbreviations
- SAFE:
-
Saline versus Albumin Fluid Evaluation
- MELD:
-
Model for end-stage liver disease
- CTP:
-
Child-Pugh Score
- MAP:
-
Mean arterial pressure
- SOFA:
-
Sequential organ failure assessment
- SIRS:
-
Systemic inflammatory response syndrome
- OR:
-
Odds ratio
- CI:
-
Confidence interval
- KM:
-
Kaplan Meier
References
Jawad I, Lukšic I, Rafnsson SB. Assessing available information on the burden of sepsis: global estimates of incidence, prevalence and mortality. J Glob Health 2012;2:010404
Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001;345:1368–1377
Finfer S, Liu B, Taylor C, et al. Resuscitation fluid use in critically ill adults. An international cross-sectional study in 391 intensive care units. Crit Care 2010;14:R185
Finfer S, Bellomo R, Boyce N, French J, Myburgh J, Norton R. The SAFE study investigators. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med. 2004;350:2247–2256.
Delaney AP, Dan A, McCaffrey J, Finfer S. The role of albumin as a resuscitation fluid for patients with sepsis: a systematic review and meta-analysis. Crit Care Med 2011;39:386–91
Caraceni P, Tufoni M, Bonavita ME. Clinical use of albumin. Blood Transfus 2013;11(4):s18-25
Olson JC, Wendon JA, Kramer DJ, et al. Intensive care of the patient with cirrhosis. Hepatology 2011;54:1864–1872
Moreau R, Hadengue A, Soupison T, et al. Septic shock in patients with cirrhosis: hemodynamic and metabolic characteristics and intensive care unit outcome. Crit Care Med 1992;20(6):746–750
Glassford NJ, Eastwood GM, Bellomo R. Physiological changes after fluid bolus therapy in sepsis: a systematic review of contemporary data. Crit Care 2014;18(6):696
Xu JY, Chen QH, Xie JF, et al. Comparison of the effects of albumin and crystalloid on mortality in adult patients with severe sepsis and septic shock: a meta-analysis of randomized clinical trials. Crit Care 2014;18:702
Tollefson BJ, Jones AE. Does resuscitation with albumin-containing solutions improve mortality in sepsis? Ann Emerg Med 2012;59:432–433
Finfer S. Clinical controversies in the management of critically ill patients with severe sepsis: resuscitation fluids and glucose control. Virulence 2013;5:200–205
Levy MM, Evans LE, Rhodes A. The surviving sepsis campaign bundle: 2018 update. Intensive Care Med 2018;44(6):925–8
Daviaud F, Grimaldi D, Dechartres A, Charpentier J, Geri G, Marin N, et al. Timing and causes of death in septic shock. Ann Intensive Care 2015;5:16
Varon J, Baron RM. A current appraisal of evidence for the approach to sepsis and septic shock. Ther Adv Infect Dis 2019;6:2049936119856517
Roger C, Zieleskiewicz L, Demattei C, Lakhal K, Piton G, Louart B, et al. Time course of fluid responsiveness in sepsis: the fluid challenge revisiting (FCREV) study. Crit Care 2019;23:179
Tu KH, Jenq CC, Tsai MH, Hsu HH, Chang MY, Tian YC, et al. Outcome scoring systems for short-term prognosis in critically ill cirrhotic patients. Shock 2011;36(5):445–450
Wehler M, Kokoska J, Reulbach U, Hahn EG, Strauss R. Short-term prognosis in critically ill patients with cirrhosis assessed by prognostic scoring systems. Hepatology 2001;34(2):255–261
Leibovici LA, GafterGvili M, Paul N, et al. Relative tachycardia in patients with sepsis: an independent risk factor for mortality. QJM 2007;100:629–634
The SAFE Study Investigators. Impact of albumin compared to saline on organ function and mortality of patients with severe sepsis. Intensive Care Med 2011;37:86–96
Marty P, Roquilly A, Vallée F, et al. Lactate clearance for death prediction in severe sepsis or septic shock patients during the first 24 hours in intensive care unit: an observational study. Ann Intensive Care 2013;3:3
Jansen TC, van Bommel J, Mulder PG, Lima AP, van der Hoven B, Rommes JH, Snellen FT, Bakker J. Prognostic value of blood lactate levels: does the clinical diagnosis at admission matter? J Trauma 2009;66:377–385
Nichol A, Bailey M, Egi M, et al. Dynamic lactate indices as predictors of outcome in critically ill patients. Crit Care 2011;15(5):R242
Zhou F, Cove ME, Peng ZY, et al. Normal saline resuscitation worsens hyperlactatemia in experimental sepsis. Crit Care 2012;16(Suppl 1):P253
Amathieu R, Al-Khafaji A, Sileanu FE, Foldes E, DeSensi R, Hilmi I, Kellum JA. Significance of oliguria in critically ill patients with chronic liver disease. Hepatology 2017;66(5):1592–1600
Xu J-Y, Chen Q-H, Xie J-F, et al. Comparison of the effects of albumin and crystalloid on mortality in adult patients with severe sepsis and septic shock: a meta-analysis of randomized clinical trials. Crit Care 2014;18(6):702
Coccolini F, Sartelli M, Catena F, Ceresoli M, Montori G, Ansaloni L. Early goal-directed treatment versus standard care in management of early septic shock: meta-analysis of randomized trials. J Trauma Acute Care Surg 2016;81:971–978
Gyawali B, Ramakrishna K, Dhamoon AS. Sepsis: The evolution in definition, pathophysiology, and management. SAGE Open Med 2019;7:2050312119835043
Malbrain MLNG, Van Regenmortel N, Saugel B, et al. Principles of fluid management and stewardship in septic shock: it is time to consider the four D’s and the four phases of fluid therapy. Ann Intensive Care 2018;8:66
Jozwiak M, Monnet X, Teboul JL. Prediction of fluid responsiveness in ventilated patients. Ann Transl Med 2018;6:352
Jones AE, Trzeciak S, Kline JA. The Sequential Organ Failure Assessment score for predicting outcome in patients with severe sepsis and evidence of hypoperfusion at the time of emergency department presentation. Crit Care Med 2009;37:1649–1654
Sakr Y, Elia C, Mascia L, Barberis B, Cardellino S, Livigni S, Fiore G, Filippini C, Ranieri VM. The influence of gender on the epidemiology of and outcome from severe sepsis. Crit Care 2013;17(2):R50
Pietropaoli AP, Glance LG, Oakes D, Fisher SG. Gender differences in mortality in patients with severe sepsis or septic shock. Gend Med 2010;7:422–437
Failla KR, Connelly CD, Ecoff L, Macauley K, Bush R. Does gender matter in septic patient outcomes? J Nurs Scholarsh 2019;51(4):438–448
Campanelli F, Landoni G, Cabrini L, Zangrillo A. Gender differences in septic intensive care unit patients. Minerva Anestesiol 2018;84(4):504–508
Sterling SA, Miller WR, Pryor J, Puskarich MA, Jones AE. The impact of timing of antibiotics on outcomes in severe sepsis and septic shock: a systematic review and meta-analysis. Crit Care Med 2015;43(9):1907–1915
Liu VX, Fielding-Singh V, Greene JD, Baker JM, Iwashyna TJ, Bhattacharya J, Escobar GJ. The timing of early antibiotics and hospital mortality in sepsis. Am J Respir Crit Care Med 2017;196(7):856–863
Kim RY, Ng AM, Persaud AK, Furmanek SP, Kothari YN, Price JD, Wiemken TL, Saad MA, Guardiola JJ, Cavallazzi RS. Antibiotic timing and outcomes in sepsis. Am J Med Sci 2018;355(6):524–549
Martínez ML, Plata-Menchaca EP, Ruiz-Rodríguez JC, Ferrer R. An approach to antibiotic treatment in patients with sepsis. J Thorac Dis 2020;12(3):1007–1021
Funding
None.
Author information
Authors and Affiliations
Contributions
CAP and SKS conceived and designed the trial. CAP, SKS, RM were the chief investigators and oversaw the trial throughout. MKS, RM, AC, AJ were trial examiners. CAP, RM, AB, GK, LGM, VS, PMA monitored the data, did analyses, and provided critical feedback to study design and activities. All authors contributed to the interpretation of data, drafting of the report and decided on its content. All authors approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have none.
Ethical approval
This trial was approved by the Institutional Ethical Review Board and was done in accordance with the tenets of the Declaration of Helsinki. Written informed consent (presuming patients were unable to provide informed consent) was obtained from all of the respective participant’s family member before enrollment.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Philips, C.A., Maiwall, R., Sharma, M.K. et al. Comparison of 5% human albumin and normal saline for fluid resuscitation in sepsis induced hypotension among patients with cirrhosis (FRISC study): a randomized controlled trial. Hepatol Int 15, 983–994 (2021). https://doi.org/10.1007/s12072-021-10164-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-021-10164-z