Abstract
Background
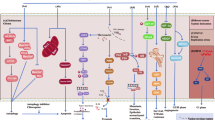
Tumor-treating fields (TTFields) is an emerging non-invasive cancer-treatment modality using alternating electric fields with low intensities and an intermediate range of frequency. TTFields affects an extensive range of charged and polarizable cellular factors known to be involved in cell division. However, it causes side-effects, such as DNA damage and apoptosis, in healthy cells.
Objective
To investigate whether thymidine can have an effect on the DNA damage and apoptosis, we arrested the cell cycle of human glioblastoma cells (U373) at G1/S phase by using thymidine and then exposed these cells to TTFields.
Methods
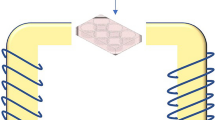
Cancer cell lines and normal cell (HaCaT) were arrested by thymidine double block method. Cells were seeded into the gap of between the insulated wires. The exposed in alternative electric fields at 120 kHz, 1.2 V/cm. They were counted the cell numbers and analyzed for cancer malignant such as colony formation, Annexin V/PI staining, γH2AX and RT-PCR.
Results
The colony-forming ability and DNA damage of the control cells without thymidine treatment were significantly decreased, and the expression levels of BRCA1, PCNA, CDC25C, and MAD2 were distinctly increased. Interestingly, however, cells treated with thymidine did not change the colony formation, apoptosis, DNA damage, or gene expression pattern.
Conclusions
These results demonstrated that thymidine can inhibit the TTFields-caused DNA damage and apoptosis, suggesting that combining TTFields and conventional treatments, such as chemotherapy, may enhance prognosis and decrease side effects compared with those of TTFields or conventional treatments alone.




Similar content being viewed by others
References
Chen W, Dong J, Haiech J, Kilhoffer MC, Zeniou M (2016) Cancer stem cell quiescence and plasticity as major challenges in cancer therapy. Stem Cells Int 2016
Dotiwala F, Harrison JC, Jain S, Sugawara N, Haber JE (2010) Mad2 prolongs DNA damage checkpoint arrest caused by a double-strand break via a centromere-dependent mechanism. Curr Biol 20:328–332
Giladi M, Schneiderman RS, Voloshin T, Porat Y, Munster M, Blat R, Sherbo S, Bomzon Z, Urman N, Itzhaki A et al (2015) Mitotic spindle disruption by alternating electric fields leads to improper chromosome segregation and mitotic catastrophe in cancer cells. Sci Rep 5:18046
Gonzalez CF, Remcho VT (2005) Harnessing dielectric forces for separations of cells, fine particles and macromolecules. J Chromatogr A 1079:59–68
Jeong H, Sung J, Oh SI, Jeong S, Koh EK, Hong S, Yoon M (2014) Inhibition of brain tumor cell proliferation by alternating electric fields. Appl Phys Lett 105:203703
Jo Y, Hwang SG, Jin YB, Sung J, Jeong YK, Baek JH, Cho JM, Kim EH, Yoon M (2018) Selective toxicity of tumor treating fields to melanoma: an in vitro and in vivo study. Cell Death Discov 4:46
Jo Y, Sung J, Jeong H, Hong S, Jeong YK, Kim EH, Yoon M (2019) Effectiveness of a fractionated therapy scheme in tumor treating fields therapy. Technol Cancer Res Treat 18:1533033819845508
Johnson C, Gali VK, Takahashi TS, Kubota T (2016) PCNA retention on DNA into G2/M phase causes genome instability in cells lacking Elg1. Cell Rep 16:684–695
Kim EH, Song HS, Yoo SH, Yoon M (2016) Tumor treating fields inhibit glioblastoma cell migration, invasion and angiogenesis. Oncotarget 7:65125–65136
Kim EH, Jo Y, Sai S, Park MJ, Kim JY, Kim JS, Lee YJ, Cho JM, Kwak SY, Baek JH et al (2019) Tumor-treating fields induce autophagy by blocking the Akt2/miR29b axis in glioblastoma cells. Oncogene 38:6630–6646
Kirson ED, Gurvich Z, Schneiderman R, Dekel E, Itzhaki A, Wasserman Y, Schatzberger R, Palti Y (2004) Disruption of cancer cell replication by alternating electric fields. Cancer Res 64:3288–3295
Kirson ED, Dbaly V, Tovarys F, Vymazal J, Soustiel JF, Itzhaki A, Mordechovich D, Steinberg-Shapira S, Gurvich Z, Schneiderman R et al (2007) Alternating electric fields arrest cell proliferation in animal tumor models and human brain tumors. Proc Natl Acad Sci USA 104:10152–10157
Kirson ED, Giladi M, Gurvich Z, Itzhaki A, Mordechovich D, Schneiderman RS, Wasserman Y, Ryffel B, Goldsher D, Palti Y (2009a) Alternating electric fields (TTFields) inhibit metastatic spread of solid tumors to the lungs. Clin Exp Metas 26:633–640
Kirson ED, Schneiderman RS, Dbaly V, Tovarys F, Vymazal J, Itzhaki A, Mordechovich D, Gurvich Z, Shmueli E, Goldsher D et al (2009b) Chemotherapeutic treatment efficacy and sensitivity are increased by adjuvant alternating electric fields (TTFields). BMC Med Phys 9:1
Liu K, Zheng MY, Lu R, Du JX, Zhao Q, Li ZG, Li YW, Zhang SW (2020) The role of CDC25C in cell cycle regulation and clinical cancer therapy: a systematic review. Cancer Cell Int 20:1–6
Moore N, Lyle S (2011) Quiescent, slow-cycling stem cell populations in cancer: a review of the evidence and discussion of significance. J Oncol 2011
Park JI, Song KH, Jung SY, Ahn J, Hwang SG, Kim J, Kim EH, Song JY (2019) Tumor-Treating Fields Induce RAW264.7 Macrophage Activation Via NK-kappaB/MAPK Signaling Pathways. Technol Cancer Res Treat 18:1533033819868225
Stupp R, Taillibert S, Kanner AA, Kesari S, Steinberg DM, Toms SA, Taylor LP, Lieberman F, Silvani A, Fink KL et al (2015) Maintenance therapy with tumor-treating fields plus temozolomide vs temozolomide alone for glioblastoma: a randomized clinical trial. JAMA 314:2535–2543
Stupp R, Taillibert S, Kanner A, Read W, Steinberg D, Lhermitte B, Toms S, Idbaih A, Ahluwalia MS, Fink K et al (2017) Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: a randomized clinical trial. JAMA 318:2306–2316
Wang Y, Cortez D, Yazdi P, Neff N, Elledge SJ, Qin J (2000) BASC, a super complex of BRCA1-associated proteins involved in the recognition and repair of aberrant DNA structures. Genes Dev 14:927–939
Acknowledgements
This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant number: HI14C3477) and Ministry of Science and ICT (NRF-2020R1A2C1101294) of the Government of the Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Hyesun Jeong, Yunhui Jo, Myonggeun Yoon, and Sunghoi Hong declare that they have no conflict of interest.
Additional information
Myonggeun Yoon and Sunghoi Hong have equally contributed to this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jeong, H., Jo, Y., Yoon, M. et al. Thymidine decreases the DNA damage and apoptosis caused by tumor‐treating fields in cancer cell lines. Genes Genom 43, 995–1001 (2021). https://doi.org/10.1007/s13258-021-01105-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-021-01105-z