Abstract
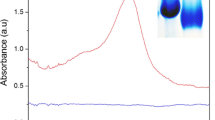
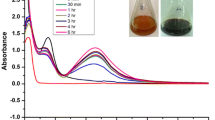
Spherical shaped silver nanoparticles with the average size of 68.7 nm were synthesized from eco-friendly and environmentally safe approach using serum carrier protein 'fetuin-A' as stabilizing agent (named as F-AgNPs). Protein capping provided excellent stability at variable temperature (~ 25–85 °C), pH (~ 4–8), salt concentrations (~ 1.5 M) and storage period (≥ 3 months). Synthesized F-AgNPs were extensively characterized using UV–visible, Fourier transform infrared, circular dichroism and intrinsic fluorescence spectroscopy, dynamic light scattering, atomic force and scanning electron microscopy, energy dispersive spectroscopic measurements. F-AgNPs were proved as highly sensitive and selective colorimetric dual sensor for iodide (I−) and ferrous (Fe2+) ions with significant linearity (1–8 and 1–10 µM, respectively), detection limits (LOD = 0.5 and 0.47 µM, respectively) and percent recoveries in real environmental (tap water) and biological (human plasma and urine) samples with adequate test performance. Cumulatively, obtained results revealed that this simple and biocompatible nanosensor has great potential for applications not only in analytical chemistry but also for biological investigations.






Similar content being viewed by others
References
A. Nayal, A. Kumar, R. K. Chhatra, and P. S. Pandey (2014). RSC Adv. 4, 39866–39869.
S. Bothra, R. Kumar, R. K. Pati, A. Kuwar, H. J. Choi, and S. K. Sahoo (2015). Spectrochim. Acta A 149, 122–126.
A. Contino, G. Maccarrone, M. Zimbone, M. Seggio, P. Musumeci, A. Giuffrida, and L. Calcagno (2017). Colloids Surf. A 529, 128–136.
T. Alizadeh and F. Zargr (2020). Mater. Chem. Phys. 240, e122118.
G. R. You, G. J. Park, S. A. Lee, K. Y. Ryu, and C. Kim (2015). Sens. Actuators B 215, 188–195.
S. A. Kumar, N. Thakur, H. J. Parab, S. P. Pandey, R. N. Shinde, A. K. Pandey, S. D. Kumar, and A. V. R. Reddy (2014). Anal. Chim. Acta 851, 87–94.
M. May and O. S. Oluwafemi (2016). Int. J. Electrochem. Sci. 11, 8096–8108.
L. Chen, W. Lu, X. Wang, and L. Chen (2013). Sens. Actuators B 182, 482–488.
M. Maruthupandi, M. Chandhru, S. K. Rani, and N. Vasimalai (2019). ACS Omega 4, 11372–11379.
S. Basiri, A. Mehdinia, and A. Jabbari (2018). Colloids Surf. A 545, 138–146.
A. Niaz, A. Bibi, M. I. Zaman, M. Khan, and A. Rahim (2018). J. Mol. Liq. 249, 1047–1051.
S. Li, Z. Peng, and R. M. Leblanc (2015). Anal. Chem. 87, 6455–6459.
K. Ranoszek-Soliwoda, E. Czechowska, E. Tomaszewska, G. Celichowski, T. Kowalczyk, T. Sakowicz, J. Szemraj, and J. Grobelny (2017). Colloids Surf. B 159, 533–539.
N. Jain, A. Bhargava, M. Rathi, R. V. Dilip, and J. Panwar (2015). PLoS ONE 10, e0134337.
C. L. Udeh (2017). Int. J. Biomed. Mater. Res. 5, 72–77.
B. Denecke, S. Gräber, C. Schäfer, A. Heiss, M. Wöltje, and W. Jahnen-Dechent (2003). Biochem. J. 376, 135–145.
R. S. Komsa-Penkova, G. M. Golemanov, Z. V. Radionova, P. T. Tonchev, S. D. Iliev, and V. V. Penkov (2017). Int. J. Biomed. Res. 10, 90–97.
S. Cartellieri, O. Hamer, H. Helmholz, and B. Niemeyer (2002). Biotechnol. Appl. Biochem. 35, 83–89.
K. Kuwamoto, Y. Takeda, A. Shirai, T. Nakagawa, S. Takeishi, S. Ihara, Y. Miyamoto, S. Shinzaki, J. H. Ko, and E. Miyoshi (2010). Mol. Med. Rep. 3, 651–656.
I. Munir, S. Ajmal, M. R. Shah, A. Ahmad, A. Hameed, and S. A. Ali (2017). Int. J. Biol. Macromol. 101, 131–145.
A. Gebregeorgis, C. Bhan, O. Wilson, and D. Raghavan (2013). J. Colloid. Interface Sci. 389, 31–41.
A. Mishra, N. K. Kaushik, M. Sardar, and D. Sahal (2013). Colloids Surf. B 111, 713–718.
L. Q. Chen, L. Fang, J. Ling, C. Z. Ding, B. Kang, and C. Z. Huang (2015). Chem. Res. Toxicol. 28, 501–509.
A. Siddiqui, H. Anwar, S. W. Ahmed, S. Naqvi, M. R. Shah, A. Ahmed, and S. A. Ali (2020). Spectrochim. Acta A 225, e117489.
S. Naqvi, H. Anwer, S. W. Ahmed, A. Siddiqui, M. R. Shah, S. Khaliq, A. Ahmed, and S. A. Ali (2020). Spectrochim. Acta A 229, e118002.
S. W. Ahmed, H. Anwer, A. Siddiqui, M. R. Shah, and S. A. Ali (2018). Sens. Actuators B 256, 429–439.
V. Prabhawathi, P. M. Sivakumar, and M. Doble (2012). Ind. Eng. Chem. Res. 51, 5230–5239.
J. Helmlinger, C. Sengstock, C. Groß-Heitfeld, C. Mayer, T. A. Schildhauer, M. Köller, and M. Epple (2016). RSC Adv. 6, 18490–18501.
E. Ugwoke, S. O. Aisida, A. A. Mirbahar, M. Arshad, I. Ahmad, T. K. Zhao, and F. I. Ezema (2020). Surf. Interfaces 18, e100419.
J. Liu, M. J. Chang, X. C. Gou, Z. G. Xu, and H. L. Zhang (2012). Colloids Surf. A 404, 112–118.
Y. K. Mohanta, S. K. Panda, R. Jayabalan, N. Sharma, A. K. Bastia, and T. K. Mohanta (2017). Front. Mol. Biosci. 4, e14.
E. Arshadi, S. Sedaghat, and O. Moradi (2018). Asian J. Green Chem. 2, 41–50.
P. Phanjom and G. Ahmed (2017). Adv. Nat. Sci. Nanosci. 8, e045016.
I. Siddiqui and Q. Husain (2019). Colloids Surf. B 173, 733–741.
S. Laera, G. Ceccone, F. Rossi, D. Gilliland, R. Hussain, G. Siligardi, and L. Calzolai (2011). Nano Lett. 11, 4480–4484.
S. Gautam, P. Dubey, and M. N. Gupta (2013). Colloids Surf. B 102, 879–883.
D. Joshi and R. K. Soni (2014). Appl. Phys. A 116, 635–641.
C. Basset, O. Averseng, P. J. Ferron, N. Richaud, A. Hagège, O. Pible, and C. Vidaud (2013). Chem. Res. Toxicol. 26, 645–653.
A. Shrivastava and V. B. Gupta (2011). Chron. Young Sci. 2, e1.
H. Cunha-Silva and M. J. Arcos-Martinez (2019). Talanta 199, 262–269.
R. Dai, X. Wang, Z. Wang, S. Mu, J. Liao, Y. Wen, J. Lv, K. Huang, and X. Xiong (2019). Microchem. J. 146, 592–599.
B. Ma, F. Zeng, F. Zheng, and S. Wu (2011). Chem. Eur. J. 17, 14844–14850.
J. Zhang, Y. Li, and S. Han (2019). Microchem. J. 147, 1141–1146.
A. Iqbal, Y. Tian, X. Wang, D. Gong, Y. Guo, K. Iqbal, Z. Wang, W. Liu, and W. Qin (2016). Sens. Actuators B 237, 408–415.
K. Zheng, C. Lai, L. He, and F. Li (2010). Sci. China Chem. 53, 1398–1405.
A. Samadi-Maybodi, V. Rezaei, and S. Rastegarzadeh (2015). Spectrochim. Acta A 136, 832–837.
N. Scheers, T. Andlid, M. Alminger, and A. S. Sandberg (2010). Electroanalysis 22, 1090–1096.
S. Sen, S. Sarkar, B. Chattopadhyay, A. Moirangthem, A. Basu, K. Dhara, and P. Chattopadhyay (2012). Analyst 137, 3335–3342.
P. G. Mahajan, D. P. Bhopate, A. A. Kamble, D. K. Dalavi, G. B. Kolekar, and S. R. Patil (2015). Anal. Methods 7, 7889–7898.
P. Vasileva, B. Donkova, I. Karadjova, and C. Dushkin (2011). Colloids Surf. A 382, 203–210.
G. L. Wang, X. Y. Zhu, Y. M. Dong, H. J. Jiao, X. M. Wu, and Z. J. Li (2013). Talanta 107, 146–153.
X. Gao, Y. Lu, S. He, X. Li, and W. Chen (2015). Anal. Chim. Acta 879, 118–125.
Acknowledgements
The indirect financial support from the Higher Education Commission (HEC) of Pakistan to SAA (NRPU/R&D/HEC 20-3891) is thankfully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10876_2021_2074_MOESM1_ESM.pptx
Supplementary file1 (PPTX 818 kb). Fig. S1. Stability profile of F-AgNPs. (a) monitoring the effect of heat after 15 min incubation at various temperatures (25–100 ºC), (b) effect of variable pH 2–13, (c) effect of salt (NaCl) concentrations (0.25, 0.5, 1, 1.5, 2 M). (d) monitoring the storage stability at 4 °C and (e) at room temperature (28 ± 2 ºC). Fig. S2. Calibration curves prepared for protein estimation and calculation of loading capacity, using standard protein BSA (blue) and fetuin-A (red) itself because of heavy posttranslational modifications. Inset: 8% Native PAGE analysis of the pure fetuin-A and F-AgNPs. BSA is also used as control. Fig. S3. Effect of different ions (100 μM) on the absorption intensity of F-AgNPs. See "Experimental" section for details. Fig. S4. Interference assay of the competing metal ions. Effect of interfering ions on F-AgNPs for I− detection, change in SPR peak alone and upon addition of equimolar concentrations of other interfering metal ions and I- with respect to F-AgNPs + I−. Fig. S5. Interference assay of the competing ions. Effect of interfering ions on F-AgNPs for Fe2+ detection. Change in SPR peak alone and upon addition of equimolar concentrations of other interfering ions and Fe2+ with respect to F-AgNPs + Fe2+. Fig. S6. Application of F-AgNPs as a sensor in real samples. UV–vis spectral changes in SPR peak of F-AgNPs + I− measured in tap water (a), human urine sample (b) and blood plasma (c). Fig. S7. Application of F-AgNPs as a sensor in real samples. UV–vis spectral changes in SPR peak of F-AgNPs + Fe2+ measured in tap water (a), human urine sample (b) and blood plasma (c).
Rights and permissions
About this article
Cite this article
Jamal, H.S., Raja, R., Ahmed, S.W. et al. Simultaneous Colorimetric Sensing of Anion (I−) and Cation (Fe2+) by Protein Functionalized Silver Nanoparticles in Real Samples. J Clust Sci 33, 1501–1514 (2022). https://doi.org/10.1007/s10876-021-02074-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-021-02074-9