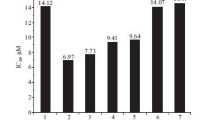
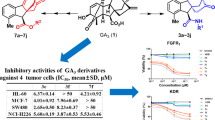
Abstract
The natural product Ginkgolide B was used as a raw material and modified by esterification on C10-OH or C1-OH to obtain 11 derivatives (1–11), which were structurally characterized with nuclear magnetic resonance spectroscopy. An MTT assay-based in vitro tumor proliferation inhibitory activity test showed that compounds 2, 3, 6, 7, 10, and 11 exhibited strong inhibitory activity against the human ovarian cancer cells SKOV3, with IC50 values of 16.05 µmol/L, 15.65 µmol/L, 32.00 µmol/L, 63.30 µmol/L, 23.20 µmol/L, and 31.10 µmol/L, respectively. Annexin V/PI double staining assay showed that compound 2 induced apoptosis in SKOV3 cells to a slightly greater extent than GB and compounds 5 and 9, with an apoptosis rate of 31.68%.



Similar content being viewed by others
References
Ahlemeyer B, Krieglstein J. Pharmacological studies supporting the therapeutic use of Ginkgo biloba extract for Alzheimer’s disease. Pharmacopsychiatry. 2003;36:8–14. https://doi.org/10.1055/s-2003-40454.
Bate C, Salmona M, Williams A. Ginkgolide B inhibits the neurotoxicity of prions or amyloid-beta1-42. J Neuroinflamm. 2004;1:4 https://doi.org/10.1186/1742-2094-1-4.
Tian QY, Gong LL. Research progress of ginkgolide. Zhongnan Pharm. 2016;14:838–41. CNKI:SUN:ZNYX.0.2016-08-016.
Nabavi SM, Habtemariam S, Daglia M, Braidy N, Loizzo MR, Tundis R. et al. Neuroprotective effects of ginkgolide B against ischemic stroke: A review of current literature. Curr Top Med Chem. 2015;15:2222–32. https://doi.org/10.2174/1568026615666150610142647.
Pan M, Yuan Y, Hui A-L. et al. Anti-platelet aggregation activity of ginkgolide B precursor drugs. Chin J Pharm. 2012;28:1435–38. https://doi.org/10.3969/j.issn.1001-1978.2012.10.024.
Huaian W, Jinsheng G. Research progress of ginkgolide B for the treatment of malignant tumors. Guangming TCM. 2017;32:2589–90. https://doi.org/10.3969/j.issn.1003-8914.2017.17.063.
Gao T, Zhao R, Yao L. et al. Platelet-activating factor induces the stemness of ovarian cancer cells via the PAF/PAFR signaling pathway[J]. Am J Transl Res. 2020;12:7249–61. PMID: 33312364; PMCID: PMC7724322.
Jiang W, Cong Q, Wang Y, Ye B, Xu C. Ginkgo may sensitize ovarian cancer cells to cisplatin: antiproliferative and apoptosis-inducing effects of ginkgolide B on ovarian cancer cells. Integr Cancer Ther. 2014;13:NP10–7. https://doi.org/10.1177/1534735411433833.
Chan W-H. The signaling cascades of ginkgolide B-induced apoptosis in MCF-7 breast cancer cells. Int J Mol Sci. 2007;8:1177–95. https://doi.org/10.3390/i8111177.
Yang B, Shuan S, Shengqiong C. et al. Study of ginkgolide B induced apoptosis in human colorectal cancer HCT116 cells. J Chengdu Med Coll. 2015;10:281–86. https://doi.org/10.3969/j.issn.1674-2257.2015.03.006.
Zhi Y, Pan J, Shen W, He P, Zheng J, Zhou X. et al. Ginkgolide B inhibits human bladder cancer cell migration and invasion through MicroRNA-223-3p. Cell Physiol Biochem. 2016;39:1787–94. https://doi.org/10.1159/000447878.
Chan WH, Hsuuw YD. Dosage effects of ginkgolide B on ethanol-induced cell death in human hepatoma G2 cells. Ann NY Acad Sci. 2007;1095:388–98. https://doi.org/10.1196/annals.1397.042.
Lou C, Lu H, Ma Z, Liu C, Zhang Y. Ginkgolide B enhances gemcitabine sensitivity in pancreatic cancer cell lines via inhibiting PAFR/NF-кB pathway. Biomed Pharmacother. 2019;109:563–72. https://doi.org/10.1016/j.biopha.2018.10.084.
Yao B, Liu B, Shi L, Li X, Ren C, Cai M. et al. PAFR selectively mediates radioresistance and irradiation-induced autophagy suppression in prostate cancer cells. Oncotarget. 2017;8:13846–54. https://doi.org/10.18632/oncotarget.14647.
Coyne CP, Narayanan L. Carnosic Acid, Tangeretin, and ginkgolide-B anti-neoplastic cytotoxicity in dual combination with dexamethasone-[anti-EGFR] in pulmonary adenocarcinoma (A549). Anti Cancer Agents Med Chem. 2019;19:802–19. https://doi.org/10.2174/1871520619666181204100226.
Sahu RP, Turner MJ, DaSilva SC, Rashid BM, Ocana JA, Perkins SM. et al. The environmental stressor ultraviolet B radiation inhibits murine antitumor immunity through its ability to generate platelet-activating factor agonists. Carcinogenesis. 2012;33:1360–67. https://doi.org/10.1093/carcin/bgs152.
Zhang L, Wang D, Jiang W. et al. Activated networking of platelet activating factor receptor and FAK/STAT1 induces malignant potential in BRCA1-mutant at-risk ovarian epithelium. BioMed Cent. 2010;8:74 https://doi.org/10.1186/1477-7827-8-74.
Aponte M, Jiang W, Lakkis M, Li MJ, Edwards D, Albitar L. et al. Activation of platelet-activating factor receptor and pleiotropic effects on tyrosine phospho-EGFR/Src/FAK/paxillin in ovarian cancers. Cancer Res. 2008;68:5839–48. https://doi.org/10.1158/0008-5472.CAN-07-5771.
Yu Y, Zhang X, Hong S, Zhang M, Cai Q, Zhang M. et al. The expression of platelet-activating factor receptor modulates the cisplatin sensitivity of ovarian cancer cells: a novel target for combination therapy. Br J Cancer. 2014;111:515–24. https://doi.org/10.1038/bjc.2014.323.
Chen Q, Chen L, Sun JB. Research progress on the sources, structural modifications and new dosage forms of ginkgolide compounds. J Nanjing. 2019 University of Traditional Chinese Medicine;35:344–50 https://doi.org/10.14148/j.issn.1672-0482.2019.0344.
Hui A-L, Wu Z-Y, Yuan Y, Zhou A, Pan M. Synthesis of ginkgolide derivatives and analogs and progress in the study of PAFR and GlyR antagonistic activities. J Org Chem. 2013;33:1263–72. https://doi.org/10.6023/cjoc201209028.
Liu KZ, Xue LP, Xu YF. et al. Review and comparative analysis of benoxate synthesis processes. Henan Sci. 2018;36:1362–66. https://doi.org/10.3969/j.issn.1004-3918.2018.09.007.
Highest L, Jia L, Zhang Yin Y. et al. Optimization of experimental reaction conditions for the synthesis of benoxate. J Xiangnan Coll. 2016;37:18–21. https://doi.org/10.3969/j.issn.1672-8173.2016.05.005.
Wang Y. Synthesis and characterization of benzoate. Exp Sci Technol. 2014;12:32–4. https://doi.org/10.3969/j.issn.1672-4550.2014.04.013.
Dai GX, Wang L, Shoutao C. Study on the catalytic synthesis process of benzoate. Coal Chem Ind. 2014;37:79–81. CNKI:SUN:HHGZ.0.2014-08-028.
Xiaozhong F, Zero L, Guangling R. et al. Improvement of the synthesis process of benoxate. J Guiyang Med Coll. 2012;37:440–1 + 443. https://doi.org/10.3969/j.issn.1000-2707.2012.04.035.
Teng SB, Qian J. Progress in the synthesis of benoxylates. Shandong Chem Ind. 2009;38:23–5. https://doi.org/10.3969/j.issn.1008-021X.2009.08.008.
Feng ZIL, Jiayuan X, Zhu ZB. et al. Synthesis of ginkgolide B structural modifiers and their effects on 6-OHDA-induced SH-SY5Y cell viability. Chin. J Med Chem. 2020;30:199–203. https://doi.org/10.14142/j.cnki.cn21-1313/r.2020.04.002.
Yong-Ming L, Jian P, Zhang Wen-NA. et al. Synthesis, in silico and in vivo blood brain barrier permeability of ginkgolide B cinnamate. Fitoterapia. 2015;106:110–14. https://doi.org/10.1016/j.fitote.2015.08.012.
Wu ZY, Pan J, Yuan Y, Hui A, Yang Y, Zhou A. et al. Brain-targeting research of 10-O-nicotinate ginkgolide B: a new prodrug of ginkgolide B. Med Chem Res. 2012;21:4028–36. https://doi.org/10.1007/s00044-011-9947-z.
Sladowski D, Steer SJ, Clothier RH, Balls M. An improved MTT assay. J Immunol Methods. 1993;157:203–07. https://doi.org/10.1016/0022-1759(93)90088-o.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (31702206), Key projects of science and Technology Department of Shaanxi Province (2018ZDXM-SF-083), and Natural Science Foundation of Shaanxi Province of China (2019JQ-530).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Feng, Z., Zhu, Z., Chen, W. et al. Ginkgolide B derivative synthesis and their effects on the viability of SKOV3 cells. Med Chem Res 30, 1265–1272 (2021). https://doi.org/10.1007/s00044-021-02732-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-021-02732-7