Abstract
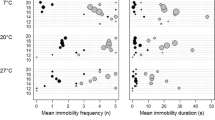
Internal parasites typically are associated with a range of negative effects on their hosts, including reduced energy, which can manifest in behavioral alterations. With this in mind, we examined effects of a naturally-occurring nematode parasite, Chondronema passali, on locomotor activity level in horned passalus beetles, Odontotaenius disjunctus from Georgia, USA. This parasite is not well-studied but can number in the thousands in severely parasitized hosts. Prior study in our lab revealed that parasitized beetles actually consume more wood than unparasitized ones do, leading us to ask here, if parasitized beetles are also more physically active. Beetles were collected from nearby forests and housed individually in our lab. We created a simple tabletop arena to observe beetle locomotor activity, which was gridded and included small stones and paper objects. We allowed individual beetles to traverse the arena for 5 min and recorded the number of grid squares crossed. Then, beetles were dissected to determine parasite presence and level of infection (on a categorical scale). A total of 140 beetles were examined across three collections. Statistical analyses of locomotor activity revealed parasite severity predicted locomotor activity, but paradoxically, lightly-infected beetles were twice as active as those without this nematode. Activity diminished with increasing worm burdens thereafter, but even the group with the most severe burdens did not move less than those with no worms. From these results we conclude that this parasite does not result in overall reduction in activity, but rather it appears to come with heightened locomotion. Alternatively, this result could stem from the fact that more active beetles are simply more likely to contract the parasite.



Similar content being viewed by others
References
Akbulut S, Stamps WT (2012) Insect vectors of the pinewood nematode: a review of the biology and ecology of Monochamus species. Forest Pathol 42:89–99
Allahverdipour HH, Talaei-Hassanloui R, Karimi J, Wang Y, Rochlin I, Gaugler R (2019) Behavior manipulation of mosquitoes by a mermithid nematode. J Invert Pathol 168:5
Altizer S, Hobson KA, Davis AK, De Roode JC, Wassenaar LI (2015) Do healthy monarchs migrate farther? Tracking natal origins of parasitized vs. uninfected monarch butterflies overwintering in Mexico. Plos one 10: 14
Bradley CA, Altizer S (2005) Parasites hinder monarch butterfly flight: implications for disease spread in migratory hosts. Ecol Lett 8:290–300
Calderon L, Davis AK (2016) Observations of Steinernema nematode and Tachinid fly parasites in horned passalus beetles, Odontotaenius disjunctus, from Georgia, U.S.a. Comp Parasitol 83:265–268
Christie JR, Chitwood BG (1931) Chondronema passali (Leidy, 1852) n. g. (Nematoda), with notes on its life history. J Wash Acad Sci 21:356–364
Cox D, Davis AK (2013) Effect of a parasitic nematode, Chondronema passali Leidy (Incertae sedis), on the size and strength of the horned passalus, Odontotaenius disjunctus Illiger (Coleoptera: Passalidae). Coleopts Bull 67:1–7
Davis AK, Prouty C (2019) The sicker the better: nematode-infected passalus beetles provide enhanced ecosystem services. Biol Lett 15:4
Davis AK, Vasquez D, LeFeuvre J, Sims S, Craft M, Vizurraga A (2017) Parasite manipulation of its host's physiological reaction to acute stress: experimental results from a natural beetle-nematode system. Physiol Biochem Zool 90:273–280
Davis AK, Calderon L, Lefeuvre J, Sims S, Pearce J, Prouty C (2020) Healing while parasitized: impact of a naturally-occurring nematode during energy-intensive wound-healing in a beetle. Physiol Entomol 45:72–80
Delahay RJ, Speakman JR, Moss R (1995) The energetic consequences of parasitism: effects of a developing infection of Trichostrongylus tenuis (Nematoda) on red grouse (Lagopus lagopus scoticus) energy-balance, body-weight and condition. Parasitology 110:473–482
Dussaubat C, Maisonnasse A, Crauser D, Beslay D, Costagliola G, Soubeyrand S, Kretzchmar A, Le Conte Y (2013) Flight behavior and pheromone changes associated to Nosema ceranae infection of honey bee workers (Apis mellifera) in field conditions. J Invert Pathol 113:42–51
Ebot-Ojong F, Jurado E, Davis AK (2019) Direct measurement of fight or flight behavior in a beetle reveals individual variation and the influence of parasitism. PLoS One 14:e0216387. https://doi.org/10.1371/journal.pone.0216387
Fellous S, Quillery E, Duncan AB, Kaltz O (2011) Parasitic infection reduces dispersal of ciliate host. Biol Lett 7:327–329
Gray IE (1946) Observations on the life history of the horned passalus. Am Midl Nat 35:728–746
Hawley D, Davis AK, Dhondt AA (2007) Transmission-relevant behaviours shift with pathogen infection in wild house finches (Carpodacus mexicanus). Can J Zool 85:752–757
Hechtel LJ, Johnson CL, Juliano SA (1993) Modification of antipredator behavior of Caecidotea intermedius by its parasite Acanthocephalus dirus. Ecology 74:710–713
Lafferty KD, Shaw JC (2013) Comparing mechanisms of host manipulation across host and parasite taxa. J Exp Biol 216:56–66
LeFeuvre J, Davis AK (2015) Effects of a naturally-occurring nematode parasite on lifting strength and captivity-related body mass patterns in horned passalus beetles, Odontotaenius disjunctus. Coleopts Bull 69:1–7
Leidy J (1852) Some observations on Nematoidea Imperfecta, and descriptions of three parasitic Infusoriae. T Am Philos Soc 10:241–244
Lewis EE, Clarke DJ (2012) Nematode parasites and entomopathogens, pp. 395-424. In F. E. Vega and H. K. Kaya [eds.], insect pathology, 2nd ed. academic press, London
Libersat F, Moore J (2000) The parasite Moniliformis moniliformis alters the escape response of its cockroach host Periplaneta americana. J Insect Behav 13:103–110
Moore J (1995) The behavior of parasitized animals. Bioscience 45:89–96
Nickle WR, Pilitt PA (1969) Chondronema passali (Leidy, 1852) Christie and Chitwood, 1931, Redescribed with observations on its early development. Proc Helm Soc Wash 36:190–193
Reinert JA (1973) Parasites associated with Popilius disjunctus in South Carolina (Coleoptera: Passalidae). Fla Entomol 56:273–276
Ulyshen MD (2018) Ecology and conservation of Passalidae, pp. 129-147. In M. D. Ulyshen [ed.], Saproxylic insects: diversity, ecology and conservation. Springer
van Dijk JGB, Kleyheeg E, Soons MB, Nolet BA, Fouchier RAM, Klaassen M (2015) Weak negative associations between avian influenza virus infection and movement behaviour in a key host species, the mallard Anas platyrhynchos. Oikos 124:1293–1303
Vasquez D Jr, Willoughby A, Davis AK (2015) Fighting while parasitized: can nematode infections affect the outcome of staged combat in beetles? PLoS One 10:e0121614. https://doi.org/10.1371/journal.pone.0121614
Villacide JM, Corley JC (2008) Parasitism and dispersal potential of Sirex noctilio: implications for biological control. Agr Forest Entomol 10:341–345
Acknowledgements
We thank the members of the Altizer lab at the University of Georgia for providing feedback on this experiment and the manuscript. Two anonymous reviewers provided feedback which improved the manuscript. Part of this project was supported by funding from the Center for Undergraduate Research Opportunities at UGA.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Davis, A.K., Hurd, C., Brandon, C. et al. Walking while Parasitized: Effects of a Naturally-Occurring Nematode on Locomotor Activity of Horned Passalus Beetles. J Insect Behav 34, 89–95 (2021). https://doi.org/10.1007/s10905-021-09769-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10905-021-09769-x