Abstract
In freshwater ecosystems, phosphorus (P) is often considered a growth-limiting nutrient. The use of fertilizers on agricultural fields has led to runoff-driven increases in P availability in streams, and the subsequent eutrophication of downstream ecosystems. Isolated storms and periodic streambed dredging are examples of two common disturbances that contribute dissolved and particulate P to agricultural streams, which can be quantified as soluble reactive P (SRP) using the molybdate-blue method on filtered water samples, or total P (TP) measured using digestions on unfiltered water reflecting all forms of P. While SRP is often considered an approximation of bioavailable P (BAP), research has shown that this is not always the case. Current methods used to estimate BAP do not account for the role of biology (e.g., NaOH extractions) or require specialized platforms (e.g., algal bioassays). Here, in addition to routine analysis of SRP and TP, we used a novel yeast-based bioassay with unfiltered sample water to estimate BAP concentrations during two storms (top 80% and > 95% flow quantiles), and downstream of a reach where management-associated dredging disturbed the streambed. We found that the BAP concentrations were often greater than SRP, suggesting that SRP is not fully representative of P bioavailability. The SRP concentrations were similarly elevated during the two storms, but remained consistently low during streambed disturbance. In contrast, turbidity and TP were elevated during all events. The BAP concentrations were significantly related to turbidity during all disturbance events, but with TP only during storms. The novel yeast assay suggests that BAP export can exceed SRP, particularly when streams are not in equilibrium, such as the rising limb of storms or during active dredging.
Similar content being viewed by others
Introduction
Phosphorus (P) is a growth-limiting nutrient for aquatic plants, algae, and microbes in many freshwater environments (Schindler and Fee 1974; Paerl et al. 2016). Excess P, from organic and inorganic fertilizers applied to farmland, can be mobilized in soils, enter adjacent waterways, and result in the eutrophication of downstream water bodies, leading to excessive algal growth and economic and ecological damage (Michalak et al. 2013; Rabalais and Turner 2019). The majority of annual P export from streams and ditches in the agricultural Midwest occurs during the top 50% of flows (Royer et al. 2006), when dissolved or particulate P moves from fields to adjacent streams, often through subsurface tile drains (Vidon and Cuadra 2011; King et al. 2015; Williams et al. 2016). Overland flow-induced erosion also results in the export of excess P (Ellison and Brett 2006; Brennan et al. 2017), especially if that occurs after fertilizer application (Gentry et al. 2007). As storms are expected to increase in frequency and volume under a changing climate in the Midwestern US, particularly during winter and spring, increased P export and eutrophication events are likely to become more common and severe in the future (Michalak et al. 2013).
Other types of human-induced disturbance can alter streambeds, resulting in the increased export of P even at baseflow conditions. For example, dredging is a common management practice used to maintain effective drainage in many low-gradient agricultural streams, which also prevents flooding in surrounding fields, and maintains crop productivity. Research characterizing the short and long-term impacts of dredging on dissolved and particulate nutrient export is limited, and the existing studies provide conflicting evidence. These studies have concluded that dredging does not affect streambed P retention (Macrae et al. 2003), or that dredged reaches can be P sinks (Smith and Huang 2010; Oldenborg and Steinman 2019) or P sources to the water column (Smith et al. 2006). Therefore, there is a need to document the impact of dredging on both dissolved and particulate P export to inform drainage management practices in the agricultural Midwest.
In aquatic systems, P concentrations are often measured either as soluble reactive P (SRP) or total P (TP). Soluble reactive P is operationally defined as the amount of dissolved P that reacts with molybdate in a filtered sample (Murphy and Riley 1962), and is assumed to approximate orthophosphate, which is readily assimilated by aquatic organisms (Wetzel 2001; Cembella et al. 1982; Karl 2000). Thus, SRP typically serves as a proxy for bioavailable P (BAP; Wetzel 2001; Reynolds and Davies 2001). However, it has been noted that BAP is likely higher than SRP due to other forms of bioavailable P not captured by the operational definition of SRP, such as many organic substances (e.g., DNA, RNA; Li and Brett 2013) and P sorbed to particles (Pacini and Gächter 1999). In contrast, TP is likely an overestimate of BAP, given that unfiltered samples contain particles with recalcitrant or bound P that is biologically unavailable (Li and Brett 2013).
Many researchers have sought methods to measure the true BAP available in freshwater environments (Boström et al. 1988), including incubations with algae (Li and Brett 2013) or bacterioplankton (Jansson et al. 2012), chemical extractions using NaOH (De Pinto et al. 1986; Pionke and Kunishi 1992; Lottig and Stanley 2007), and with qPCR (Yang et al. 2018). These approaches have been successful in estimating BAP concentrations and identified when and where SRP is not a good proxy for BAP (Hudson et al. 2000; Jansson et al. 2012; Li and Brett 2015), but these methods are often costly and time-consuming (Mohamed et al. 2019).
In this study, we compared the impacts of storms and baseflow streambed disturbance (i.e., dredging) on SRP, BAP, and TP concentrations and export in an agricultural stream (Shatto Ditch, IN). To estimate BAP concentrations, we tested the utility of a simple yeast-based bioassay on freshwater samples (Shepherd et al. 2021). We chose yeast because they grow to levels linearly related to low concentrations of a limiting nutrient (Saldanha et al. 2004), they are robust to environmental perturbations (e.g., pH, temperature, and light), and they are resistant to herbicides and insecticides that may be found in agricultural streams (Ravishankar et al. 2020). Unlike algae, yeast have relatively fast incubation times (i.e., days to complete an assay for yeast as compared to weeks for algae), and they behave consistently and predictably in culture systems (Botstein and Fink 2011). Furthermore, this bioassay does not require the use of hazardous or toxic chemicals unlike analyses for SRP and TP.
We used this approach to measure BAP concentrations, along with SRP and TP, from grab samples collected during three disturbances: (1) a small rain event (discharge, Q = top 80% of flows), (2) downstream of a reach being actively dredged, and (3) above and below the dredged reach during a rain-on-snow event (Q = top 10% of flows) that occurred 7 months after dredging. We compare the concentration and export of different operational definitions of P during each of these disturbances and their relationships with each other, as well as with stream turbidity. We predicted that SRP < BAP < TP in most samples, with more variation among P operational definitions when disturbance pushes the system out of equilibrium (e.g., during the rising limb of storms or active dredging), and less variation when the stream is returning to or in equilibrium (e.g., the falling limb of storms or when dredging ceases). With this work, we highlight the importance of understanding how different operational measurements of P, particularly BAP, vary with different aspects of stream disturbance.
Methods
Study site
We conducted this study in the Shatto Ditch Watershed (SDW), a 13.5 km2 agricultural watershed in north-central Indiana (Fig. 1). Land use in the watershed is > 85% row crop agriculture, in corn/soybean rotation, and there is one poultry feedlot operation located within the watershed boundaries. Winter cover crops (primarily annual and cereal ryegrass) have been planted during the fallow period on 62–69% of the croppable acres since October 2013 (Hanrahan et al. 2018). The stream is approximately 2 m wide at the watershed outlet and has a flashy discharge regime that varies seasonally, with discharge (Q) ranging from 10 to 3800 L s−1 (median Q = 70 L s−1). The data in this study were collected as a part of a long-term project that has supported bi-weekly grab sampling for water chemistry since 2008 (Mahl et al. 2015; Hanrahan et al. 2018) and a recent evaluation of sediment P dynamics (Trentman et al. 2020). Continuous discharge (15-min intervals) at the watershed outlet were recorded using a USGS gaging station (gage# 03331224), rainfall was measured at 5-min intervals using a tipping-bucket rain gage (Onset; model: S-RGA-M002) deployed in the watershed, and estimates of snowfall and snow-water equivalents were obtained from the local National Weather Service Office (North Webster, IN).
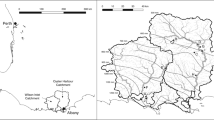
Map of the Shatto Ditch Watershed and the stationary location for each high frequency sampling event. The inset shows the sampling location for storm 1 and storm 2-B. The sampling location for storm 2-A was located at the top of the dredged reach and is on the map. The streamed disturbance took place over three days, with the disturbance taking place over a new 200 m section of the stream each day (represented in the inset). The pictures represent a peak flow during storm 1, b active dredging during the streambed disturbance, and c the rising limb of storm 2
Targeted water sampling
We conducted targeted, high-frequency sampling during three watershed disturbance events where we anticipated elevated export of elevated export of P from either the terrestrial landscape or stream sediments. The events include a small storm (i.e., low precipitation), a large storm (i.e., high precipitation), and streambed disturbance associated with dredging; all events took place within a single year (Fig. 2). For each event, we used a ISCO Automated Water Sampler (3700; Teledyne, Lincoln, NE) to collect stream water into clean 1L plastic bottles at event-specific timed intervals, and we programmed the samplers to rinse the sample tube once before and after collecting each sample. From each bottle, we collected filtered (Type A/E glass fiber filter; Pall, Ann Arbor, MI) and unfiltered samples in 50 mL centrifuge tubes, which were frozen for later laboratory analyses for SRP, BAP, and TP. We inverted each bottle multiple times to ensure that the sample was well-mixed before aliquoting for individual analysis. We also measured the turbidity of a 15 mL aliquot of each bottle using a calibrated portable turbidimeter (2100Q; Hach, Loveland, CO). The longitudinal location of the sampler varied slightly (up to ~ 400 m) depending on the type of event sampled (Fig. 1).
The first event occurred in April 5–7, 2017 during a small storm (hereafter “storm 1”; Fig. 2), where we collected hourly samples through the peak of the hydrograph. The second event occurred immediately downstream of a reach that was being dredged to remove stream sediments in order to increase the streambed slope and velocity (hereafter “streambed disturbance”), which took place prior to the construction of small floodplains (i.e., a two-stage ditch; ~ 3 m on each side of the stream) along a 2 km stretch of the stream. The contractors first dredged a section of the reach (~ 200 m), then returned to the beginning of that section to construct the adjacent floodplain, and this process was repeated in ~ 200 m sections, with the contractors moving sequentially upstream each day. We sampled during the first three days of activity and captured periods of both active streambed dredging and while lateral floodplains were being constructed. We placed the ISCO sampler at the base of the dredged reach throughout, to monitor what was exported from that reach, with the activity during this disturbance often beginning early in the morning and stopping in the afternoon, coincident with the workday. The stream turbidity stabilized near baseline within a few hours after dredging was completed.
Dredging of the first section began on July 31, 2017, and due to logistical constraints, we were only able to initiate sampling during the first round of floodplain construction. We set the ISCO to sample every 20–60 min (depending on activity), and the type of activity (either dredging or floodplain construction) was documented. During the streambed disturbance, 70% of sample collection occurred during active dredging and 30% of samples were collected during floodplain construction. On Day 1, dredging occurred first, followed by two hours of floodplain construction, and five hours of dredging. On Day 2, floodplain construction occurred first for three hours, followed by dredging for four hours. On Day 3, dredging occurred first for an hour, followed by two hours of floodplain construction, and another three hours of dredging.
The third event was a rain-on-snow precipitation event (hereafter “storm 2”) that occurred in February 2018 (Fig. 2), resulting in historic flooding in the region and the largest documented Q recorded in 12 years at the SDW. We collected samples at two sites, above (Storm 2-A) and below (Storm 2-B) the 2 km dredged reach (Fig. 1). We estimated Q at Storm 2-B using area-weighting with the downstream USGS gage. On February 18, 2018, temperatures rose above freezing, initiating snowmelt on the landscape, which was then followed by two periods of rainfall (total = 11.7 cm) over two days, and resulted in two discharge peaks. We began collecting samples at 1–3 h intervals on the morning of February 19, 2019, collecting three samples before the onset of rainfall, but after snowmelt. We stopped sampling for 20 h beginning on February 20 (rising limb of the second peak) due to safety concerns that the stream would overflow its banks and damage sampling equipment. We resumed sampling again at noon February 21 after the stream began to recede, and we stopped sampling for this event after five days, before the stream fully returned to baseflow but capturing the bulk of the storm and its falling limb.
Nutrient measurements
We measured SRP from filtered samples using the molybdate-blue ascorbic acid method and a Lachat Quickchem Analyzer (Hach Company, Loveland CO; method = 10-115-01-1-Q). We measured TP on unfiltered samples using an in-line UV persulfate digester on a Lachat Quickchem Analyzer (method = 10-115-01-3-E). For TP, we used sodium pyrophosphate, sodium tripolyphosphate, and trimethyl phosphate check standards to determine digest efficiency; recovery efficiencies were typically between 70 and 95%.
BAP yeast assay
We estimated BAP on a subset of samples using a yeast assay (hereafter “bioassay”) based on a method developed by Shepherd et al. (2021). We had originally intended to analyze all the available samples; however, this effort was curtailed due to lab shutdown associated with the SARS-CoV-2 pandemic. We selected samples across the hydrograph of the larger storm, with more focus on when the hydrograph was changing, and across all days of the dredging event. Given this, we believe the subset of BAP samples provided here are representative of the full dataset. The bioassay works by incubating cultured P-starved yeast with unfiltered sample water and media that ensures P-limitation of the yeast. After a 4-day incubation, we measured the abundance of yeast using absorbance (corrected for background turbidity), and estimated the BAP of the samples by comparing the observed yeast densities to those obtained from a standard curve of yeast incubated in media containing known amounts of KH2PO4.
The yeast used in this bioassay was Saccharomyces cerevisiae strain DBY10148, a strain adapted to growth in continuous culture systems (Saldanha et al. 2004). Before the assay, we depleted the internal P stores of the cells by growing the yeast in P-limiting media (see below) for four days at 30 °C, until they reached saturation, at which point they were stored at 4 °C until the assay. To initiate each bioassay, we added 100 µL of each sample, in triplicate, to a 96 well plate, not using wells along the edge of the plate to avoid edge-related artifacts during absorbance measurements. Instead, we filled outer wells with double deionized water and antibiotics (ampicillin and kanamycin) to prevent microbial activity in the water, which could contaminate the samples. We then added 100 µL of a mixture containing the P-limited media and P-starved yeast to each sample well; the P-limited media contained sufficient concentrations of micronutrients (Ca, Na, Mg, K, S), glucose, ammonium, trace metals, and vitamins to ensure that P would be the only growth-limiting factor, and antibiotics were included to prevent the growth of bacteria. We created our standard curve on a separate plate, adding increasing amounts of KH2PO4 with medium and yeast (total volume = 200 µL), and then we stored the plates in an incubator at 30 °C for four days. On the fourth day, we thawed a previously aliquoted subset of each sample and added it to adjacent wells in triplicate with media as above, to correct each sample for background turbidity. We transferred the plate to a plate reader that was set to 30 °C and shook the samples in an orbital formation for 1 min, at 807 cycles per minute, in order to mix and suspend the yeast in each sample well. We then immediately measured the absorbance of yeast at 600 nm, and we determined BAP concentration (µg L−1) by comparing the background-corrected absorbance value for each sample to the standard curve. For additional details, see Shepherd et al. (2021)
Results
Stream hydrology during disturbances
Storm 1 had relatively dry antecedent precipitation conditions with minimal 7-day antecedent rainfall, no 7-day antecedent snowfall, and no snowpack before the onset of the storm (Table 1). There was a single discharge peak with Q increasing from 280 L s−1 to a peak of 720 L s−1, while the average Q across the sampling period was 310 L s−1 (Fig. 3). For the streambed disturbance, there was no rainfall in the previous 7-days, nor during the sampling period (Table 1), and Q averaged 180 L s−1, declining by 60 L s−1 over the 3-day sampling period (Fig. 4). Storm 2 had higher 7-day antecedent rainfall than storm 1, and 2.5–5 cm of snow-water equivalent on the landscape, before storm onset (Table 1), and was larger than storm 1 based on bulk precipitation and streamflow. At Storm 2-A, Q peaked at 900 L s−1 and 1700 L s−1 during the first and second peaks, respectively (Fig. 5a). At Storm 2-B, Q peaked at 1780 and 3650 L s−1 for the first and second peaks, respectively (Fig. 5b), and average Q during the sampling period was nearly sixfold higher than during storm 1.
P concentrations and turbidity during disturbances
The peak SRP concentration during storm 1 was 224 µg L−1 and was lower than peak TP (286 µg L−1; Fig. 3). The average SRP concentration during storm 1 was 95 µg L−1 compared to the average TP concentration of 143 µg L−1. Finally, maximum turbidity for storm 1 was 48 NTUs, and turbidity averaged 27 NTUs across the sampling period (Fig. 3).
For the streambed disturbance, SRP concentrations were relatively low, and concentrations did not change significantly between dredging and floodplain construction periods, although the average SRP concentrations decreased from day 1 to day 3 (Fig. 4). The average TP concentrations were generally an order of magnitude higher than SRP, but, like SRP, decreased from day 1 to day 3 (Fig. 4). On each day, water column turbidity was higher during dredging activities than during floodplain construction, and in contrast to SRP and TP, the average turbidity increased from day 1 to day 3 (Fig. 4).
Using our yeast-based assay for BAP (Shepherd et al. 2021), we estimated BAP concentrations on a subset of samples from the streambed disturbance, and observed that BAP concentrations were variable on day 1, ranging between 13 and 179 µg L−1. On days 2 and 3, BAP concentrations were typically higher during dredging and lower during floodplain construction (Fig. 4). Like turbidity, the average BAP concentration increased with each consecutive day (Fig. 4). The BAP concentrations were generally closer to the TP values than the SRP values, although, on the final day of sampling, BAP was generally greater than TP.
For storm 2, SRP concentration at peak flow at storm 2-A was 320 µg L−1 for the first peak and 163 µg L−1 before the second peak (Fig. 5a), while at storm 2-B, SRP concentration at peak flow was 238 for the first peak and 155 µg L−1 before the second peak (Fig. 5b). The average SRP concentrations were similar at sites storm 2-A and storm 2-B (209 vs 206 µg L−1, respectively), but the average SRP concentration at storm 2-B was twofold higher during storm 2 (i.e., top 10% of flows) compared to storm 1 (i.e., top 80% of flows). The TP concentrations during peak flow at storm 2-A were nearly threefold lower than the peak concentration of storm 2-B (Fig. 5). The average TP concentrations at storm 2-A were lower than at storm 2-B (364 vs 425 µg L−1, respectively), while the average TP concentration at storm 2-B was threefold higher during storm 2 compared to storm 1. The turbidity at peak flow at storm 2-A was five-fold lower than peak flow at storm 2-B (Fig. 5). The average turbidity at storm 2-A was lower than at storm 2-B (493 vs 88 NTUs, respectively), while the average turbidity at storm 2-B was 20-fold higher during storm 2 than storm 1.
Again, we measured BAP on a subset of available samples from storm 2-B and concentrations ranged between 81 µg L−1 measured after the first peak and 617 µg L−1 on the last available sample before the second peak (Fig. 5b). The BAP concentrations were closer to the SRP values than the TP values at the onset of sample collection and during the falling limb of the storm, while BAP was closer to TP during the highest period of rainfall and increases in discharge. The average BAP concentration for all samples was 274 µg L−1.
Storm hysteresis patterns of P species and turbidity
For both storms, all P species and turbidity exhibited clockwise hysteresis (Fig. 6), as indicated by higher measurements on the rising limb than the falling limb of the storm hydrograph. During storm 1, the hysteresis patterns between SRP, TP, and turbidity were similar in shape, but with higher overall concentrations of TP compared to SRP (Fig. 5a). Also, SRP and TP concentrations returned to near pre-storm (i.e., background) values despite Q being double background flows, while turbidity remained higher than pre-storm values at the end of sampling (Fig. 6a). During the rising limb of storm 2, at both sites, the increase in TP was much larger than the increase in SRP (Figs. 6b, c), and at storm 2-B, BAP increased during the rising limb with values between SRP and TP (Fig. 6c). During the falling limb of storm 2, SRP and TP were still higher than pre-storm values when sampling ended at site 2-A, while turbidity had returned to pre-storm values. At storm 2-B during the falling limb, all P species and turbidity returned to pre-storm values by the end of sampling (Fig. 6b, c).
Hysteresis graphs relating P concentrations (SRP, BAP, and TP) and turbidity with discharge during storm 1 (a), and storm 2 above (b) and storm 2 below (c) the dredged reach. The gray arrows represent the general hysteresis pattern, and lighter and darker colors represent samples collected earlier and later in the hydrograph. In panel C we show a subset of the data across the hydrograph in order to better identify hysteresis trends
Mass loss of P species during storms
We calculated the yield for each P species exported during each storm (i.e., kg exported per hectare of drainage area), as well as P export (by species) during each day of the streambed disturbance, by interpolating loads between each sample (Table 2). During storm 1, both SRP and TP loss was highest on the rising limb, and lower on the falling limb; total SRP and TP yield was 0.025 kg ha−1 and 0.037 kg ha−1, respectively, and the proportion of SRP to TP yield was 69%. During the streambed disturbance, total SRP export was 0.3 kg compared to BAP = 2.0 kg and TP = 1.9 kg (Table 2), thus slightly more P was exported in bioavailable form than was characterized by TP, and the proportion of SRP to TP loss was 16%. During storm 2, SRP and TP yields were 1.2 and 1.5 times lower at storm 2-A than at storm 2-B. Total SRP and TP yields at storm 2-B were 0.42 kg ha−1 and 0.91 kg ha−1, respectively, which were eightfold and 15-fold higher than for storm 1, and the BAP yield at storm 2-B was 0.47 kg ha−1. Finally, the total yield ratios were SRP:BAP = 89%, BAP:TP = 51%, while SRP:TP = 46%, which was very similar to storm 1. Finally, most of the mass loss for all P species occurred during the falling limb at both sites storm 2-A and storm 2-B (Table 2).
BAP and its relationship with SRP and TP
We compared the relationships between turbidity and the P species (BAP, SRP, TP), analyzing the data from storms separately from the streambed disturbance. We found that SRP and TP were significantly related but showed different relationships with streambed disturbance vs. storms (Fig. 7a). The variation in TP concentrations was better explained by SRP concentrations during storms relative to the streambed disturbance (R2 = 0.51 vs 0.17, respectively), and the TP concentrations were always higher than SRP (Fig. 7a). The slope for the relationship between SRP and TP was nearly double during storms compared to the streambed disturbance event (Fig. 7a). We did not see a significant relationship between SRP and BAP; however, BAP was higher than SRP for the majority of samples (Fig. 7b). The BAP was more likely to be similar to the SRP during storms, as opposed to the streambed disturbance. The BAP concentrations were significantly related to TP concentrations during storms, with TP concentrations being generally higher than BAP (Fig. 7c). In contrast, BAP and TP were not related during the streambed disturbance, and nearly half of these samples had higher BAP than TP concentrations. Finally, BAP concentrations were significantly related to turbidity during both storms and streambed disturbance, and turbidity explained similar amounts of variation in BAP with both storms and streambed disturbance (R2 = 0.34 vs 0.35, respectively; Fig. 7d).
Discussion
The estimation of bioavailable P is critical for understanding limits on algal productivity in freshwaters, especially to better understand algal blooms in lakes and estuaries; however, current approaches are logistically challenging or do not accurately reflect the role of biology in extracting P from sediments (Mohamed et al. 2019). Here, we used a simple yeast-growth assay, with a 4-day incubation time and minimal specialized equipment (Shepherd et al. 2021), to examine patterns in BAP relative to SRP and TP sampled during multiple disturbances in an agricultural stream. We found unique P signatures among individual storms and streambed disturbance, suggesting that BAP availability and downstream export can be heterogeneous. Moreover, the novel yeast assay provides quantitative evidence that BAP export can exceed SRP, particularly when streams are not in equilibrium, which enhances our understanding of BAP export during disturbances.
P export during storms
We found that water column concentrations of SRP, BAP, and TP behaved similarly in response to elevated precipitation during both storm events, albeit at different magnitudes. The BAP concentration was similar to SRP during pre-storm samples, and higher than SRP during periods of precipitation. In all, the BAP:TP yield at storm 2-B was 51%, which was 5% higher than the SRP: TP yield (at 46%). Storm runoff from fields may contain bioavailable P not characterized by SRP, including highly labile P monoesters, which have been identified as the most prominent form of organic P entering streams from agricultural fields (Darch et al. 2014). Furthermore, Li and Brett (2013) found that many organic compounds (i.e., DNA, RNA, etc.) were also bioavailable, but not represented by measured SRP concentrations. Thus, we suggest that our BAP assay was able to capture forms of P that are bioavailable but not represented with estimates of SRP.
All P species exhibited clockwise hysteresis, which means that P concentrations were lower as precipitation ceased and flow receded and is representative of a quick transfer of P to the water column with the onset of precipitation (Williams 1989). Clockwise hysteresis patterns seem to be common in agricultural streams in the SRP and TP range we observed (House and Warwick 1998; Bowes et al. 2005; Outram et al. 2016), but we note that anticlockwise hysteresis have been documented, particularly during the fallow season (Sadeghi et al. 2017; Bender et al. 2018). The clockwise hysteresis patterns in the SDW could indicate that soil P sources are exhausted, leading to lower P on the falling limb, but this seems unlikely given that Mehlich-III P concentrations in the SDW soils are consistently high \((\overline{x }\)= 170 mg kg−1; J. Tank, unpublished data). The higher SRP concentrations during the rising limb could be explained by the quick delivery of dissolved P to the stream through subsurface tile drains; which has been observed in tile drains of SDW (M. Trentman, unpublished data) and other tile drain studies (Gentry et al. 2007; Vidon et al. 2012). Likewise, the disturbance and scouring of stream sediments during storms could lead to immediate movement of sediments and particulate P to the water column (Outram et al. 2016), especially after the stream bed has been recently disturbed. This situation likely occurred during storm 2, where turbidity and TP increased disproportionately relative to SRP, as highlighted by comparing sites above and below the dredged reach. Overall, our data suggest that elevated P export during the two storms in SDW was limited to periods of precipitation and increased flows.
Average SRP concentrations were similar between storm 1 and storm 2, despite higher average TP concentrations and turbidity during storm 2, which was contrary to the prediction that SRP concentrations would increase with increasing storm size (Sharpley et al. 2008). However, others have noted that during storms, there is increased potential for the sorption of dissolved P to available Fe in suspended sediments (Pacini and Gächter 1999; Rosenberg and Schroth 2017), and we note that turbidity at Storm 2-B > 4500 NTUs, reflecting an abundance of suspended solids in the water column. There is some indication that P sorbed to particles are bioavailable (Pacini and Gächter 1999), which is likely captured by the BAP measurements at Storm 2-B. Storm driven sediment loads may have sorbed added dissolved P resulting in “buffered” SRP concentrations during the larger storm.
Overall, our data suggest that storms may export more BAP than is estimated with SRP, particularly during the rising limb of the hydrograph. High-frequency estimates of BAP during storms are uncommon in the literature (but see Pacini and Gächter 1999), indicating that our data provide important context to improving understanding of storm-induced BAP transport to downstream waters. The measurement of BAP export during extreme storms is vital given that these events can often make up a majority of annual P export (Royer et al. 2006). Moreover, extreme events will be especially important given the anticipated effects of a changing climate, where precipitation amounts and intensity are expected to increase, including the increased likelihood of extreme events resulting from rain-on-snow (Easterling et al. 2017; Musselman et al. 2018).
P export during streambed disturbance
Dredging of streambed sediments represents an extreme form of disturbance that can have significant negative effects on stream biota (Zawal et al. 2016; Dabkowski et al. 2016; Bączyk et al. 2018), but the effects on sediment biogeochemistry are variable (Smith et al. 2006; Smith and Huang 2010; Oldenborg and Steinman 2019). Here, we found that the impacts of dredging increased the downstream transport of unique forms of P compared to that transported during storms. This is likely because the form and type of sediment P below the sediment–water interface differ significantly from those of sediments eroded from surrounding fields (Noll et al. 2009; Audette et al. 2018). The proportion of BAP (relative to TP) was higher during the dredging event than during storm 2, which may be explained by stream sediments disturbed by dredging having a higher proportion of bioavailable P as compared to those from runoff during storms. Consistent with these observations, Logan et al. (1979) found that the proportion of BAP (as assessed by chemical extraction) in stream sediments in Western Ohio and Michigan streams was as high as 89%, perhaps due to the buildup of P bound to organic matter in anoxic sediments trapped below the sediment water interface. Moreover, lab assays have also shown that revealing buried (and anoxic) sediments, that are organic rich, and exposing them to oxic conditions increased phosphatase activity and stimulated mineralization rates (Parsons et al. 2017).
Stream sediments can be dissolved P sinks, storing P deep below the sediment–water interface (Withers and Jarvie 2008; Casillas-Ituarte et al. 2020). Previous studies have measured P in stream sediment profiles and noted the presence of a ‘geochemical cap’ that forms just below the sediment–water interface, preventing the release of stored P back to the water column (Jarvie et al. 2008; Burns et al. 2015). One might expect that removing this layer via dredging would lead to increased dissolved P entering the water column (House 2003; Palmer-Felgate et al. 2009), yet we observed minimal release of SRP during the disturbance event in SDW, compared to increases in BAP and TP during dredging. Furthermore, average daily SRP concentrations declined to near baseflow concentrations by day 3 of activity, and as the active dredging moved away from the sampling point. A potential explanation for this observation is that the SRP may have sorbed to suspended particles (House 2003; Rosenberg and Schroth 2017), which would be removed during the SRP-associated filtration step. In contrast, average BAP concentration and turbidity was highest on day 3. Declines in SRP and increases in BAP may be linked given that some sorbed P may be bioavailable (Pacini and Gächter 1999).
During the three days of streambed disturbance, the total export of BAP was slightly higher than that of TP, driven mainly by relatively high BAP concentrations during the final day of the streambed disturbance. We acknowledge that these outcomes could be a result of methodological limitations in estimating TP, as our recovery rate was between 70 and 95% depending on the form of P tested. Similarly, multiple studies have shown that using persulfate digestion can underestimate TP concentrations, especially in samples containing mineral sediments (Syers et al. 1973; Logan et al. 1979). Dredging, more so than storms, may reveal relatively older sediments containing Al and Fe minerals with increased crystalline structures (Schlesinger and Bernhardt 2013), which could reduce digestion efficiencies (Logan et al. 1979). Additionally, for the BAP assay, yeast are known to acidify their environment and excrete ethanol, which could stimulate the breakdown of more complex forms of P (Rossini et al. 1993), and result in an overestimate of P that is bioavailable to other organisms (i.e., algae). Therefore, there is some uncertainty in our estimates of BAP and TP, which should be considered when comparing measurement methods in this study. Future work could evaluate how the yeast BAP assay compares directly with algal (Li and Brett 2013) or bacterioplankton (Jansson et al. 2012) measurements of BAP.
Conclusions and conceptual framework
We used the data from this study to inform a conceptual framework that describes our understanding and predictions of the role of two contrasting disturbances (storms vs dredging) on the export of different operational definitions of P from an agricultural stream based on direct comparisons of samples from this study (e.g., Fig. 7). We acknowledge that this conceptual framework is based on a dataset that does not perfectly capture the range of hydrological conditions, (e.g., limited sampling at baseflow conditions); however, high-frequency samples of BAP collected alongside SRP and TP measurements are rare in the literature, so we used the available data to conceptualize our observations and predictions.
At baseflow, when stream sediments should be in geochemical equilibrium with overlying stream water, it is likely that SRP, BAP, and TP are similar in concentration and mostly bioavailable. While our data on baseflow conditions is limited to periods before or after disturbances, we found that the three operational definitions of P were sometimes similar (i.e., the end of falling limb of both storms). The amount of P in the water column during baseflow is likely to be low and constant, driven by the cycling of BAP in and out of biological reservoirs or abiotic sorption sites (Wetzel 2001). During streambed disturbance, we found that BAP and TP concentrations were elevated, likely due to the increase in particulate P in suspended particles. The SRP concentrations were relatively low during this period, possibly due the sorption of dissolved P with newly revealed particles, while BAP was relatively high, possibly due to P-rich organic matter being revealed from previously anoxic sediments. Finally, we observed that the proportion of different P operational definitions can vary with the storm hydrograph, with the biggest difference in concentration of P species occurring during the rising limb and storm peak. We suggest that BAP is likely elevated above SRP during this period due to the presence of bioavailable organic P in runoff. Overall, we show that there is disturbance driven variation in the amount BAP exported from this agricultural stream, and that BAP was often larger than SRP, the commonly used proxy for BAP.
While we expect that these results would be similar in many agricultural streams, variation in benthic stream substrate and land use in the watershed may create conditions with different relationships between SRP and BAP than we observed here. Specifically, BAP measurements in streams with larger substrate may deviate less from SRP, particularly during streambed disturbance, given that larger substrata typically contain lower bioavailable P (Logan et al. 1979; Pacini and Gächter 1999). The particular land use types in the watershed may also be important, given that the proportion of agriculture or forest in a watershed can control the amount of bioavailable P in stream sediments (Trentman et al. 2021), and would likely mediate BAP concentrations during both types of disturbances. Finally, conservation practices, such as the addition of cover crops during the winter-spring fallow period, can increase P retention on the landscape (Christopher et al. 2021), preventing P delivery from fields to waterways. Future work characterizing BAP in streams should include sites with these conditions to further our understanding of BAP export from agricultural streams.
Our results have important implications for the management of agricultural lands and the eutrophication of sensitive freshwater ecosystems, like the US Great Lakes. Long-term monitoring of the western Lake Erie tributaries has indicated an increase in SRP inputs to the lake (Jarvie et al. 2017; Koltun 2021), and while long-term estimates of BAP entering the lake have been measured using chemical extractions (Baker et al. 2014), these estimates do not account for organic forms of P that are biologically available. The presence of organic P has been highlighted by Shinohara et al. (2018) using NMR spectroscopy to show that a majority of the bioavailable P entering Lake Erie from the Grand River (a Canadian tributary to Lake Erie dominated by agricultural land use) was from particulate organic phosphorus. Thus, our simple bioassay could improve characterization of bioavailable P entering Lake Erie, and enhance our understanding of the extent and timing of eutrophication more generally.
Data availability
The data can be accessed after publication at the following website: https://github.com/mtrentma.
References
Audette Y, O’Halloran IP, Nowell PM et al (2018) Speciation of phosphorus from agricultural muck soils to stream and lake sediments. J Environ Qual 47:884–892. https://doi.org/10.2134/jeq2018.02.0068
Bączyk A, Wagner M, Okruszko T, Grygoruk M (2018) Influence of technical maintenance measures on ecological status of agricultural lowland rivers—systematic review and implications for river management. Sci Total Environ 627:189–199. https://doi.org/10.1016/j.scitotenv.2018.01.235
Baker DB, Confesor R, Ewing DE et al (2014) Phosphorus loading to Lake Erie from the Maumee, Sandusky and Cuyahoga rivers: the importance of bioavailability. J Great Lakes Res 40:502–517. https://doi.org/10.1016/j.jglr.2014.05.001
Bender MA, dos Santos DR, Tiecher T et al (2018) Phosphorus dynamics during storm events in a subtropical rural catchment in southern Brazil. Agric Ecosyst Environ 261:93–102. https://doi.org/10.1016/j.agee.2018.04.004
Boström B, Persson G, Broberg’ B (1988) Bioavailability of different phosphorus forms in freshwater systems. Hydrobiologia 170:133–155 https://doi.org/10.1007/BF00024902
Botstein D, Fink GR (2011) Yeast: an experimental organism for 21st century biology. Genetics 189:695–704. https://doi.org/10.1534/genetics.111.130765
Bowes MJ, House WA, Hodgkinson RA, Leach DV (2005) Phosphorus–discharge hysteresis during storm events along a river catchment: the River Swale, UK. Water Res 39:751–762. https://doi.org/10.1016/j.watres.2004.11.027
Brennan RB, Scott JT, Sharpley AN et al (2017) Linking soil erosion to instream dissolved phosphorus cycling and periphyton growth. JAWRA J Am Water Resour Assoc 53:809–821. https://doi.org/10.1111/1752-1688.12534
Burns EE, Comber S, Blake W et al (2015) Determining riverine sediment storage mechanisms of biologically reactive phosphorus in situ using DGT. Environ Sci Pollut Res 22:9816–9828. https://doi.org/10.1007/s11356-015-4109-3
Casillas-Ituarte NN, Sawyer AH, Danner KM et al (2020) Internal phosphorus storage in two headwater agricultural streams in the Lake Erie Basin. Environ Sci Technol 54:176–183. https://doi.org/10.1021/acs.est.9b04232
Cembella AD, Antia NJ, Harrison PJ (1982) The utilization of inorganic and organic phosphorous compounds as nutrients by eukaryotic microalgae: a multidisciplinary perspective: Part I. CRC Crit Rev Microbiol 10:317–391. https://doi.org/10.3109/10408418209113567
Christopher SF, Tank JL, Mahl UH, Hanrahan BR, Royer TV (2021) Effect of winter cover crops on soil nutrients in two row-cropped watersheds in Indiana (USA).J. Environ. Qual. Accepted Author Manuscript. https://doi.org/10.1002/jeq2.20217
Dabkowski P, Buczynski P, Zawal A et al (2016) The impact of dredging of a small lowland river on water beetle fauna (Coleoptera). J Limnol. https://doi.org/10.4081/jlimnol.2016.1270
Darch T, Blackwell MSA, Hawkins JMB et al (2014) A meta-analysis of organic and inorganic phosphorus in organic fertilizers, soils, and water: implications for water quality. Crit Rev Environ Sci Technol 44:2172–2202. https://doi.org/10.1080/10643389.2013.790752
De Pinto JV, Young TC, McIlroy LM (1986) Great lakes water quality improvement. Environ Sci Technol 20:752–759. https://doi.org/10.1021/es00150a001
Easterling DR, Kunkel KE, Arnold JR et al (2017) Precipitation change in the United States. In: Wuebbles DJ, Fahey DW, Hibbard KA, Dokken DJ, Stewart BC, Maycock TK (eds) Climate Science Special Report: Fourth National Climate Assessment, vol I. U.S Global Change Research Program, Washington DC, pp 207–230
Ellison ME, Brett MT (2006) Particulate phosphorus bioavailability as a function of stream flow and land cover. Water Res 40:1258–1268. https://doi.org/10.1016/j.watres.2006.01.016
Gentry LE, David MB, Royer TV et al (2007) Phosphorus transport pathways to streams in tile-drained agricultural watersheds. J Environ Qual 36:408–415. https://doi.org/10.2134/jeq2006.0098
Hanrahan BR, Tank JL, Christopher SF et al (2018) Winter cover crops reduce nitrate loss in an agricultural watershed in the central U.S. Agric Ecosyst Environ 265:513–523. https://doi.org/10.1016/j.agee.2018.07.004
House WA (2003) Geochemical cycling of phosphorus in rivers. Appl Geochem 18:739–748. https://doi.org/10.1016/S0883-2927(02)00158-0
House WA, Warwick MS (1998) Hysteresis of the solute concentration/discharge relationship in rivers during storms. Water Res 32:2279–2290. https://doi.org/10.1016/S0043-1354(97)00473-9
Hudson JJ, Taylor WD, Schindler DW (2000) Phosphate concentrations in lakes. Nature 406:54–56. https://doi.org/10.1038/35017531
Jansson M, Berggren M, Laudon H, Jonsson A (2012) Bioavailable phosphorus in humic headwater streams in boreal Sweden. Limnol Oceanogr 57:1161–1170. https://doi.org/10.4319/lo.2012.57.4.1161
Jarvie HP, Mortimer RJG, Palmer-Felgate EJ et al (2008) Measurement of soluble reactive phosphorus concentration profiles and fluxes in river-bed sediments using DET gel probes. J Hydrol 350:261–273. https://doi.org/10.1016/j.jhydrol.2007.10.041
Jarvie HP, Johnson LT, Sharpley AN et al (2017) Increased soluble phosphorus loads to Lake Erie: unintended consequences of conservation practices? J Environ Qual 46:123–132. https://doi.org/10.2134/jeq2016.07.0248
Karl DM (2000) Phosphorus, the staff of life. Nature 406:31–33. https://doi.org/10.1038/35017683
King KW, Williams MR, Macrae ML et al (2015) Phosphorus transport in agricultural subsurface drainage: a review. J Environ Qual 44:467–485. https://doi.org/10.2134/jeq2014.04.0163
Koltun GF (2021) Estimated total phosphorus loads for selected sites on Great Lakes tributaries, water years 2014–2018: U.S. Geological Survey Open-File Report 2020–1145, p 13. https://doi.org/10.3133/ofr20201145
Li B, Brett MT (2013) The influence of dissolved phosphorus molecular form on recalcitrance and bioavailability. Environ Pollut 182:37–44. https://doi.org/10.1016/j.envpol.2013.06.024
Li B, Brett MT (2015) The relationship between operational and bioavailable phosphorus fractions in effluents from advanced nutrient removal systems. Int J Environ Sci Technol 12:3317–3328. https://doi.org/10.1007/s13762-015-0760-y
Logan TJ, Oloya TO, Yaksich SM (1979) Phosphate characteristics and bioavailability of suspended sediments from streams draining into Lake Erie. J Great Lakes Res 5:112–123. https://doi.org/10.1016/S0380-1330(79)72136-8
Lottig NR, Stanley EH (2007) Benthic sediment influence on dissolved phosphorus concentrations in a headwater stream. Biogeochemistry 84:297–309. https://doi.org/10.1007/s10533-007-9116-0
Macrae ML, English MC, Schiff SL, Stone MA (2003) Phosphate retention in an agricultural stream using experimental additions of phosphate. Hydrol Process 17:3649–3663. https://doi.org/10.1002/hyp.1356
Mahl UH, Tank JL, Roley SS, Davis RT (2015) Two-stage ditch floodplains enhance N-removal capacity and reduce turbidity and dissolved P in agricultural streams. JAWRA J Am Water Resour Assoc 51:923–940. https://doi.org/10.1111/1752-1688.12340
Michalak AM, Anderson EJ, Beletsky D et al (2013) Record-setting algal bloom in Lake Erie caused by agricultural and meteorological trends consistent with expected future conditions. PNAS 110:6448–6452. https://doi.org/10.1073/pnas.1216006110
Mohamed MN, Wellen C, Parsons CT et al (2019) Understanding and managing the re-eutrophication of Lake Erie: knowledge gaps and research priorities. Freshw Sci 38:675–691. https://doi.org/10.1086/705915
Murphy J, Riley JP (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36. https://doi.org/10.1016/S0003-2670(00)88444-5
Musselman KN, Lehner F, Ikeda K et al (2018) Projected increases and shifts in rain-on-snow flood risk over western North America. Nat Clim Change 8:808–812. https://doi.org/10.1038/s41558-018-0236-4
Noll MR, Szatkowski AE, Magee EA (2009) Phosphorus fractionation in soil and sediments along a continuum from agricultural fields to nearshore lake sediments: potential ecological impacts. J Great Lakes Res 35:56–63. https://doi.org/10.1016/j.jglr.2008.08.004
Oldenborg KA, Steinman AD (2019) Impact of sediment dredging on sediment phosphorus flux in a restored riparian wetland. Sci Total Environ 650:1969–1979. https://doi.org/10.1016/j.scitotenv.2018.09.298
Outram FN, Cooper RJ, Sünnenberg G et al (2016) Antecedent conditions, hydrological connectivity and anthropogenic inputs: factors affecting nitrate and phosphorus transfers to agricultural headwater streams. Sci Total Environ 545–546:184–199. https://doi.org/10.1016/j.scitotenv.2015.12.025
Pacini N, Gächter R (1999) Speciation of riverine particulate phosphorus during rain events. Biogeochemistry 47:87–109. https://doi.org/10.1007/BF00993098
Paerl HW, Scott JT, McCarthy MJ et al (2016) It takes two to tango: when and where dual nutrient (N & P) reductions are needed to protect lakes and downstream ecosystems. Environ Sci Technol 50:10805–10813. https://doi.org/10.1021/acs.est.6b02575
Palmer-Felgate EJ, Jarvie HP, Withers PJA et al (2009) Stream-bed phosphorus in paired catchments with different agricultural land use intensity. Agric Ecosyst Environ 134:53–66. https://doi.org/10.1016/j.agee.2009.05.014
Parsons CT, Rezanezhad F, O’Connell DW, Cappellen PV (2017) Sediment phosphorus speciation and mobility under dynamic redox conditions. Biogeosciences 14:3585–3602. https://doi.org/10.5194/bg-14-3585-2017
Pionke HB, Kunishi HM (1992) Phosphorus status and content of suspended sediment in a Pennsylvania watershed. Soil Sci 153:452–462
Rabalais NN, Turner RE (2019) Gulf of mexico hypoxia: past, present, and future. Limnol Oceanogr Bull 28:117–124. https://doi.org/10.1002/lob.10351
Ravishankar A, Pupo A, Gallagher JEG (2020) Resistance mechanisms of Saccharomyces cerevisiae to commercial formulations of glyphosate involve DNA damage repair, the cell cycle, and the cell wall structure. G3 (Bethesda) 10:2043–2056. https://doi.org/10.1534/g3.120.401183
Reynolds CS, Davies PS (2001) Sources and bioavailability of phosphorus fractions in freshwaters: a British perspective. Biol Rev 76:27–64. https://doi.org/10.1017/S1464793100005625
Rosenberg BD, Schroth AW (2017) Coupling of reactive riverine phosphorus and iron species during hot transport moments: impacts of land cover and seasonality. Biogeochemistry 132:103–122. https://doi.org/10.1007/s10533-016-0290-9
Rossini D, Porro D, Brambilla L et al (1993) In Saccharomyces cerevisiae, protein secretion into the growth medium depends on environmental factors. Yeast 9:77–84. https://doi.org/10.1002/yea.320090110
Royer TV, David MB, Gentry LE (2006) Timing of riverine export of nitrate and phosphorus from agricultural watersheds in Illinois: implications for reducing nutrient loading to the Mississippi River. Environ Sci Technol 40:4126–4131. https://doi.org/10.1021/es052573n
Sadeghi SH, Ebrahimi Mohammadi S, Singh VP, Chapi K (2017) Non-point source contribution and dynamics of soluble and particulate phosphorus from main tributaries of the Zarivar Lake watershed, Iran. Environ Monit Assess 189:238. https://doi.org/10.1007/s10661-017-5937-z
Saldanha AJ, Brauer MJ, Botstein D (2004) Nutritional homeostasis in batch and steady-state culture of yeast. MBoC 15:4089–4104. https://doi.org/10.1091/mbc.e04-04-0306
Schindler DW, Fee EJ (1974) Experimental lakes area: whole-lake experiments in eutrophication. J Fish Res Bd Can 31:937–953. https://doi.org/10.1139/f74-110
Schlesinger WH, Bernhardt ES (2013) Biogeochemistry, 3rd edn. Elsevier, Amsterdam
Sharpley AN, Kleinman PJA, Heathwaite AL et al (2008) Phosphorus loss from an agricultural watershed as a function of storm size. J Environ Qual 37:362–368. https://doi.org/10.2134/jeq2007.0366
Shepherd HAM, Trentman MT, Tank JL, Praner J, Cervantes A, Chaudhary P, Gezelter J, Marrs AJ, Myers A, Welsh JR, Wu Y-FO, Goodson HV (2021) Development of a yeast-based assay for bioavailable phosphorous. bioRxiv. https://doi.org/10.1101/2021.02.28.433264.
Shinohara R, Ouellette L, Nowell P et al (2018) The composition of particulate phosphorus: a case study of the Grand River, Canada. J Great Lakes Res 44:527–534. https://doi.org/10.1016/j.jglr.2018.03.006
Smith DR, Huang C (2010) Assessing nutrient transport following dredging of agricultural drainage ditches. Trans ASABE 53:429–436. https://doi.org/10.13031/2013.29583
Smith DR, Warnemuende EA, Haggard BE, Huang C (2006) Dredging of drainage ditches increases short-term transport of soluble phosphorus. J Environ Qual 35:611–616. https://doi.org/10.2134/jeq2005.0301
Syers JK, Harris RF, Armstrong DE (1973) Phosphate chemistry in lake sediments 1. J Environ Qual 2:1–14. https://doi.org/10.2134/jeq1973.00472425000200010001x
Trentman MT, Tank JL, Jones SE et al (2020) Seasonal evaluation of biotic and abiotic factors suggests phosphorus retention in constructed floodplains in three agricultural streams. Sci Total Environ 729:138744. https://doi.org/10.1016/j.scitotenv.2020.138744
Trentman MT, Tank JL, Braund D, Entrekin SA (2021) Agricultural layering explains variation in sediment P dynamics in streams draining two distinct agricultural biomes. Aquat Sci 83:7. https://doi.org/10.1007/s00027-020-00764-6
Vidon P, Cuadra PE (2011) Phosphorus dynamics in tile-drain flow during storms in the US Midwest. Agric Water Manag 98:532–540. https://doi.org/10.1016/j.agwat.2010.09.010
Vidon P, Hubbard H, Cuadra P, Hennessy M (2012) Storm phosphorus concentrations and fluxes in artificially drained landscapes of the US Midwest. https://doi.org/10.4236/as.2012.34056
Wetzel RG (2001) Limnology: lake and river ecosystems, 3rd edn. Academic Press, Cambridge
Williams GP (1989) Sediment concentration versus water discharge during single hydrologic events in rivers. J Hydrol 111:89–106. https://doi.org/10.1016/0022-1694
Williams MR, King KW, Ford W et al (2016) Effect of tillage on macropore flow and phosphorus transport to tile drains. Water Resour Res 52:2868–2882. https://doi.org/10.1002/2015WR017650
Withers PJA, Jarvie HP (2008) Delivery and cycling of phosphorus in rivers: a review. Sci Total Environ 400:379–395. https://doi.org/10.1016/j.scitotenv.2008.08.002
Yang Y, Yang J, Zhang X (2018) A qPCR method to quantify bioavailable phosphorus using indigenous aquatic species. Environ Sci Eur 30:32. https://doi.org/10.1186/s12302-018-0163-z
Zawal A, Czachorowski S, Stępień E et al (2016) Early post-dredging recolonization of caddisflies (Insecta: Trichoptera) in a small lowland river (NW Poland). Limnology 17:71–85. https://doi.org/10.1007/s10201-015-0466-3
Acknowledgements
We acknowledge funding for fieldwork through grants from the US Department of Agriculture (USDA) Regional Conservation Partnership Program, the Walton Family Foundation, and the Indiana Soybean Alliance. The development of the yeast assay was funded by a Grant from the National Science Foundation (DBI-1556349). We also thank the private landowners for access to the field sites. Finally, we thank the numerous members of the Tank and Goodson Labs for their assistance in the field and with lab analyses.
Funding
US Department of Agriculture (USDA) Regional Conservation Partnership Program, the National Science Foundation, the Walton Family Foundation, and the Indiana Soybean Alliance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declared that they have no conflict of interest.
Additional information
Responsible Editor: Adam Langley
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Trentman, M.T., Tank, J.L., Shepherd, H.A.M. et al. Characterizing bioavailable phosphorus concentrations in an agricultural stream during hydrologic and streambed disturbances. Biogeochemistry 154, 509–524 (2021). https://doi.org/10.1007/s10533-021-00803-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-021-00803-w