Abstract
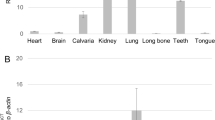
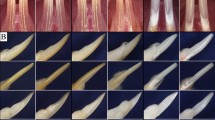
It is widely accepted that cellular processes are controlled by protein phosphorylation and has become increasingly clear that protein degradation, localization and conformation as well as protein–protein interaction are the examples of subsequent cellular events modulated by protein phosphorylation. Enamel matrix proteins belong to members of the secretory calcium binding phosphoprotein (SCPP) family clustered on chromosome 4q21, and most of the SCPP phosphoproteins have at least one S-X-E motifs (S; serine, X; any amino acid, E; glutamic acid). It has been reported that mutations in C4orf26 gene, located on chromosome 4q21, are associated with autosomal recessive type of Amelogenesis Imperfecta (AI), a hereditary condition that affects enamel formation/mineralization. The enamel phenotype observed in patients with C4orf26 mutations is hypomineralized and partially hypoplastic, indicating that C4orf26 protein may function at both secretory and maturation stages of amelogenesis. The previous in vitro study showed that the synthetic phosphorylated peptide based on C4orf26 protein sequence accelerates hydroxyapatite nucleation. Here we show the molecular cloning of Gm1045, mouse homologue of C4orf26, which has 2 splicing isoforms. Immunohistochemical analysis demonstrated that the immunolocalization of Gm1045 is mainly observed in enamel matrix in vivo. Our report is the first to show that FAM20C, the Golgi casein kinase, phosphorylates C4orf26 and Gm1045 in cell cultures. The extracellular localization of C4orf26/Gm1045 was regulated by FAM20C kinase activity. Thus, our data point out the biological importance of enamel matrix-kinase control of SCPP phosphoproteins and may have a broad impact on the regulation of amelogenesis and AI.




Similar content being viewed by others
References
Collier PM, Sauk JJ, Rosenbloom SJ, Yuan ZA, Gibson CW (1997) An amelogenin gene defect associated with human X-linked amelogenesis imperfecta. Arch Oral Biol 42:235–242
Hart TC, Hart PS, Gorry MC, Michalec MD, Ryu OH, Uygur C, Ozdemir D, Firatli S, Aren G, Firatli E (2003) Novel ENAM mutation responsible for autosomal recessive amelogenesis imperfecta and localised enamel defects. J Med Genet 40:900–906
Kida M, Ariga T, Shirakawa T, Oguchi H, Sakiyama Y (2002) Autosomal-dominant hypoplastic form of amelogenesis imperfecta caused by an enamelin gene mutation at the exon-intron boundary. J Dent Res 81:738–742
Ravassipour DB, Hart PS, Hart TC, Ritter AV, Yamauchi M, Gibson C, Wright JT (2000) Unique enamel phenotype associated with amelogenin gene (AMELX) codon 41 point mutation. J Dent Res 79:1476–1481
Hart PS, Hart TC, Simmer JP, Wright JT (2002) A nomenclature for X-linked amelogenesis imperfecta. Arch Oral Biol 47:255–260
Rajpar MH, Harley K, Laing C, Davies RM, Dixon MJ (2001) Mutation of the gene encoding the enamel-specific protein, enamelin, causes autosomal-dominant amelogenesis imperfecta. Hum Mol Genet 10:1673–1677
Parry DA, Brookes SJ, Logan CV, Poulter JA, El-Sayed W, Al-Bahlani S, Al Harasi S, Sayed J, el Raif M, Shore RC, Dashash M, Barron M, Morgan JE, Carr IM, Taylor GR, Johnson CA, Aldred MJ, Dixon MJ, Wright JT, Kirkham J, Inglehearn CF, Mighell AJ (2012) Mutations in C4orf26, encoding a peptide with in vitro hydroxyapatite crystal nucleation and growth activity, cause amelogenesis imperfecta. Am J Hum Genet 91:565–571
Tagliabracci VS, Engel JL, Wen J, Wiley SE, Worby CA, Kinch LN, Xiao J, Grishin NV, Dixon JE (2012) Secreted kinase phosphorylates extracellular proteins that regulate biomineralization. Science 336:1150–1153
Raine J, Winter RM, Davey A, Tucker SM (1989) Unknown syndrome - microcephaly, hypoplastic nose, exophthalmos, gum hyperplasia, cleft-palate, low set ears, and osteosclerosis. J Med Genet 26:786–788
Kingston HM, Freeman JS, Hall CM (1991) A new lethal sclerosing bone dysplasia. Skeletal Radiol 20:117–119
Kan AE, Kozlowski K (1992) New distinct lethal osteosclerotic bone dysplasia (raine syndrome). Am J Med Genet 43:860–864
Acevedo AC, Poulter JA, Alves PG, de Lima CL, Castro LC, Yamaguti PM, Paula LM, Parry DA, Logan CV, Smith CEL, Johnson CA, Inglehearn CF, Mighell AJ (2015) Variability of systemic and oro-dental phenotype in two families with non-lethal Raine syndrome with FAM20C mutations. BMC Med Genet. https://doi.org/10.1186/s12881-015-0154-5
Elalaoui SC, Al-Sheqaih N, Ratbi I, Urquhart JE, O’Sullivan J, Bhaskar S, Williams SS, Elalloussi M, Lyahyai J, Sbihi L, Jaouad IC, Sbihi A, Newman WG, Sefiani A (2016) Non lethal Raine syndrome and differential diagnosis. Eur J Med Genet 59:577–583
Tagliabracci VS, Wiley SE, Guo X, Kinch LN, Durrant E, Wen J, Xiao J, Cui J, Nguyen KB, Engel JL, Coon JJ, Grishin N, Pinna LA, Pagliarini DJ, Dixon JE (2015) A single kinase generates the majority of the secreted phosphoproteome. Cell 161:1619–1632
Kawano S, Morotomi T, Toyono T, Nakamura N, Uchida T, Ohishi M, Toyoshima K, Harada H (2002) Establishment of dental epithelial cell line (HAT-7) and the cell differentiation dependent on Notch signaling pathway. Connect Tissue Res 43:409–412
Mochida Y, Takeda K, Saitoh M, Nishitoh H, Amagasa T, Ninomiya-Tsuji J, Matsumoto K, Ichijo H (2000) ASK1 inhibits interleukin-1-induced NF-kappa B activity through disruption of TRAF6-TAK1 interaction. J Biol Chem 275:32747–32752
Ohyama Y, Lin JH, Govitvattana N, Lin IP, Venkitapathi S, Alamoudi A, Husein D, An C, Hotta H, Kaku M, Mochida Y (2016) FAM20A binds to and regulates FAM20C localization. Sci Rep 6:27784
Mochida Y, Kaku M, Yoshida K, Katafuchi M, Atsawasuwan P, Yamauchi M (2011) Podocan-like protein: a novel small leucine-rich repeat matrix protein in bone. Biochem Biophys Res Commun 410:333–338
Prasad MK, Laouina S, El Alloussi M, Dollfus H, Bloch-Zupan A (2016) Amelogenesis imperfecta: 1 family, 2 phenotypes, and 2 mutated genes. J Dent Res 95:1457–1463
Nalbant D, Youn H, Nalbant SI, Sharma S, Cobos E, Beale EG, Du Y, Williams SC (2005) FAM20: an evolutionarily conserved family of secreted proteins expressed in hematopoietic cells. BMC Genomics 6:11
O’Sullivan J, Bitu CC, Daly SB, Urquhart JE, Barron MJ, Bhaskar SS, Martelli-Junior H, dos Santos Neto PE, Mansilla MA, Murray JC, Coletta RD, Black GC, Dixon MJ (2011) Whole-Exome sequencing identifies FAM20A mutations as a cause of amelogenesis imperfecta and gingival hyperplasia syndrome. Am J Hum Genet 88:616–620
Cho SH, Seymen F, Lee KE, Lee SK, Kweon YS, Kim KJ, Jung SE, Song SJ, Yildirim M, Bayram M, Tuna EB, Gencay K, Kim JW (2012) Novel FAM20A mutations in hypoplastic amelogenesis imperfecta. Hum Mutat 33:91–94
Strawich E, Glimcher MJ (1985) Synthesis and degradation in vivo of a phosphoprotein from rat dental enamel. Identification of a phosphorylated precursor protein in the extracellular organic matrix. Biochem J 230:423–433
Termine JD, Torchia DA, Conn KM (1979) Enamel matrix: structural proteins. J Dent Res 58:773–781
Fincham AG, Belcourt AB, Termine JD (1982) Changing patterns of enamel matrix proteins in the developing bovine tooth. Caries Res 16:64–71
Fincham AG, Hu Y, Lau EC, Slavkin HC, Snead ML (1991) Amelogenin post-secretory processing during biomineralization in the postnatal mouse molar tooth. Arch Oral Biol 36:305–317
Fincham AG, Moradian-Oldak J, Sarte PE (1994) Mass-spectrographic analysis of a porcine amelogenin identifies a single phosphorylated locus. Calcif Tissue Int 55:398–400
Salih E, Huang JC, Strawich E, Gouverneur M, Glimcher MJ (1998) Enamel specific protein kinases and state of phosphorylation of purified amelogenins. Connect Tissue Res 38:225–235 (discussion 241-226)
Kwak SY, Wiedemann-Bidlack FB, Beniash E, Yamakoshi Y, Simmer JP, Litman A, Margolis HC (2009) Role of 20-kDa amelogenin (P148) phosphorylation in calcium phosphate formation in vitro. J Biol Chem 284:18972–18979
Le Norcy E, Kwak SY, Allaire M, Fratzl P, Yamakoshi Y, Simmer JP, Margolis HC (2011) Effect of phosphorylation on the interaction of calcium with leucine-rich amelogenin peptide. Eur J Oral Sci 119(Suppl 1):97–102
Masica DL, Gray JJ, Shaw WJ (2011) Partial high-resolution structure of phosphorylated and non-phosphorylated leucine-rich amelogenin protein adsorbed to hydroxyapatite. J Phys Chem C Nanomater Interfaces 115:13775–13785
Wiedemann-Bidlack FB, Kwak SY, Beniash E, Yamakoshi Y, Simmer JP, Margolis HC (2011) Effects of phosphorylation on the self-assembly of native full-length porcine amelogenin and its regulation of calcium phosphate formation in vitro. J Struct Biol 173:250–260
Shin NY, Yamazaki H, Beniash E, Yang X, Margolis SS, Pugach MK, Simmer JP, Margolis HC (2020) Amelogenin phosphorylation regulates tooth enamel formation by stabilizing a transient amorphous mineral precursor. J Biol Chem 295:1943–1959
Fukae M, Tanabe T, Murakami C, Dohi N, Uchida T, Shimizu M (1996) Primary structure of the porcine 89-kDa enamelin. Adv Dent Res 10:111–118
Chan HC, Mai L, Oikonomopoulou A, Chan HL, Richardson AS, Wang SK, Simmer JP, Hu JC (2010) Altered enamelin phosphorylation site causes amelogenesis imperfecta. J Dent Res 89:695–699
Kobayashi K, Yamakoshi Y, Hu JC, Gomi K, Arai T, Fukae M, Krebsbach PH, Simmer JP (2007) Splicing determines the glycosylation state of ameloblastin. J Dent Res 86:962–967
Poulter JA, Murillo G, Brookes SJ, Smith CE, Parry DA, Silva S, Kirkham J, Inglehearn CF, Mighell AJ (2014) Deletion of ameloblastin exon 6 is associated with amelogenesis imperfecta. Hum Mol Genet 23:5317–5324
Prasad MK, Geoffroy V, Vicaire S, Jost B, Dumas M, Le Gras S, Switala M, Gasse B, Laugel-Haushalter V, Paschaki M, Leheup B, Droz D, Dalstein A, Loing A, Grollemund B, Muller-Bolla M, Lopez-Cazaux S, Minoux M, Jung S, Obry F, Vogt V, Davideau JL, Davit-Beal T, Kaiser AS, Moog U, Richard B, Morrier JJ, Duprez JP, Odent S, Bailleul-Forestier I, Rousset MM, Merametdijan L, Toutain A, Joseph C, Giuliano F, Dahlet JC, Courval A, El Alloussi M, Laouina S, Soskin S, Guffon N, Dieux A, Doray B, Feierabend S, Ginglinger E, Fournier B, de la Dure MM, Alembik Y, Tardieu C, Clauss F, Berdal A, Stoetzel C, Maniere MC, Dollfus H, Bloch-Zupan A (2016) A targeted next-generation sequencing assay for the molecular diagnosis of genetic disorders with orodental involvement. J Med Genet 53:98–110
Ma P, Yan W, Tian Y, He J, Brookes SJ, Wang X (2016) The Importance of Serine Phosphorylation of Ameloblastin on Enamel Formation. J Dent Res 95:1408–1414
Lamkin MS, Lindhe P (2001) Purification of kinase activity from primate parotid glands. J Dent Res 80:1890–1894
Nam Y, Madapallimattam G, Drzymala L, Bennick A (1997) Characterization of human sublingual-gland protein kinase by phosphorylation of a peptide related to secreted proteins. Arch Oral Biol 42:527–537
Madapallimattam G, Bennick A (1986) Phosphorylation of salivary proteins by salivary gland protein kinase. J Dent Res 65:405–411
Yang X, Yan W, Tian Y, Ma P, Opperman LA, Wang X (2016) Family with sequence similarity member 20C is the primary but not the only kinase for the small-integrin-binding ligand N-linked glycoproteins in bone. FASEB J 30:121–128
Acknowledgements
We thank Dr. Hidemitsu Harada (Iwate Medical University, Japan) for providing HAT-7 cells. We also thank for Dr. Sundharamani Venkitapathi for his assistance. This study was supported by grants from the National Institute of Dental and Craniofacial Research, NIH (DE019527 to YM.) and Boston University Henry Goldman School of Dental Medicine (YM.). NG and PP were supported by The Chulalongkorn Academic Advancement Into Its 2nd Century Project.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: NG, MK and YM. Performed the experiments: NG, MK, YO, HJ and IPL. Analyzed the data: NG, MK, PP, HM and YM. Wrote the paper: NG, MK, HM, PP and YM. Read and approved the final manuscript by all authors.
Corresponding authors
Ethics declarations
Conflict of interest
Nattanan Govitvattana, Masaru Kaku, Yoshio Ohyama, Haytham Jaha, I-Ping Lin, Hanna Mochida, Prasit Pavasant and Yoshiyuki Mochida have declares no competing financial interests.
Ethical approval
The use of animals and all animal procedures in this study were approved by the Institutional Animal Care and Use Committee (IACUC) of Boston University Medical campus (approved protocol number: AN-15053), and all efforts were made to minimize suffering animals. This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Accession Number
The nucleotide sequences for mouse Gm1045-a and Gm1045-b genes have been deposited in the GenBank database under GenBank Accession numbers; KF477194 and KC602377, respectively.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Govitvattana, N., Kaku, M., Ohyama, Y. et al. Molecular Cloning of Mouse Homologue of Enamel Protein C4orf26 and Its Phosphorylation by FAM20C. Calcif Tissue Int 109, 445–454 (2021). https://doi.org/10.1007/s00223-021-00847-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-021-00847-y