Abstract
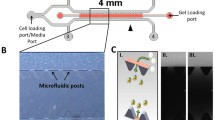
The most challenging phase in the clinical translation of drugs is the complex process of screening various drugs and evaluating their therapeutic effects in vitro and in vivo. Microfluidic models have been recognized as an interesting alternative to animal models for drug screening. Enhanced permeation and retention (EPR) effect is one of the most widely used standard for drug delivery in solid tumors with high vascular density and active angiogenesis. The nascent blood vessels in the tumor vicinity have large gaps between the endothelial junctions which allow nanoparticles to pass through them, resulting in selective extravasation and passive accumulation in the tumor regions. In this study, an attempt has been made to mimic some of the physiological characteristics in solid tumors such as endothelial gap junctions, obstructed blood vessels and EPR using a microfluidic platform for drug screening under dynamic culture conditions. The microfluidic chip was fabricated using soft lithography technique. Numerical simulations were performed to analyze the flow patterns inside the chip. Fluorescent gold nanoclusters (Au NCs) were synthesized and their accumulation in the tumor cells cultured inside the microfluidic chip was studied. The experimental results showed an increased uptake of Au NCs in the cells near the endothelial gap junctions in comparison to the cells away from the junctions. The observations in the study correlated with the leaky nature of the tumor vasculature, owing to the enhancement of vascularization and thereby EPR effect. Hence, the fabricated microfluidic device has the potential of minimizing the number of pre-clinical trials using animal models, allowing easier drug screening.





Similar content being viewed by others
References
Abu Lila AS, Kiwada H, Ishida T (2013) The accelerated blood clearance (ABC) phenomenon: clinical challenge and approaches to manage. J Control Release 172(1):38–47. https://doi.org/10.1016/j.jconrel.2013.07.026
Baish JW et al (2011) Scaling rules for diffusive drug delivery in tumor and normal tissues. Proc Natl Acad Sci USA 108(5):1799–1803. https://doi.org/10.1073/pnas.1018154108
Bobo D, Robinson KJ, Islam J, Thurecht KJ, Corrie SR (2016) Nanoparticle-based medicines: a review of FDA-approved materials and clinical trials to date. Pharm Res 33(10):2373–2387. https://doi.org/10.1007/s11095-016-1958-5
Chen Y, Gao D, Wang Y, Lin S, Jiang Y (2018) A novel 3D breast-cancer-on-chip platform for therapeutic evaluation of drug delivery systems. Anal Chim Acta 1036:97–106. https://doi.org/10.1016/j.aca.2018.06.038
Debnath J, Brugge JS (2005) Modelling glandular epithelial cancers in three-dimensional cultures. Nat Rev Cancer 5(9):675–688. https://doi.org/10.1038/nrc1695
El-Kareh AW, Secomb TW (2004) A theoretical model for intraperitoneal delivery of cisplatin and the effect of hyperthermia on drug penetration distance. Neoplasia 6(2):117–127. https://doi.org/10.1593/neo.03205
Fang J, Islam W, Maeda H (2020) Exploiting the dynamics of the EPR effect and strategies to improve the therapeutic effects of nanomedicines by using EPR effect enhancers. Adv Drug Deliv Rev. https://doi.org/10.1016/j.addr.2020.06.005
Forster JC, Harriss-Phillips WM, Douglass MJ, Bezak E (2017) A review of the development of tumor vasculature and its effects on the tumor microenvironment. Hypoxia 5:21–32. https://doi.org/10.2147/HP.S133231
Gabriel EM, Fisher DT, Evans S, Takabe K, Skitzki JJ (2018) Intravital microscopy in the study of the tumor microenvironment: from bench to human application. Oncotarget 9(28):20165–20178. https://doi.org/10.18632/oncotarget.24957
Hashizume H et al (2000) Openings between defective endothelial cells explain tumor vessel leakiness. Am J Pathol 156(4):1363–1380. https://doi.org/10.1016/S0002-9440(10)65006-7
Huynh E, Zheng G (2015) Cancer nanomedicine: addressing the dark side of the enhanced permeability and retention effect. Nanomedicine 10(13):1993–1995. https://doi.org/10.2217/nnm.15.86
Irvine DJ, Dane EL (2020) Enhancing cancer immunotherapy with nanomedicine. Nat Rev Immunol 20(5):321–334. https://doi.org/10.1038/s41577-019-0269-6
Iyer AK, Khaled G, Fang J, Maeda H (2006) Exploiting the enhanced permeability and retention effect for tumor targeting. Drug Discov Today 11(17–18):812–818. https://doi.org/10.1016/j.drudis.2006.07.005
Jain RK (2001) Delivery of molecular and cellular medicine to solid tumors. Adv Drug Deliv Rev 46(1–3):149–168. https://doi.org/10.1016/S0169-409X(00)00131-9
Kalyane D, Raval N, Maheshwari R, Tambe V, Kalia K, Tekade RK (2019) Employment of enhanced permeability and retention effect (EPR): nanoparticle-based precision tools for targeting of therapeutic and diagnostic agent in cancer. Mater Sci Eng C 98(January):1252–1276. https://doi.org/10.1016/j.msec.2019.01.066
Le Guével X, Hötzer B, Jung G, Hollemeyer K, Trouillet V, Schneider M (2011) Formation of fluorescent metal (Au, Ag) nanoclusters capped in bovine serum albumin followed by fluorescence and spectroscopy. J Phys Chem C 115(22):10955–10963. https://doi.org/10.1021/jp111820b
Leu AJ, Berk DA, Lymboussaki A, Alitalo K, Jain RK (2000) Absence of functional lymphatics within a murine sarcoma: a molecular and functional evaluation. Cancer Res 60(16):4324–4327
Maeda H, Khatami M (2018) Analyses of repeated failures in cancer therapy for solid tumors: poor tumor-selective drug delivery, low therapeutic efficacy and unsustainable costs. Clin Transl Med 7(1):11. https://doi.org/10.1186/s40169-018-0185-6
Maeda H, Tsukigawa K, Fang J (2016) A retrospective 30 years after discovery of the enhanced permeability and retention effect of solid tumors: next-generation chemotherapeutics and photodynamic therapy—problems, solutions, and prospects. Microcirculation 23(3):173–182. https://doi.org/10.1111/micc.12228
Man F, Lammers T, de Rosales RTM (2018) Imaging nanomedicine-based drug delivery: a review of clinical studies. Mol Imaging Biol 20(5):683–695. https://doi.org/10.1007/s11307-018-1255-2
Matsumura Y, Maeda H (1986) A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res 46(8):6387–6392
Ozcelikkale A, Ghosh S, Han B (2013) Multifaceted transport characteristics of nanomedicine: needs for characterization in dynamic environment. Mol Pharm 10(6):2111–2126. https://doi.org/10.1021/mp3005947
Ozcelikkale A, Moon HR, Linnes M, Han B (2017) In vitro microfluidic models of tumor microenvironment to screen transport of drugs and nanoparticles. Wiley interdisciplinary reviews. Nanomed Nanobiotechnol 9(5). https://doi.org/10.1002/wnan.1460
Park K (2013) Questions on the role of the EPR effect in tumor targeting. J Control Release 172(1):391. https://doi.org/10.1016/j.jconrel.2013.10.001
Roberts WG, Hasan T (1992) Role of neovasculature and vascular permeability on the tumor retention of photodynamic agents. Cancer Res 52(4):924–930
Sindhwani S et al (2020) The entry of nanoparticles into solid tumours. Nat Mater 19(5):566–575. https://doi.org/10.1038/s41563-019-0566-2
Vidal-Meza M, Zhou R, Barua S, Wang C, Park J (2016) Simulation of interstitial nanoparticle flow for development of tumor- on-a-chip device. Proceedings of the 2016 COMSOL conference, Boston, MA, USA pp 5–7
Xiao S, Tang Y, Lv Z, Lin Y, Chen L (2019) Nanomedicine—advantages for their use in rheumatoid arthritis theranostics. J Control Release 316:302–316. https://doi.org/10.1016/j.jconrel.2019.11.008
Youn YS, Bae YH (2018) Perspectives on the past, present, and future of cancer nanomedicine. Adv Drug Deliv Rev 130:3–11. https://doi.org/10.1016/j.addr.2018.05.008
Acknowledgements
Authors would like to acknowledge DBT (BT/NNT/28/1386/2017), DST INSPIRE (INSPIRE/04/2015/00037) and MHRD IMPRINT (4291), Government of India for the research funding. Author MS would like to acknowledge the Ministry of Education, Government of India, for M Tech (bm17mtech11004) fellowship. Author TA would like to thank DST, Government of India for Inspire fellowship (IF160291). Authors would like to acknowledge “The Vasudha Foundations”, Hyderabad for their generous financial support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shaji, M., Mudigunda V., S., Appidi, T. et al. Microfluidic design of tumor vasculature and nanoparticle uptake by cancer cells. Microfluid Nanofluid 25, 46 (2021). https://doi.org/10.1007/s10404-021-02446-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-021-02446-7