Abstract
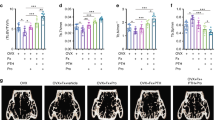
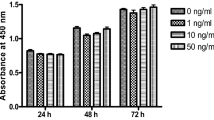
Vasoactive intestinal peptide (VIP) as a neuromodulator and neurotransmitter played a significant role in modulating bone homeostasis. Our previous study reported an essential role of VIP in in vitro BMSCs osteogenesis and in vivo bone defect repair. VIP was also revealed to have a promoting effect on embryonic skeletal element development. However, the role of VIP in fracture healing is not known yet. We hypothesized that the disorder of sympathetic nervous system impairs bone structure and fracture healing, whereas VIP may rescue the sympathetic inhibition effects and promote fracture healing. We employed a 6-hydroxydopamine (6-OHDA) induced sympathectomy mice model (sympathectomized mice), in which successful sympathetic inhibition was confirmed by a decreased level of norephedrine (NE) in the spleen. In the sympathectomized mice, the femoral micro-architecture, bone density and mechanical properties were all impaired compared to the vehicle control mice. The femoral fracture was created in the vehicle or sympathectomized mice. Vehicle mice were locally injected with PBS as a negative control, and the sympathectomized mice were treated with injection of PBS or VIP. VIP expression at the fracture site was significantly decreased in sympathectomized mice. The fracture healing was repressed upon 6-OHDA treatment and rescued by VIP treatment. Micro-CT examination showed that the femoral bone micro-architecture at the fracture sites and mechanical properties were all impaired. Simultaneously, the expression level of osteogenic markers OCN and OPN were reduced in sympathectomized mice compared with vehicle group. While the VIP treatment rescued the repression effects of 6-OHDA on bone remodeling and significantly promoted bone quality and mechanical properties as well as increased osteogenesis marker expression in the sympathectomized mice. VIP administration promoted bone fracture healing by inhibiting bone resorption, making it a putative new alternative treatment strategy for fracture healing.





Similar content being viewed by others
References
Lin W, Xu L, Zwingenberger S, Gibon E, Goodman SB, Li G (2017) Mesenchymal stem cells homing to improve bone healing. J Orthop Translat 9:19–27
Niedermair T, Straub RH, Brochhausen C, Grassel S (2020) Impact of the sensory and sympathetic nervous system on fracture healing in ovariectomized mice. Int J Mol Sci 21:405–422
Hukkanen M, Konttinen YT, Rees RG, Santavirta S, Terenghi G, Polak JM (1992) Distribution of nerve endings and sensory neuropeptides in rat synovium, meniscus and bone. Int J Tissue React 14:1–10
Jones KB, Mollano AV, Morcuende JA, Cooper RR, Saltzman CL (2004) Bone and brain: a review of neural, hormonal, and musculoskeletal connections. Iowa Orthop J 24:123–132
Elefteriou F, Campbell P, Ma Y (2014) Control of bone remodeling by the peripheral sympathetic nervous system. Calcif Tissue Int 94:140–151
Grassel SG (2014) The role of peripheral nerve fibers and their neurotransmitters in cartilage and bone physiology and pathophysiology. Arthritis Res Ther 16:485–497
Li J, Ahmad T, Spetea M, Ahmed M, Kreicbergs A (2001) Bone reinnervation after fracture: a study in the rat. J Bone Miner Res 16:1505–1510
Aitken SJ, Landao-Bassonga E, Ralston SH, Idris AI (2009) β2-Adrenoreceptor ligands regulate osteoclast differentiation in vitro by direct and indirect mechanisms. Arch Biochem Biophys 482:96–103
Huang HH, Brennan TC, Muir MM, Mason RS (2009) Functional alpha1- and beta2-adrenergic receptors in human osteoblasts. J Cell Physiol 220:267–275
Opolka A, Straub RH, Pasoldt A, Grifka J, Grassel S (2012) Substance P and norepinephrine modulate murine chondrocyte proliferation and apoptosis. Arthritis Rheum 64:729–739
Niedzwiedzki T, Filipowska J (2015) Bone remodeling in the context of cellular and systemic regulation: the role of osteocytes and the nervous system. J Mol Endocrinol 55:23–36
Hill EL, Elde R (1991) Distribution of CGRP-, VIP-, D beta H-, SP-, and NPY-immunoreactive nerves in the periosteum of the rat. Cell Tissue Res 264:469–480
Hohmann EL, Elde RP, Rysavy JA, Einzig S, Gebhard RL (1986) Innervation of periosteum and bone by sympathetic vasoactive intestinal peptide-containing nerve fibers. Science 232:868–871
Winding B, Wiltink A, Foged NT (1997) Pituitary adenylyl cyclase-activating polypeptides and vasoactive intestinal peptide inhibit bone resorption by isolated rabbit osteoclasts. Exp Physiol 82:871–886
Lundberg P, Lundgren I, Mukohyama H, Lehenkari PP, Horton MA, Lerner UH (2001) Vasoactive intestinal peptide (VIP)/pituitary adenylate cyclase-activating peptide receptor subtypes in mouse calvarial osteoblasts: presence of VIP-2 receptors and differentiation-induced expression of VIP-1 receptors. Endocrinology 142:339–347
Hohmann EL, Levine L, Tashjian AH Jr. (1983) Vasoactive intestinal peptide stimulates bone resorption via a cyclic adenosine 3’,5’-monophosphate-dependent mechanism. Endocrinology 112:1233–1239
Lerner UH, Lundberg P, Ransjo M, Persson P, Hakanson R (1994) Helodermin, helospectin, and PACAP stimulate cyclic AMP formation in intact bone, isolated osteoblasts, and osteoblastic cell lines. Calcif Tissue Int 54:284–289
Lundberg P, Lie A, Bjurholm A, Lehenkari PP, Horton MA, Lerner UH, Ransjo M (2000) Vasoactive intestinal peptide regulates osteoclast activity via specific binding sites on both osteoclasts and osteoblasts. Bone 27:803–810
Shi L, Feng L, Zhu ML, Yang ZM, Wu TY, Xu J, Liu Y, Lin WP, Lo JHT, Zhang JF, Li G (2020) Vasoactive intestinal peptide stimulates bone marrow-mesenchymal stem cells osteogenesis differentiation by activating Wnt/beta-catenin signaling pathway and promotes rat skull defect repair. Stem Cells Dev 29:655–666
Shi L, Wang C, Yan Y, Wang G, Zhang J, Feng L, Yang X, Li G (2020) Function study of vasoactive intestinal peptide on chick embryonic bone development. Neuropeptides 83:102077
Niedermair T, Kuhn V, Doranehgard F, Stange R, Wieskotter B, Beckmann J, Salmen P, Springorum HR, Straub RH, Zimmer A, Grifka J, Grassel S (2014) Absence of substance P and the sympathetic nervous system impact on bone structure and chondrocyte differentiation in an adult model of endochondral ossification. Matrix Biol 38:22–35
Wirth T, Westendorf AM, Bloemker D, Wildmann J, Engler H, Mollerus S, Wadwa M, Schafer MK, Schedlowski M, del Rey A (2014) The sympathetic nervous system modulates CD4(+)Foxp3(+) regulatory T cells via noradrenaline-dependent apoptosis in a murine model of lymphoproliferative disease. Brain Behav Immun 38:100–110
Szpunar MJ, Belcher EK, Dawes RP, Madden KS (2016) Sympathetic innervation, norepinephrine content, and norepinephrine turnover in orthotopic and spontaneous models of breast cancer. Brain Behav Immun 53:223–233
Shi L, Feng L, Liu Y, Duan JQ, Lin WP, Zhang JF, Li G (2018) MicroRNA-218 promotes osteogenic differentiation of mesenchymal stem cells and accelerates bone fracture healing. Calcif Tissue Int 103:227–236
Haffner-Luntzer M, Kemmler J, Heidler V, Prystaz K, Schinke T, Amling M, Kovtun A, Rapp AE, Ignatius A, Liedert A (2016) Inhibition of midkine augments osteoporotic fracture healing. PLoS ONE 11:e0159278
Sun Y, Xu J, Xu L, Zhang J, Chan K, Pan X, Li G (2017) MiR-503 promotes bone formation in distraction osteogenesis through suppressing Smurf1 expression. Sci Rep 7:409–418
Chen Y, Lin S, Sun Y, Pan X, Xiao L, Zou L, Ho KW, Li G (2016) Translational potential of ginsenoside Rb1 in managing progression of osteoarthritis. J Orthop Translat 6:27–33
Qiao Y, Wang Y, Zhou Y, Jiang F, Huang T, Chen L, Lan J, Yang C, Guo Y, Yan S, Wei Z, Li J (2019) The role of nervous system in adaptive response of bone to mechanical loading. J Cell Physiol 234:7771–7780
Cutz E, Chan W, Track NS, Goth A, Said SI (1978) Release of vasoactive intestinal polypeptide in mast cells by histamine liberators. Nature 275:661–662
Martinez C, Delgado M, Abad C, Gomariz RP, Ganea D, Leceta J (1999) Regulation of VIP production and secretion by murine lymphocytes. J Neuroimmunol 93:126–138
Takeda S, Elefteriou F, Levasseur R, Liu X, Zhao L, Parker KL, Armstrong D, Ducy P, Karsenty G (2002) Leptin regulates bone formation via the sympathetic nervous system. Cell 111:305–317
Elefteriou F, Ahn JD, Takeda S, Starbuck M, Yang X, Liu X, Kondo H, Richards WG, Bannon TW, Noda M, Clement K, Vaisse C, Karsenty G (2005) Leptin regulation of bone resorption by the sympathetic nervous system and CART. Nature 434:514–520
Lambert E, Phillips S, Tursunalieva A, Eikelis N, Sari C, Dixon J, Straznicky N, Grima M, Schlaich M, Lambert G (2018) Inverse association between sympathetic nervous system activity and bone mass in middle aged overweight individuals. Bone 111:123–128
Cherruau M, Facchinetti P, Baroukh B, Saffar JL (1999) Chemical sympathectomy impairs bone resorption in rats: a role for the sympathetic system on bone metabolism. Bone 25:545–551
Pagani F, Sibilia V, Cavani F, Ferretti M, Bertoni L, Palumbo C, Lattuada N, De Luca E, Rubinacci A, Guidobono F (2008) Sympathectomy alters bone architecture in adult growing rats. J Cell Biochem 104:2155–2164
Hu K, Zhou H, Zhang G, Qin R, Hou R, Kong L, Ding Y (2010) The effect of chemical sympathectomy and stress on bone remodeling in adult rats. Neuro Endocrinol Lett 31:807–813
Togari A (2002) Adrenergic regulation of bone metabolism: possible involvement of sympathetic innervation of osteoblastic and osteoclastic cells. Microsc Res Tech 58:77–84
Suzuki A, Palmer G, Bonjour JP, Caverzasio J (1999) Regulation of alkaline phosphatase activity by p38 MAP kinase in response to activation of Gi protein-coupled receptors by epinephrine in osteoblast-like cells. Endocrinology 140:3177–3182
Takeuchi T, Tsuboi T, Arai M, Togari A (2001) Adrenergic stimulation of osteoclastogenesis mediated by expression of osteoclast differentiation factor in MC3T3-E1 osteoblast-like cells. Biochem Pharmacol 61:579–586
Arai M, Nagasawa T, Koshihara Y, Yamamoto S, Togari A (2003) Effects of beta-adrenergic agonists on bone-resorbing activity in human osteoclast-like cells. Biochim Biophys Acta 1640:137–142
Harada S, Rodan GA (2003) Control of osteoblast function and regulation of bone mass. Nature 423:349–355
Gaur T, Lengner CJ, Hovhannisyan H, Bhat RA, Bodine PV, Komm BS, Javed A, van Wijnen AJ, Stein JL, Stein GS, Lian JB (2005) Canonical WNT signaling promotes osteogenesis by directly stimulating Runx2 gene expression. J Biol Chem 280:33132–33140
Roca H, Franceschi RT (2008) Analysis of transcription factor interactions in osteoblasts using competitive chromatin immunoprecipitation. Nucleic Acids Res 36:1723–1730
Gu XC, Zhang XB, Hu B, Zi Y, Li M (2016) Neuropeptide Y accelerates post-fracture bone healing by promoting osteogenesis of mesenchymal stem cells. Neuropeptides 60:61–66
Amano S, Arai M, Goto S, Togari A (2007) Inhibitory effect of NPY on isoprenaline-induced osteoclastogenesis in mouse bone marrow cells. Biochim Biophys Acta 1770:966–973
Ding WG, Zhang ZM, Zhang YH, Jiang SD, Jiang LS, Dai LY (2010) Changes of substance P during fracture healing in ovariectomized mice. Regul Pept 159:28–34
Appelt J, Baranowsky A, Jahn D, Yorgan T, Kohli P, Otto E, Farahani SK, Graef F, Fuchs M, Herrera A, Amling M, Schinke T, Frosch KH, Duda GN, Tsitsilonis S, Keller J (2020) The neuropeptide calcitonin gene-related peptide alpha is essential for bone healing. EBioMedicine 59:102970
Acknowledgements
This work was partially supported by grants from the National Natural Science Foundation of China (Grant No. 81772322); Hong Kong Government Research Grant Council, General Research Fund (Grant Nos. 14120118, 14160917, C7030-18G and T13-402/17-N); Hong Kong Innovation Technology Commission Funds (Grant No. PRP/050/19FX); Hong Kong Medical Research Funds (Grant Nos. 16170951 and 17180831). This study also received support from the SMART program, Lui Che Woo Institute of Innovative Medicine, The Chinese University of Hong Kong.
Author information
Authors and Affiliations
Contributions
GL and LF led and supervised all the experiments. GL, JFZ, and LF designed experiments. LS, YL, ZMY, TYW, JX, JJZ and WPL conducted experiments. LS and LF analyzed data. LS, HTL, LF, and GL prepared, wrote and revised the manuscript. All authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
There is no conflict of interest.
Human and Animal Rights and Informed Consent
No human specimen were applied in this study. The animal experiments were performed in accordance with the Code of Ethics of the World Medical Association. The ethics approval (14/118/MIS) of this study was obtained from the Animal Research Ethics Committee (AEEC) of The Chinese University of Hong Kong.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shi, L., Liu, Y., Yang, Z. et al. Vasoactive Intestinal Peptide Promotes Fracture Healing in Sympathectomized Mice. Calcif Tissue Int 109, 55–65 (2021). https://doi.org/10.1007/s00223-021-00820-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-021-00820-9