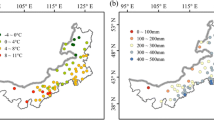
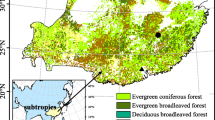
Abstract
Vegetation phenology is an indicator of vegetation response to natural environmental changes and is of great significance for the study of global climate change and its impact on terrestrial ecosystems. The normalized difference vegetation index (NDVI) and enhanced vegetation index (EVI), extracted from the Moderate Resolution Imaging Spectrometer (MODIS), are widely used to monitor phenology by calculating land surface reflectance. However, the applicability of the vegetation index based on ‘greenness’ to monitor photosynthetic activity is hindered by poor observation conditions (e.g., ground shadows, snow, and clouds). Recently, satellite measurements of solar-induced chlorophyll fluorescence (SIF) from OCO-2 sensors have shown great potential for studying vegetation phenology. Here, we tested the feasibility of SIF in extracting phenological metrics in permafrost regions of the northeastern China, exploring the characteristics of SIF in the study of vegetation phenology and the differences between NDVI and EVI. The results show that NDVI has obvious SOS advance and EOS lag, and EVI is closer to SIF. The growing season length based on SIF is often the shortest, while it can represent the true phenology of vegetation because it is closely related to photosynthesis. SIF is more sensitive than the traditional remote sensing indices in monitoring seasonal changes in vegetation phenology and can compensate for the shortcomings of traditional vegetation indices. We also used the time series data of MODIS NDVI and EVI to extract phenological metrics in different permafrost regions. The results show that the length of growing season of vegetation in predominantly continuous permafrost (zone I) is longer than in permafrost with isolated taliks (zone II). Our results have certain significance for understanding the response of ecosystems in cold regions to global climate change.
Similar content being viewed by others
References
Baker N R, 2008. Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annual Review of Plant Biology, 59(1): 89–113. doi: https://doi.org/10.1146/annurev.arplant.59.032607.092759
Boyd D S, Almond S, Dash J et al., 2011. Phenology of vegetation in Southern England from Envisat MERIS terrestrial chlorophyll index (MTCI) data. International Journal of Remote Sensing, 32(23): 8421–8447. doi: https://doi.org/10.1080/01431161.2010.542194
Bradley A V, Gerard F F, Barbier N et al., 2011. Relationships between phenology, radiation and precipitation in the Amazon region. Global Change Biology, 17(6): 2245–2260. doi: https://doi.org/10.1111/j.1365-2486.2011.02405.x
Chang Q, Xiao X M, Jiao W Z et al., 2019. Assessing consistency of spring phenology of snow-covered forests as estimated by vegetation indices, gross primary production, and solar- induced chlorophyll fluorescence. Agricultural and Forest Meteorology, 275: 305–316. doi: https://doi.org/10.1016/j.agrformet.2019.06.002
Cleland E E, Chuine I, Menzel A et al., 2007. Shifting plant phenology in response to global change. Trends in Ecology & Evolution, 22(7): 357–365. doi: https://doi.org/10.1016/j.tree.2007.04.003
Delbart N, Le Toan T, Kergoat L et al., 2006. Remote sensing of spring phenology in boreal regions: a free of snow-effect method using NOAA-AVHRR and SPOT-VGT data (1982–2004). Remote Sensing of Environment, 101(1): 52–62. doi: https://doi.org/10.1016/j.rse.2005.11.012
Deng G R, Zhang H Y, Guo X Y et al., 2019. Asymmetric effects of daytime and nighttime warming on boreal forest spring phenology. Remote Sensing, 11(14): 1651. doi: https://doi.org/10.3390/rs11141651
Frankenberg C, Butz A, Toon G C, 2011. Disentangling chlorophyll fluorescence from atmospheric scattering effects in O2 Aband spectra of reflected sun-light. Geophysical Research Letters, 38(3): L03801. doi: https://doi.org/10.1029/2010GL045896
Frankenberg C, O’Dell C, Berry J et al., 2014. Prospects for chlorophyll fluorescence remote sensing from the Orbiting Carbon Observatory-2. Remote Sensing of Environment, 147: 1–12. doi: https://doi.org/10.1016/j.rse.2014.02.007
Fu Y H, Piao S L, de Beeck M O et al., 2014. Recent spring phenology shifts in western Central Europe based on multiscale observations. Global Ecology & Biogeography, 23(11): 1255–1263. doi: https://doi.org/10.1111/geb.12210
Fu Y Y, He H S, Zhao J J et al., 2018. Climate and spring phenology effects on autumn phenology in the Greater Khingan Mountains, Northeastern China. Remote Sensing, 10(3): 449. doi: https://doi.org/10.3390/rs10030449
Gocic M, Trajkovic S, 2013. Analysis of changes in meteorological variables using Mann-Kendall and Sen’s slope estimator statistical tests in Serbia. Global and Planetary Change, 100: 172–182. doi: https://doi.org/10.1016/j.gloplacha.2012.10.014
Gonsamo A, Chen J M, Price D T et al., 2012. Land surface phenology from optical satellite measurement and CO2 eddy covariance technique. Journal of Geophysical Research: Biogeosciences, 117: G03032. doi: https://doi.org/10.1029/2012JG002070
Goward S N, Markham B, Dye D G et al., 1991. Normalized difference vegetation index measurements from the advanced very high resolution radiometer. Remote Sensing of Environment, 35(2v3): 257–277. doi: https://doi.org/10.1016/0034-4257(91)90017-Z
Guanter L, Frankenberg C, Dudhia A et al., 2012. Retrieval and global assessment of terrestrial chlorophyll fluorescence from GOSAT space measurements. Remote Sensing of Environment, 121: 236–251. doi: https://doi.org/10.1016/j.rse.2012.02.006
Guanter L, Zhang Y G, Jung M et al., 2014. Global and time-resolved monitoring of crop photosynthesis with chlorophyll fluorescence. Proceedings of the National Academy of Sciences of the United States of America, 111(14): E1327–E1333. doi: https://doi.org/10.1073/pnas.1320008111
Guo M, Li J, Huang S B et al., 2020. Feasibility of using MODIS products to simulate sun-induced chlorophyll fluorescence (SIF) in boreal forests. Remote Sensing, 12(4): 680. doi: https://doi.org/10.3390/rs12040680
Hmimina G, Dufrêne E, Pontailler J Y et al., 2013. Evaluation of the potential of MODIS satellite data to predict vegetation phenology in different biomes: an investigation using ground-based NDVI measurements. Remote Sensing of Environment, 132: 145–158. doi: https://doi.org/10.1016/j.rse.2013.01.010
Huete A, Didan K, Miura T et al., 2002. Overview of the radiometric and biophysical performance of the MODIS vegetation indices. Remote Sensing of Environment, 83(1–2): 195–213. doi: https://doi.org/10.1016/S0034-4257(02)00096-2
Irwin J O, 1934. Correlation methods in psychology. British Journal of Psychology, 25(1): 86–91. doi: https://doi.org/10.1111/j.2044-8295.1934.tb00727.x
Jeong S J, Schimel D, Frankenberg C et al., 2017. Application of satellite solar-induced chlorophyll fluorescence to understanding large-scale variations in vegetation phenology and function over northern high latitude forests. Remote Sensing of Environment, 190: 178–187. doi: https://doi.org/10.1016/j.rse.2016.11.021
Joiner J, Yoshida Y, Vasilkov A P et al., 2011. First observations of global and seasonal terrestrial chlorophyll fluorescence from space. Biogeosciences, 8(3): 637–651. doi: https://doi.org/10.5194/bg-8-637-2011
Joiner J, Yoshida Y, Vasilkov A P et al., 2014. The seasonal cycle of satellite chlorophyll fluorescence observations and its relationship to vegetation phenology and ecosystem atmosphere carbon exchange. Remote Sensing of Environment, 152: 375–391. doi: https://doi.org/10.1016/j.rse.2014.06.022
Jonsson P, Eklundh L, 2002. Seasonality extraction by function fitting to time-series of satellite sensor data. IEEE Transactions on Geoscience and Remote Sensing, 40(8): 1824–1832. doi: https://doi.org/10.1109/TGRS.2002.802519
Jönsson P, Eklundh L, 2004. TIMESAT—a program for analyzing time-series of satellite sensor data. Computers & Geosciences, 30(8): 833–845. doi: https://doi.org/10.1016/j.cageo.2004.05.006
Justice C O, Townshend J R G, Holben B N et al., 1985. Analysis of the phenology of global vegetation using meteorological satellite data. International Journal of Remote Sensing, 6(8): 1271–1318. doi: https://doi.org/10.1080/01431168508948281
Köhler P, Guanter L, Kobayashi H et al., 2018. Assessing the potential of sun-induced fluorescence and the canopy scattering coefficient to track large-scale vegetation dynamics in Amazon forests. Remote Sensing of Environment, 204: 769–785. doi: https://doi.org/10.1016/j.rse.2017.09.025
Lee J E, Frankenberg C, Van der Tol C et al., 2013. Forest productivity and water stress in Amazonia: observations from GOSAT chlorophyll fluorescence. Proceedings of the Royal Society B: Biological Sciences, 280(1761): 20130171. doi: https://doi.org/10.1098/rspb.2013.0171
Li X, Xiao J F, He B B, 2018. Chlorophyll fluorescence observed by OCO-2 is strongly related to gross primary productivity estimated from flux towers in temperate forests. Remote Sensing of Environment, 204: 659–671. doi: https://doi.org/10.1016/j.rse.2017.09.034
Li X, Xiao J F, 2019. A global, 005-degree product of solar-induced chlorophyll fluorescence derived from OCO-2, MODIS, and Reanalysis Data. Remote Sensing, 11(5): 517. doi: https://doi.org/10.3390/rs11050517
Liu Q, Fu Y H, Zeng Z Z et al., 2016. Temperature, precipitation, and insolation effects on autumn vegetation phenology in temperate China. Global Change Biology, 22(2): 644–655. doi: https://doi.org/10.1111/gcb.13081
Liu Xiaotian, Zhou Lei, Shi Hao et al., 2018. Phenological characteristics of temperate coniferous and broad-leaved mixed forests based on multiple remote sensing vegetation indices, chlorophyll fluorescence and CO2 flux data. Acta Ecologica Sinica, 38(10): 3482–3494. (in Chinese)
Lloyd D, 1990. A phenological classification of terrestrial vegetation cover using shortwave vegetation index imagery. International Journal of Remote Sensing, 11(12): 2269–2279. doi: https://doi.org/10.1080/01431169008955174
Luo Y W, Su B, Currie W S et al., 2004. Progressive nitrogen limitation of ecosystem responses to rising atmospheric carbon dioxide. BioScience, 54(8): 731–739. doi: https://doi.org/10.1641/0006-3568(2004)054[0731:Pnloer]2.0.Co;2
Mao Dehuang, Wang Zongming, Luo Ling et al., 2012. Dynamic changes of vegetation net primary productivity in permafrost zone of Northeast China in 1982–2009 in response to global change. Chinese Journal of Applied Ecology, 23(26): 1511–1519. (in Chinese)
Mann H B, 1945. Nonparametric tests against trend. Econometrica, 13(3): 245–259. doi: https://doi.org/10.2307/1907187
Meroni M, Rossini M, Guanter L et al., 2009. Remote sensing of solar-induced chlorophyll fluorescence: review of methods and applications. Remote Sensing of Environment, 113(10): 2037–2051. doi: https://doi.org/10.1016/j.rse.2009.05.003
Mi Desheng, 1990. Map of snow, ice and frozen ground in China. Journal of Glaciology and Geocryology, 12(4): 175–181. (in Chinese)
Myneni R B, Keeling C D, Tucker C J et al., 1997. Increased plant growth in the northern high latitudes from 1981 to 1991. Nature, 386(6626): 698–702. doi: https://doi.org/10.1038/386698a0
Piao S L, Friedlingstein P, Ciais P et al., 2007. Growing season extension and its impact on terrestrial carbon cycle in the Northern Hemisphere over the past 2 decades. Global Biogeochemical Cycles, 21(3): GB3018. doi: https://doi.org/10.1029/2006gb002888
Qiu B, Li W K, Wang X Q et al., 2019. Satellite-observed solar-induced chlorophyll fluorescence reveals higher sensitivity of alpine ecosystems to snow cover on the Tibetan Plateau. Agricultural and Forest Meteorology, 271: 126–134. doi: https://doi.org/10.1016/j.agrformet.2019.02.045
Richardson A D, Anderson R S, Arain M A et al., 2012. Terrestrial biosphere models need better representation of vegetation phenology: results from the North American Carbon Program Site Synthesis. Global Change Biology, 18(2): 566–584. doi: https://doi.org/10.1111/j.1365-2486.2011.02562.x
Richardson A D, Keenan T F, Migliavacca M et al., 2013. Climate change, phenology, and phenological control of vegetation feedbacks to the climate system. Agricultural and Forest Meteorology, 169: 156–173. doi: https://doi.org/10.1016/j.agrformet.2012.09.012
Rocha A V, Shaver G R, 2009. Advantages of a two band EVI calculated from solar and photosynthetically active radiation fluxes. Agricultural and Forest Meteorology, 149(9): 1560–1563. doi: https://doi.org/10.1016/j.agrformet.2009.03.016
Savitzky A, Golay M J E, 1964. Smoothing and differentiation of data by simplified least squares procedures. Analytical Chemistry, 36(8): 1627–1639. doi: https://doi.org/10.1021/ac60214a047
Sen P K, 1968. Estimates of the regression coefficient based on Kendall’s Tau. Journal of the American Statistical Association, 63(324): 1379–1389. doi: https://doi.org/10.1080/01621459.1968.10480934
Shen M G, Sun Z Z, Wang S P et al., 2013. No evidence of continuously advanced green-up dates in the Tibetan Plateau over the last decade. Proceedings of the National Academy of Sciences of the United States of America, 110(26): E2329. doi: https://doi.org/10.1073/pnas.1304625110
Shen M G, Tang Y H, Desai A R et al., 2014. Can EVI-derived land-surface phenology be used as a surrogate for phenology of canopy photosynthesis? International Journal of Remote Sensing, 35(3): 1162–1174. doi: https://doi.org/10.1080/01431161.2013.875636
Stanimirova R, Cai Z Z, Melaas E K et al., 2019. An empirical assessment of the MODIS land cover dynamics and TIMESAT land surface phenology algorithms. Remote Sensing, 11(19): 2201. doi: https://doi.org/10.3390/rs11192201
Tabari H, Somee B S, Zadeh M R, 2011. Testing for long-term trends in climatic variables in Iran. Atmospheric Research, 100(1): 132–140. doi: https://doi.org/10.1016/j.atmosres.2011.01.005
Tang H, Li Z W, Zhu Z L et al., 2015. Variability and climate change trend in vegetation phenology of recent decades in the Greater Khingan Mountain area, Northeastern China. Remote Sensing, 7(9): 11914–11932. doi: https://doi.org/10.3390/rs70911914
Tang J W, Körner C, Muraoka H et al., 2016. Emerging opportunities and challenges in phenology: a review. Ecosphere, 7(8): e01436. doi: https://doi.org/10.1002/ecs2.1436
Tucker C J, Pinzon J E, Brown M E et al., 2005. An extended AVHRR 8-km NDVI dataset compatible with MODIS and SPOT vegetation NDVI data. International Journal of Remote Sensing, 26(20): 4485–4498. doi: https://doi.org/10.1080/01431160500168686
Walther S, Voigt M, Thum T et al., 2016. Satellite chlorophyll fluorescence measurements reveal large-scale decoupling of photosynthesis and greenness dynamics in boreal evergreen forests. Global Change Biology, 22(9): 2979–2996. doi: https://doi.org/10.1111/gcb.13200
Wang C, Beringer J, Hutley L B et al., 2019. Phenology dynamics of dryland ecosystems along the North Australian tropical transect revealed by satellite solar-induced chlorophyll fluorescence. Geophysical Research Letters, 46(10): 5294–5302. doi: https://doi.org/10.1029/2019GL082716
Wang X, Dannenberg M P, Yan D et al., 2020. Globally consistent patterns of asynchrony in vegetation phenology derived from optical, microwave, and fluorescence satellite data. Journal of Geophysical Research: Biogeosciences, 125(7): e2020JG005732. doi: https://doi.org/10.1029/2020JG005732
Wei Z, Jin H J, Zhang J M et al., 2011. Prediction of permafrost changes in Northeastern China under a changing climate. Science China Earth Sciences, 54(6): 924–935. doi: https://doi.org/10.1007/s11430-010-4109-6
Wu C Y, Chen J M, Black T A et al., 2013. Interannual variability of net ecosystem productivity in forests is explained by carbon flux phenology in autumn. Global Ecology and Biogeography, 22(8): 994–1006. doi: https://doi.org/10.1111/geb.12044
Wu C Y, Gonsamo A, Gough C M et al., 2014. Modeling growing season phenology in North American forests using seasonal mean vegetation indices from MODIS. Remote Sensing of Environment, 147: 79–88. doi: https://doi.org/10.1016/j.rse.2014.03.001
Yu L, Wen J, Chang C Y et al., 2019. High-Resolution Global Contiguous SIF of OCO-2. Geophysical Research Letters, 46(3): 1449–1458. doi: https://doi.org/10.1029/2018GL081109
Yu L X, Liu T X, Bu K et al., 2017. Monitoring the long term vegetation phenology change in Northeast China from 1982 to 2015. Scientific Reports, 7(1): 14770. doi: https://doi.org/10.1038/s41598-017-14918-4
Yu Xinfang, Zhuang Dafang, 2006. Monitoring forest pheno-phases of Northeast China based on MODIS NDVI data. Resources Science, 28(4): 111–117. (in Chinese)
Zarco-Tejada P J, Morales A, Testi L et al., 2013. Spatio-temporal patterns of chlorophyll fluorescence and physiological and structural indices acquired from hyperspectral imagery as compared with carbon fluxes measured with eddy covariance. Remote Sensing of Environment, 133: 102–115. doi: https://doi.org/10.1016/j.rse.2013.02.003
Zhang X Y, Friedl M A, Schaaf C B, 2006. Global vegetation phenology from Moderate Resolution Imaging Spectroradiometer (MODIS): evaluation of global patterns and comparison with in situ measurements. Journal of Geophysical Research: Biogeoences, 111: G04017. doi: https://doi.org/10.1029/2006JG000217
Zhang Y, Joiner J, Alemohammad S H et al., 2018. A global spatially contiguous solar-induced fluorescence (CSIF) dataset using neural networks. Biogeosciences, 15(19): 5779–5800. doi: https://doi.org/10.5194/bg-15-5779-2018
Zhao J J, Wang Y Y, Zhang Z X et al., 2016. The variations of land surface phenology in Northeast China and its responses to climate change from 1982 to 2013. Remote Sensing, 8(5): 400. doi: https://doi.org/10.3390/rs8050400
Zhou Yuke, 2020. Analysis of controlling factors for vegetation productivity in Northeast China. Acta Geographica Sinica, 75(1): 53–67. (in Chinese)
Zhou Youwu, Guo Dongxin, 1982. Principal characteristics of permafrost in China. Journal of Glaciology and Geocryology, 4(1): 1–19. (in Chinese)
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item
Under the auspices of National Key Research and Development Projects (No. 2018YFE0207800), National Natural Science Foundation of China (No. 41871103)
Rights and permissions
About this article
Cite this article
Wen, L., Guo, M., Yin, S. et al. Vegetation Phenology in Permafrost Regions of Northeastern China Based on MODIS and Solar-induced Chlorophyll Fluorescence. Chin. Geogr. Sci. 31, 459–473 (2021). https://doi.org/10.1007/s11769-021-1204-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11769-021-1204-x