Abstract
Echinacoside (ECH), acteoside (ACT), and isoacteoside (ISAT), the typical phenylethanoid glycosides (PhGs) in cistanches herba, have various pharmacological activities. However, the ECH, ACT and ISAT have extremely low oral bioavailability, which is related to their metabolism under the intestinal flora. Previous studies showed that intestinal metabolites were the hepatoprotective substances in vivo, but the research on whether PhGs has effects without intestinal bacteria has not been studied. In this paper, ECH, ACT and ISAT were incubated with human or rat intestinal bacteria for 36 h. After incubating with human bacteria for 36 h, three prototype compounds were not detected and were mainly biotransformed to 3-HPP and HT. In the network pharmacology, a total of 6 common targets were obtained by analysing the prototypes, the metabolites and the liver injury. It was found that the combinations of three metabolites and common targets were more stable than those of the prototypes and common targets by molecular docking. Meanwhile, hepatocellular apoptosis, proliferation, inflammation and oxidative responses might play important roles in the mechanisms of the metabolites exerting hepatoprotective activities. Then normal and pseudo-sterile mice experiments were adopted to further compare the hepatoprotective activities of prototypes and metabolites. Animal experiment results showed that the prototypes and the metabolites in the normal mice had significantly hepatoprotective activity. Interestingly, in the pseudo-germfree mice, the metabolites showed significant hepatoprotective effect, but the prototypes had not effect. It indicated that the prototype cannot exert liver protective activity without the effect of intestinal bacteria.
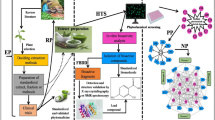
Graphic abstract









Similar content being viewed by others
Abbreviations
- ACT:
-
Acteoside
- AKR1B1:
-
Aldo–Keto Reductase Family 1 Member B
- AKR1B10:
-
Aldo–Keto Reductase Family 1 Member B10
- ALT:
-
Aminotransferase
- ANOVA:
-
Analysis of variance
- APP:
-
Amyloid-beta precursor protein
- AST:
-
Aspartate aminotransferase
- CA:
-
Caffeic acid
- COMT:
-
Catechol O-methyltransferase
- CYP2C19:
-
Cytochrome P450 2C19
- CYP3A4:
-
Cytochrome P450 3A4
- ECH:
-
Echinacoside
- EGFR:
-
Epidermal growth factor receptor
- ERBB2:
-
Receptor tyrosine–protein kinase erbB-2
- ESR1:
-
Estrogen receptor
- GalN/LPS:
-
Galactosamine/lipopolysaccharides
- GSH:
-
Glutathione
- H&E:
-
Haematoxylin–eosin
- HMGB1:
-
High mobility group box 1
- HT:
-
Hydroxytyrosol
- IL-6:
-
Interleukin 6
- ISAT:
-
Isoacteoside
- MAPK1:
-
Mitogen-activated protein kinase 1
- MDA:
-
Malondialdehyde
- MMP1:
-
Interstitial collagenase
- MMP2:
-
72 KDa type IV collagenase
- MMP9:
-
Matrix metalloproteinase-9
- PhGs:
-
Phenylethanoid glycosides;
- PPI:
-
Protein–protein interaction
- PTGS2:
-
Prostaglandin G/H synthase 2
- RELA:
-
Transcription factor p65
- SOD:
-
Superoxide dismutase
- STAT1:
-
Signal transducer and activator of transcription 1-alpha/beta
- STAT3:
-
Signal transducer and activator of transcription 3
- TLR4:
-
Toll-like receptor 4
- TNF-α:
-
Tumor necrosis factor α
- TYR:
-
Tyrosinase
- 3-HPP:
-
3-Hydroxyphenylpropionic acid
References
Trefts E, Gannon M, Wasserman DH (2017) The liver. Curr Biol 27:R1147–R1151
Wang H, Naghavi M, Allen C et al (2016) Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388:1459–1544
Tian W, Xu Y (2015) Decoding liver injury: a regulatory role for histone modifications. Int J Biochem Cell Biol 67:188–193
Brenner C, Galluzzi L, Kepp O, Kroemer G (2013) Decoding cell death signals in liver inflammation. J Hepatol 59:583–594
Xu D, Xu M, Jeong S, Qian Y, Wu H, Xia Q, Kong X (2019) The role of Nrf2 in liver disease: novel molecular mechanisms and therapeutic approaches. Front Pharmacol 9:1428
Malhi H, Guicciardi ME, Gores GJ (2010) Hepatocyte death: a clear and present danger. Physiol Rev 90:1165–1194
Serfaty L, Lemoine M (2008) Definition and natural history of metabolic steatosis: clinical aspects of NAFLD, NASH and cirrhosis. Diabetes Metabol 34:634–637
Commission CP (2015) Herba cistanche. In: Commission CP (Chinese Pharmacopoeia Commission) (eds) Pharmacopoeia of the People’s Republic of China. China Medical Science Press, Beijing
Wu C-R, Lin H-C, Su M-H (2014) Reversal by aqueous extracts of Cistanche tubulosa from behavioral deficits in Alzheimer’s disease-like rat model: relevance for amyloid deposition and central neurotransmitter function. BMC Complement Altern Med 14:1–11
Nan Z-D, Zeng K-W, Shi S-P, Zhao M-B, Jiang Y, Tu P-F (2013) Phenylethanoid glycosides with anti-inflammatory activities from the stems of Cistanche deserticola cultured in Tarim desert. Fitoterapia 89:167–174
Li J, Li J, Aipire A, Gao L, Huo S, Luo J, Zhang F (2016) Phenylethanoid glycosides from Cistanche tubulosa inhibits the growth of B16-F10 cells both in vitro and in vivo by induction of apoptosis via mitochondria-dependent pathway. J Cancer 7:1877–1887
Guo Y, Cao L, Zhao Q, Zhang L, Chen J, Liu B, Zhao B (2016) Preliminary characterizations, antioxidant and hepatoprotective activity of polysaccharide from Cistanche deserticola. Int J Biol Macromol 93:678–685
Zhang B, Yang L-L, Ding S-Q, Liu J-J, Dong Y-H, Li Y-T, Li N, Zhao X-J, Hu C-L, Jiang Y, Ma X-Q (2019) Anti-osteoporotic activity of an edible traditional chinese medicine Cistanche deserticola on bone metabolism of ovariectomized rats through RANKL/RANK/TRAF6-mediated signaling pathways. Front Pharmacol 10:1412
Zhang L, Yue X-X, Zhang L, Zhao J-F, Chen Y-M, Cao Z-J, Liu Y-l (2016) Anti-osteoporosis effect of Cistanche deserticola Ma extract in ovariectomized rats. Trop J Pharm Res 15:1929–1933
Cai R-L, Yang M-H, Shi Y, Chen J, Li Y-C, Qi Y (2010) Antifatigue activity of phenylethanoid-rich extract from Cistanche deserticola. Phytother Res 24:313–315
Jiang Y, Tu P-F (2009) Analysis of chemical constituents in Cistanche species. J Chromatogr A 1216:1970–1979
Li X, Gou C, Yang H, Qiu J, Gu T, Wen T (2014) Echinacoside ameliorates d-galactosamine plus lipopolysaccharide-induced acute liver injury in mice via inhibition of apoptosis and inflammation. Scand J Gastroenterol 49:993–1000
Zhao J, Liu T, Ma L, Yan M, Zhao Y, Gu Z, Huang Y (2009) Protective effect of acteoside on immunological liver injury induced by Bacillus Calmette-Guerin plus lipopolysaccharide. Planta Med 75:1463–1469
Morikawa T, Pan Y, Ninomiya K, Imura K, Matsuda H, Yoshikawa M, Yuan D, Muraoka O (2010) Acylated phenylethanoid oligoglycosides with hepatoprotective activity from the desert plant Cistanche tubulosa. Bioorg Med Chem 18:1882–1890
Feng B, Song Y, Xu Q, Xu P, Zeng Q, Shan B, Liu K, Su D (2018) Simultaneous determination of savaside A, acteoside, and isoacteoside in rat plasma by UHPLC-MS/MS: comparative pharmacokinetic and bioavailability characteristics of Monochasma savatieri via different routes of administration. J Sep Sci 41:4408–4418
Cui Q, Pan Y, Yan X, Qu B, Liu X, Xiao W (2017) A metabolic way to investigate related hurdles causing poor bioavailability in oral delivery of isoacteoside in rats employing ultrahigh-performance liquid chromatography/quadrupole time-of-flight tandem mass spectrometry. Rapid Commun Mass Spectrom 31:371–380
Cui Q, Pan Y, Bai X, Zhang W, Chen L, Liu X (2016) Systematic characterization of the metabolites of echinacoside and acteoside from Cistanche tubulosa in rat plasma, bile, urine and feces based on UPLC-ESI-Q-TOF-MS. Biomed Chromatogr 30:1406–1415
Cui Q, Pan Y, Xu X, Zhang W, Wu X, Qu S, Liu X (2016) The metabolic profile of acteoside produced by human or rat intestinal bacteria or intestinal enzyme in vitro employed UPLC-Q-TOF-MS. Fitoterapia 109:67–74
Li Y, Zhou G, Peng Y, Tu P, Li X (2016) Screening and identification of three typical phenylethanoid glycosides metabolites from Cistanches Herba by human intestinal bacteria using UPLC/Q-TOF-MS. J Pharm Biomed Anal 118:167–176
Pirozzi C, Lama A, Simeoli R, Paciello O, Pagano TB, Mollica MP, Di Guida F, Russo R, Magliocca S, Canani RB, Raso GM, Calignano A, Meli R (2016) Hydroxytyrosol prevents metabolic impairment reducing hepatic inflammation and restoring duodenal integrity in a rat model of NAFLD. J Nutr Biochem 30:108–115
Cui Q, Pan Y, Zhang W, Zhang Y, Ren S, Wang D, Wang Z, Liu X, Xiao W (2018) Metabolites of dietary acteoside: profiles, isolation, identification, and hepatoprotective capacities. J Agric Food Chem 66:2660–2668
Zuo F (2002) Studies on pharmacokineties and the relation to intestinal baeteria of Huangqin-Tangdeeoetion. Beijing University of Chinese Medicine, Beijing
Maes M, Vinken M, Jaeschke H (2016) Experimental models of hepatotoxicity related to acute liver failure. Toxicol Appl Pharmacol 290:86–97
Li M, Wang S, Li X, Jiang L, Wang X, Kou R, Wang Q, Xu L, Zhao N, Xie K (2018) Diallyl sulfide protects against lipopolysaccharide/d-galactosamine-induced acute liver injury by inhibiting oxidative stress, inflammation and apoptosis in mice. Food Chem Toxicol 120:500–509
Galanos C, Freudenberg MA, Reutter W (1979) Galactosamine-induced sensitization to the lethal effects of endotoxin. Proc Natl Acad Sci USA 76:5939–5943
Ma X, Peng S, Zhou X, Li S, Jin P (2019) The amphioxus ERK2 gene is involved in innate immune response to LPS stimulation. Fish Shellfish Immunol 86:64–69
Taniguchi M, Takeuchi T, Nakatsuka R, Watanabe T, Sato K (2004) Molecular process in acute liver injury and regeneration induced by carbon tetrachloride. Life Sci 75:1539–1549
Li W, Liang XP, Kellendonk C, Poli V, Taub R (2002) STAT3 contributes to the mitogenic response of hepatocytes during liver regeneration. J Biol Chem 277:28411–28417
Roskoski R Jr (2012) ERK1/2 MAP kinases: Structure, function, and regulation. Pharmacol Res 66:105–143
Hu Y, Yang X, Wu S, Xiao J (2020) COX-2 in liver fibrosis. Clin Chim Acta 506:196–203
Motino O, Agra N, Brea Contreras R, Dominguez-Moreno M, Garcia-Monzon C, Vargas-Castrillon J, Carnovale CE, Bosca L, Casado M, Mayoral R, Pilar Valdecantos M, Valverde AM, Frances DE, Martin-Sanz P (2016) Cyclooxygenase-2 expression in hepatocytes attenuates non-alcoholic steatohepatitis and liver fibrosis in mice. BBA-Mol Basis Dis 1862:1710–1723
Hardesty J, Jacobs J, Warner J, Warner D, Song Y, McClain C, Kirpich I (2020) The EGFR liver regeneration pathway is downregulated in alcoholic hepatitis. The Faseb Journa 34:1–1
Bhushan B, Stoops JW, Mars WM, Orr A, Bowen WC, Paranjpe S, Michalopoulos GK (2019) TCPOBOP-induced hepatomegaly and hepatocyte proliferation are attenuated by combined disruption of MET and EGFR signaling. Hepatology 69:1702–1718
Feng M, Wang H, Wang Q, Guan W (2013) Matrix metalloprotease 9 promotes liver recovery from ischemia and reperfusion injury. J Surg Res 180:156–161
Wu Y, Li L, Wen T, Li Y-Q (2007) Protective effects of echinacoside on carbon tetrachloride-induced hepatotoxicity in rats. Toxicology 232:50–56
Xiong Q, Hase K, Tezuka Y, Namba T, Kadota S (1999) Acteoside inhibits apoptosis in d-galactosamine and lipopolysaccharide-induced liver injury. Life Sci 65:421–430
Pan S, Liu L, Pan H, Ma Y, Wang D, Kang K, Wang J, Sun B, Sun X, Jiang H (2013) Protective effects of hydroxytyrosol on liver ischemia/reperfusion injury in mice. Mol Nutr Food Res 57:1218–1227
Xia H, He Q, Wang H, Wang Y, Yang Y, Li Y, Zhang J, Chen Z, Yang J (2019) Treatment with either COX-2 inhibitor or 5-LOX inhibitor causes no compensation between COX-2 pathway and 5-LOX pathway in chronic aluminum overload-induced liver injury in rats. Fundam Clin Pharmacol 33:535–543
Acknowledgements
Our present study is supported by National Natural Science Foundation of China (NSFC Nos. 81773879, 81973465), Overseas Training Project of Liaoning Colleges and Universities (2018LNGXGJWPY-YB024), The Fourth Investigation of Traditional Chinese Medicine resources in Liaoning Province of China (2018017, 2019019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guo, Y., Cui, Q., Ren, S. et al. The hepatoprotective efficacy and biological mechanisms of three phenylethanoid glycosides from cistanches herba and their metabolites based on intestinal bacteria and network pharmacology. J Nat Med 75, 784–797 (2021). https://doi.org/10.1007/s11418-021-01508-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-021-01508-y