Abstract
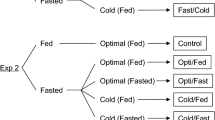
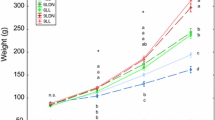
We examined the combined effects of fasting in freshwater and water temperature during the transition from freshwater to seawater on growth/metabolic parameters in juvenile chum salmon Oncorhynchus keta. Juveniles of 1 g in freshwater were first subjected to a 5-day fasting period at 10 or 5 °C, acclimated to either 10 or 5 °C seawater and fed ad libitum for 30 days. Control fish were transferred from 10 °C freshwater to 10 °C seawater and fed ad libitum throughout the experimental period. Serum insulin-like growth factor (IGF)-I was measured to evaluate growth status/potential and liver glycogen as an index of energy storage. Fasting in freshwater for 5 days negatively affected body size. Growth of juveniles kept at colder temperatures was retarded in seawater for at least 20 days, which may partly be explained by a lower feeding rate in cold seawater. Serum IGF-I levels were lower in fasted fish in freshwater at both temperatures and colder seawater had a negative effect on restoring serum IGF-I levels after refeeding for 20 days. Liver glycogen content was low in fish fasted in freshwater for 5 days. After refeeding in seawater for 10 days, liver glycogen content increased significantly in juveniles kept at colder temperatures. Colder water temperatures in both salinities positively affected glycogen content for 30 days after transfer to seawater, suggesting that juveniles allocated energy stores to the liver rather than growth under suboptimal feeding and temperature conditions. The findings of the present study suggested that relatively cold freshwater could negatively affect juvenile chum salmon growth soon after sea entry.










Similar content being viewed by others
References
Bax NJ (1983) Early marine mortality of marked juvenile chum salmon (Oncorhynchus keta) released into Hood Canal, Puget Sound, Washington, in 1980. Can J Fish Aquat Sci 40:426–435
Beacham TD, Neville CM, Tucker S, Trudel M (2017) Is there evidence for biologically significant size-selective mortality of coho salmon during the first winter of marine residence? Trans Am Fish Soc 146:395–407
Beacham TD, Araujo HA, Tucker S, Trudel M (2018) Validity of inferring size-selective mortality and a critical size limit in Pacific salmon from scale circulus spacing. PLoS ONE 13:e0199418
Beamish RJ, Mahnken C (2001) A critical size and period hypothesis to explain natural regulation of salmon abundance and the linkage to climate and climate change. Prog Oceanogr 49:423–437
Beckman BR (2011) Perspectives on concordant and discordant relations between insulin-like growth factor 1 (IGF1) and growth in fishes. Gen Comp Endocrinol 170:233–252
Beckman BR, Shimizu M, Gadberry BA, Cooper KA (2004a) Response of the somatotropic axis of juvenile coho salmon to alterations in plane of nutrition with an analysis of the relationships among growth rate and circulating IGF-I and 41 kDa IGFBP. Gen Comp Endocrinol 135:334–344
Beckman BR, Shimizu M, Gadberry BA, Parkins PJ, Cooper KA (2004b) The effect of temperature change on the relations among plasma IGF-I, 41-kDa IGFBP, and growth rate in postsmolt coho salmon. Aquaculture 241:601–619
Chang C-H, Huang J-J, Yeh C-Y, Tang C-H, Hwang L-Y, Lee T-H (2018) Salinity effects on strategies of glycogen utilization in livers of euryhaline milkfish (Chanos chanos) under hypothermal stress. Font Physiol 9:81
Claireaux G, Chabot D (2016) Responses by fishes to environmental hypoxia: integration through fry’s concept of aerobic metabolic scope. J Fish Biol 88:232–251
Daughaday WH, Rotwein P (1989) Insulin-like growth factors I and II. Peptide, messenger ribonucleic acid and gene structures, serum, and tissue concentrations. Endocr Rev 10:68–91
Dreiling CE, Brown DE, Casale L, Kelly L (1987) Muscle glycogen: comparison of iodine binding and enzyme digestion assays and application to meat samples. Meat Sci 20:167–177
Duffy EJ, Beauchamp DA (2011) Rapid growth in the early marine period improves the marine survival of Chinook salmon (Oncorhynchus tshawytscha) in Puget Sound, Washington. Can J Fish Aquat Sci 68:232–240
Duncan DH, Beaudreau AH (2019) Spatiotemporal variation and size-selective predation on hatchery- and wild-born juvenile chum salmon at marine entry by nearshore fishes in Southeast Alaska. Mar Coast Fish 11:372–390
Farley EV, Moss JH, Beamish RJ (2007) A review of the critical size, critical period hypothesis for juvenile Pacific salmon. N Pac Anadr Fish Comm Bull 4:311–317
Fukuwaka M, Suzuki T (2002) Early sea mortality of mark-recaptured juvenile chum salmon in open coastal waters. J Fish Biol 60:3–12
Gabillard JC, Weil C, Rescan PY, Navarro I, Gutierrez J, Le Bail PY (2005) Does the GH/IGF system mediate the effect of water temperature on fish growth? A review. Cybium 29:107–117
Healey MC (1982) Timing and relative intensity of size-selective mortality of juvenile chum salmon (Oncorhynchus keta) during early sea life. Can J Fish Aquat Sci 39:952–957
Hoar WS (1988) The physiology of smolting salmonids. In: Hoar WS, Randall D (eds) Fish physiology 11B. Academic Press, Orland, pp 274–343
Honda K, Kawakami T, Suzuki K, Watanabe K, Saito T (2017) Growth rate characteristics of juvenile chum salmon Oncorhynchus keta originating from the Pacific coast of Japan and reaching Konbumori, eastern Hokkaido. Fish Sci 83:987–996
Honda K, Shirai K, Komatsu S, Saito T (2020) Sea-entry conditions of juvenile chum salmon Oncorhynchus keta that improve post-sea-entry survival: a case study of the 2012 brood-year stock released from the Kushiro River, eastern Hokkaido, Japan. Fish Sci 86:783–792
Kaneko N, Taniyama N, Inatani Y, Nagano Y, Fujiwara M, Torao M, Miyakoshi Y, Shimizu M (2015) Circulating insulin-like growth factor I in juvenile chum salmon: relationship with growth rate and changes during downstream and coastal migration in northeastern Hokkaido, Japan. Fish Physiol Biochem 41:991–1003
Kaneko N, Torao M, Koshino Y, Fujiwara M, Miyakoshi Y, Shimizu M (2019) Evaluation of growth status using endocrine growth indices, insulin-like growth factor (IGF)-I and IGF-binding protein-1b, in out-migrating juvenile chum salmon. Gen Comp Endocrinol 274:50–59
Kashiwagi M, Sato R (1969) Studies on the osmoregulation of the chum salmon, Oncorhynchus keta (Walbaum). I. The tolerance of eyed period eggs, alevins and fry of the chum salmon to sea water. Tohoku J Agric Res 20:41–47
Kasugai K, Takeuchi K, Miyakoshi Y, Nagata M (2014) Estimation of number of downstream migrating chum salmon fry in the Nishibetsu River in 2006. Sci Rep Hokkaido Fish Res Inst 85:37–40 (In Japanese with English abstract)
Kitada S (2014) Japanese chum salmon stock enhancement: current perspective and future challenges. Fish Sci 80:237–249
Le Roith D, Bondy C, Yakar S, Liu JL, Butler A (2001) The somatomedin hypothesis: 2001. Endocr Rev 22:53–74
McCormick SD (2013) Smolt physiology and endocrinology. In: McCormick SD et al (eds) Euryhaline fishes. Academic Press, Oxford, UK, pp 199–251
Miyakoshi Y, Nagata M, Kitada S, Kaeriyama M (2013) Historical and current hatchery programs and management of chum salmon in Hokkaido, northern Japan. Rev Fish Sci 21:469–479
Mizuno S (2012) Studies on improvement of seed production techniques in Salmonids and Osmerids. Aqua-Biosci 5:103–143
Morita K, Nakashima A (2015) Temperature seasonality during fry out-migration influences the survival of hatchery-reared chum salmon Oncorhynchus keta. J Fish Biol 87:1111–1117
Morita K, Saito T, Miyakoshi Y, Fukuwaka MA, Nagasawa T, Kaeriyama M (2006) A review of Pacific salmon hatchery programmes on Hokkaido Island, Japan. ICES J Mar Sci 63:1353–1363
Morita K, Nakashima A, Kikuchi M (2015) River temperature drives salmon survivorship: is it determined prior to ocean entry? Roy Soc Open Sci 2:140312
Moss JH, Beauchamp DA, Cross AD, Myers KW, Farley EV, Murphy JM, Helle JH (2005) Evidence for size-selective mortality after the first summer of ocean growth by pink salmon. Trans Am Fish Soc 134:1313–1322
Nagata M, Miyakoshi Y, Ando D, Fujiwara M, Sawada M, Shimada H, Asami H (2007) Influence of coastal seawater temperature on the distribution and growth of juvenile chum salmon, with recommendations for altered release strategies. N Pac Anadr Fish Comm Bull 4:223–235
Nagata M, Miyakoshi Y, Urabe H, Fujiwara M, Sasaki Y, Kasugai K, Torao M, Ando D, Kaeriyama M (2012) An overview of salmon enhancement and the need to manage and monitor natural spawning in Hokkaido, Japan. Environ Biol Fish 94:311–323
Nagata M, Miyakoshi Y, Fujiwara M, Kasugai K, Ando D, Torao M, Saneyoshi H, Irvine JR (2016) Adapting Hokkaido hatchery strategies to regional ocean conditions can improve chum salmon survival and reduce variability. N Pac Anadr Fish Comm Bull 6:73–85
Nakamura S, Kaneko N, Nonaka T, Kurita D, Miyakoshi Y, Shimizu M (2019) Fasting in freshwater severely affects growth of juvenile chum salmon when entering cold seawater. Fish Sci 85:655–665
Ohlsson C, Mohan S, Sjögren K, Tivesten Å, Isgaard J, Isaksson O, Jansson JO, Svensson J (2009) The role of liver-derived insulin-like growth factor-I. Endocr Rev 30:494–535
Picha ME, Turano MJ, Beckman BR, Borski RJ (2008) Endocrine biomarkers of growth and applications to aquaculture: A minireview of growth hormone, insulin-like growth factor (IGF)-I, and IGF-binding proteins as potential growth indicators in fish. N Am J Aquacult 70:196–211
Quabius ES, Balm PHM, Bonga SEW (1997) Interrenal stress responsiveness of tilapia (Oreochromis mossambicus) is impaired by dietary exposure to PCB 126. Gen Comp Endocrinol 108:472–482
Reinecke M (2010) Influences of the environment on the endocrine and paracrine fish growth hormone-insulin-like growth factor-I system. J Fish Biol 76:1233–1254
Saito T, Shimizu I, Seki J, Kaga T, Hasegawa E, Saito H, Nagasawa K (2010) Can research on the early marine life stage of juvenile chum salmon Oncorhynchus keta forecast returns of adult salmon? A case study from eastern Hokkaido, Japan. Fish Sci 76:909–920
Saito T, Kaga T, Hasegawa E, Nagasawa K (2011) Effects of juvenile size at release and early marine growth on adult return rates for Hokkaido chum salmon (Oncorhynchus keta) in relation to sea surface temperature. Fish Oceanogr 20:278–293
Salo EO (1991) Life history of chum salmon (Onocorhynchus keta). In: Groot C, Margolis L (eds) Pacific salmon life histories. UBC Press, Vancouver, BC, Canada, pp 231–310
Seki J (2005) Study of characteristics of feeding habitat of juvenile chum salmon and their food environment in the Pacific coastal waters, central part of Hokkaido. Bull Nat Salmon Resour Ctr 7:1–104 (In Japanese with English abstract)
Seki J (2013) Development of hatchery techniques for releasing juvenile chum salmon in Japan. J Fish Technol 6:69–82 ((In Japanese with English abstract))
Shimizu M, Swanson P, Fukada H, Hara A, Dickhoff WW (2000) Comparison of extraction methods and assay validation for salmon insulin-like growth factor-I using commercially available components. Gen Comp Endocrinol 119:26–36
Small BC, Peterson BC (2005) Establishment of a time-resolved fluoroimmunoassay for measuring plasma insulin-like growth factor I (IGF-I) in fish: effect of fasting on plasma concentrations and tissue mRNA expression of IGF-I and growth hormone (GH) in channel catfish (Ictalurus punctatus). Domest Anim Endocrinol 28:202–215
Takahashi S, Hasegawa K, Ito H, Ban M, Miyauchi Y (2016) Comparisons of growth of chum salmon fry released into rivers of which temperature and prey abundance conditions were different. Nippon Suisan Gakkaishi 82:559–568 (In Japanese with English abstract)
Taniyama N, Kaneko N, Inatani Y, Miyakoshi Y, Shimizu M (2016) Effects of seawater transfer and fasting on the endocrine and biochemical growth indices in juvenile chum salmon (Oncorhynchus keta). Gen Comp Endocrinol 236:146–156
Tucker S, Hipfner JM, Trudel M (2016) Size- and condition-dependent predation: a seabird disproportionately targets substandard individual juvenile salmon. Ecology 97:461–471
Urawa S, Beacham TD, Fukuwaka M, Kaeriyama M (2018) Ocean ecology of chum salmon. In: Beamish RJ (ed) The ocean ecology of Pacific salmon and Trout. American Fisheries Society, Bethesda, MD, pp 161–317
Vijayan MM, Moon TW (1992) Acute handling stress alters hepatic glycogen metabolism in food-deprived rainbow trout (Oncorhynchus mykiss). Can J Fish Aquat Sci 49:2260–2266
Vijayan MM, Maule AG, Schreck CB, Moon TW (1993) Hormonal control of hepatic glycogen metabolism in food-deprived, continuously swimming coho salmon (Oncorhynchus kisutch). Can J Fish Aquat Sci 50:1676–1682
Wertheimer AC, Thrower FP (2007) Mortality rates of chum salmon during their early marine residency. In: Grimes CB, Brodeur RD, Haldorson LJ, McKinnell SM (eds) The ecology of juvenile salmon in the northeast Pacific Ocean: regional comparisons. American Fisheries Society, Bethesda, MD, pp 233–247
Wong MKS, Nobata S, Hyodo S (2019) Enhanced osmoregulatory ability marks the smoltification period in developing chum salmon (Oncorhynchus keta). Comp Biochem Physiol A 238:110565
Zavolokin AV, Strezhneva EV (2013) Size-selective mortality of Sea of Okhotsk pink salmon in the ocean in the winter and spring. Russ J Mar Biol 39:501–508
Acknowledgements
We thank the staff of the Kitami Salmon Enhancement Programs Association for providing juvenile chum salmon. This work was supported by grants from the Japan Society for the Promotion of Science (JSPS), KAKENHI Grant Number 18K05801, and the JSPS Bilateral Joint Research Project (Open Partnership with Norway) Grant Number JPJSBP120209901. We acknowledge Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oikawa, J., Nakamura, S., Kaneko, N. et al. Effects of fasting and water temperatures during transition from freshwater to seawater on juvenile chum salmon growth and metabolism. Fish Sci 87, 579–588 (2021). https://doi.org/10.1007/s12562-021-01526-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-021-01526-5