Abstract
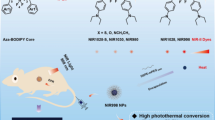
Photothermal therapy (PTT) is one of recently emerging anticancer treatment modalities that involve the use of laser and photosensitizers. To fully explore the therapeutic potential of PTT, it is critical to deliver photosensitizers to tumors specifically. Maltodextrin is a hydrophilic polysaccharide consisting of D-glucose units and has been used as a pharmaceutical excipient because of its excellent biocompatibility. IR820 is a derivative of indocyanine green (ICG), a clinically used imaging agent and has potential as a photosensitizer for PTT. However, the clinical translation of IR820 has been limited by its lack of cancer targeting ability and poor stability in physiological environments. In this study, we hypothesized that IR820-conjugated maltodextrin (MDir) preferentially accumulates in tumors and serves as photothermal agents. Combination of MDir and near-infrared (NIR) laser irradiation significantly increased the temperature, leading to photothermal-mediated apoptosis of cancer cells. In a mouse xenograft models, MDir preferentially accumulated in tumors and induced almost complete thermal ablation of tumors upon NIR laser irradiation. Based on its tumor targeting ability, photothermal conversion efficiency and excellent biocompatibility, we anticipate that MDir holds great translational potential as a fluorescence imaging-guided photothermal agent for cancer treatment.

Similar content being viewed by others
References
Y. C. Wang, K. C. L. Black, H. Luehmann, W. Y. Li, Y. Zhang, X. Cai, D. H. Wan, S. Y. Liu, M. Li, P. Kim, Z. Y. Li, L. H. V. Wang, Y. J. Liu, and Y. A. Xia, ACS Nano, 7, 2068 (2013).
S. Shen, S. Wang, R. Zheng, X. Y. Zhu, X. G. Jiang, D. L. Fu, and W. L. Yang, Biomaterials, 39, 67 (2015).
C. Shirata, J. Kaneko, Y. Inagaki, T. Kokudo, M. Sato, S. Kiritani, N. Akamatsu, J. Arita, Y. Sakamoto, K. Hasegawa, and N. Kokudo, Sci. Rep., 7, (2017).
W. Yang, J. Noh, H. Park, S. Gwon, B. Singh, C. Song, and D. Lee, Biomaterials, 154, 48 (2018).
A. W. Mao, Gastrointest. Tumors, 3, 59 (2016).
M. Alter, U. Hillen, U. Leiter, M. Sachse, and R. Gutzmer, J. Deutsch. Dermatol. Ges., 13, 863 (2015).
T. Tsuda, M. Kaibori, H. Hishikawa, R. Nakatake, T. Okumura, E. Ozeki, I. Hara, Y. Morimoto, K. Yoshii, and M. Kon, Plos One, 12, (2017).
A. Hannah, G. Luke, K. Wilson, K. Homan, and S. Emelianov, ACS Nano, 8, 250 (2014).
C. C. Niu, Y. Xu, S. B. An, M. Zhang, Y. H. Hu, L. Wang, and Q. H. Peng, Sci. Rep., 7, (2017).
G. R. Cherrick, S. W. Stein, C. M. Leevy, and C. S. Davidson, J. Clin. Invest., 39, 592 (1960).
K. Sano, M. Ohashi, K. Kanazaki, A. Makino, N. Ding, J. Deguchi, Y. Kanada, M. Ono, and H. Saji, Bioconjug. Chem., 28, 1024 (2017).
K. K. Wang, Y. F. Zhang, J. Wang, A. Yuan, M. J. Sun, J. H. Wu, and Y. Q. Hu, Sci. Rep., 6, 24721 (2016).
A. Fernandez-Fernandez, R. Manchanda, T. J. Lei, D. A. Carvajal, Y. Tang, S. Z. R. Kazmi, and A. J. McGoron, Mol. Imaging, 11, 99 (2012).
A. Fernandez-Fernandez, R. Manchanda, D. A. Carvajal, T. Lei, S. Srinivasan, and A. J. McGoron, Int. J. Nanomedicine, 9, 4631 (2014).
B. Bahmani, C. Y. Lytle, A. M. Walker, S. Gupta, V. I. Vullev, and B. Anvari, Int. J. Nanomedicine, 8, 1609 (2013).
N. Beziere, N. Lozano, A. Nunes, J. Salichs, D. Queiros, K. Kostarelos, and V. Ntziachristos, Biomaterials, 37, 415 (2015).
F. M. Veronese and G. Pasut, Drug Discov Today, 10, 1451 (2005).
K. Kanazaki, K. Sano, A. Makino, F. Yamauchi, A. Takahashi, T. Homma, M. Ono, and H. Saji, J. Control. Release, 226, 115 (2016).
D. L. Hofman, V. J. Van Buul, and F. J. Brouns, Crit. Rev. Food Sci. Nutrition, 56, 2091 (2016).
N. N. Pavlova and C. B. Thompson, Cell Metab., 23, 27 (2016).
A. Vazquez, J. J. Kamphorst, E. K. Markert, Z. T. Schug, S. Tardito, and E. Gottlieb, J. Cell Sci., 129, 3367 (2016).
X. Ning, W. Seo, S. Lee, K. Takemiya, M. Rafi, X. Feng, D. Weiss, X. Wang, L. Williams, and V. M. Camp, Angew. Chem. Int. Ed., 53, 14096 (2014).
X. Tan, J. Wang, X. Pang, L. Liu, Q. Sun, Q. You, F. Tan, and N. Li, ACS Appl. Mater. Interfaces, 8, 34991 (2016).
J. Zhou, X. Du, C. Berciu, S. J. Del Signore, X. Chen, N. Yamagata, A. A. Rodal, D. Nicastro, and B. Xu, Mol. Ther., 26, 648 (2018).
K. Z. Huang, M. Y. Gao, L. Fan, Y. Y. Lai, H. W. Fan, and Z. C. Hua, Biomater. Sci., 6, 2925 (2018).
X. X. Tan, J. P. Wang, X. J. Pang, L. Liu, Q. Sun, Q. You, F. P. Tan, and N. Li, ACS Appl. Mater. Interfaces, 8, 34991 (2016).
J. Noh, E. Jung, D. Yoo, C. Kang, C. Kim, S. Park, G. Khang, and D. Lee, ACS Appl. Mater. Interfaces, 10, 40424 (2018).
Acknowledgment
This work was supported by a grant of a grant of Ministry of Science and ICT through National Research Foundation (2020M2D9A3094215), Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rampa, D.R., Ko, C., Lee, Y. et al. NIR Dye-Conjugated Maltodextrin for Photothermal Therapy of Cancer. Macromol. Res. 29, 306–312 (2021). https://doi.org/10.1007/s13233-021-9038-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-021-9038-z