Abstract
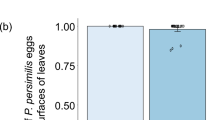
Environmental variables may markedly influence egg parasitoid performance and must be considered when choosing the best release strategy. Thus, this study aimed to investigate the effects of light, temperature, soil moisture, and precipitation on the emergence and parasitism of parasitoid releases of unprotected and encapsulated pupae. The presence of light favored the parasitism of Trichogramma pretiosum Riley, 1879, and Telenomus remus (Nixon, 1937) but did not impact the parasitism of Telenomus podisi (Ashmead, 1893). The release strategy adopted (release of encapsulated or unprotected pupae) also impacted the results. On one hand, card capsules gave protection against rainfall, limited to 10 mm regarding Tr. pretiosum, while on the other hand, card capsules led to a reduction in the number of captured adults for Te. podisi at 25°C and 30°C. Therefore, Tr. pretiosum, Te. remus, and Te. podisi can be released using both encapsulated and unprotected pupae, with advantages and disadvantages for each strategy, depending on each studied environmental variable. In addition, parasitoid pupae should be released so that the majority emerge during daylight, especially for Te. remus and Tr. pretiosum, since parasitism was greatly reduced in dark environments. Telenomus podisi is not affected by this variable as it demonstrated similar parasitism in light and dark environments.


Similar content being viewed by others
References
Aquilino KM, Cardinale BJ, Ives AR (2005) Reciprocal effects of host plant and natural enemy diversity on herbivore suppression: an empirical study of a model tritrophic system. Oikos 108:275–282. https://doi.org/10.1111/j.0030-1299.2005.13418.x
Bai B, Smith SM (1993) Effect of host availability on reproduction and survival of the parasitoid wasp Trichogramma minutum. Ecol Entomol 18(4):279–286. https://doi.org/10.1111/j.1365-2311.1993.tb01102.x
Botelho PSM (1997) Eficiência de Trichogramma em campo. In: Parra JRP, Zucchi RA (eds) Trichogrammae o controle biológico aplicado. FEALQ, Piracicaba, pp 303–318
Bourchier RS, Smith SM (1996) Influence of environmental conditions and parasitoid quality on field performance of Trichogramma minutum. Entomol Exp Appl 80(3):461–468. https://doi.org/10.1111/j.1570-7458.1996.tb00960.x
Braz EC, Bueno AF, Colombo F, Queiroz AP (2021) Temperature impact on Telenomus podisi emergence in field releases of unprotected and encapsulated parasitoid pupae Neotrop Entomol in press
Bueno AF, Batistela MJ, Bueno RCOF, França-Neto JB, Nishikawa MAN, Filho AL (2011) Effects of integrated pest management, biological control and prophylactic use of insecticides on the management and sustainability of soybean. Crop Prot 30:937–945. https://doi.org/10.1016/j.cropro.2011.02.021
Bueno RCOF, Parra JRP, Bueno AF (2012) Trichogramma pretiosum parasitism of Pseudoplusia includens and Anticarsia gemmatalis eggs at different temperatures. Biol Control 60(2):154–162. https://doi.org/10.1016/j.biocontrol.2011.11.005
Burr IW, Foster LA (1972) A test for equality of variances. University of Purdue, West Lafayette, p 26
Castellanos NL, Bueno AF, Haddi K (2019) The fitness and economic benefits of rearing the parasitoid Telenomus podisi under fluctuating temperature regime. Neotrop Entomol 48:934–948. https://doi.org/10.1007/s13744-019-00717-1
Cônsoli FL, Rossi MM, Parra JRP (1999) Developmental time and characteristics of the immature stages of Tichogramma galloi and Tr. pretiosum (Himenoptera, Trichogrammatidae). Rev Bras Entomol 43(3-4):271–275
Conte O, Oliveira FT, Harger N, Corrêa-Ferreira BS (2014) Resultados do Manejo Integrado de Pragas da Soja na safra 2013/14 no Paraná. Londrina, EMBRAPA-CNPSo, EMBRAPA-CNPSo, (Documentos 356), p 56
Dutton A, Bigler F (1995) Flight activity assessment of the egg parasitoid Trichogramma brassicae (Hym.: Trichogrammatidae) in laboratory and field conditions. Entomophaga 40:223–233. https://doi.org/10.1007/BF02373070
Godoy KB, Avila CJ, Duarte MM, Arce CCM (2010) Parasitismo e sítios de diapausa de adultos do percevejo marrom, Euschistus heros na região da Grande Dourados, MS. Cienc Rural 40(5):1199–1202. https://doi.org/10.1590/S0103-84782010005000074
Guedes JVC, Arnemann JÁ, Stürmer GR, Melo AA, Bigolin M, Perini CR, Sari BG (2012) Percevejos da soja: novos cenários, novo manejo. Revista Plantio Direto 12(1):24–30
Guillén L, Aluja M, Equihua M, Sivinski J (2002) Performance of two fruit fly (Diptera: Tephritidae) pupal parasitoids [Coptera haywardi (Hymenoptera: Diapriidae) and Pachycrepoideus vindemiae (Hymenoptera: Pteromalidae)] under different environmental soil conditions. Biol Control 23:219–227. https://doi.org/10.1006/bcon.2001.1011
Larios GLB, Ohno K, Fukuhara F (2007) Effects of photoperiod and temperature on preimaginal development and summer diapause of Chrysocharis pubicornis (Zetterstedt) (Hymenoptera: Eulophidae), a pupal parasitoid of leafminers (Diptera: Agromyzidae). Appl Entomol Zool 42(2):189–197
Lewis CN, Whitfield JB (1999) Braconid wasp (Hymenoptera: Braconidae) diversity in forest plots under different silvicultural methods. Environ Entomol 28:986–997. https://doi.org/10.1093/ee/28.6.986
Mcdougall SJ, Mills NJ (1997) The influence of hosts, temperature and food sources on the longevity of Trichogramma platneri. Entomol Exp Appl 83:195–203. https://doi.org/10.1046/j.1570-7458.1997.00172.x
Mélo-Filho LR, Guenther M (2015) A resistência sistêmica induzida como alternativa sustentável ao uso de agrotóxicos. Revista Agroambiente 8:27–38
Mills N, Pickel C, Mansfield S, McDougall S, Buchner R, Caprile-Emeritus J, Stocker R (2000) Mass releases of wasps can reduce damage from codling moth. Calif Agric 54(6):22–25. https://doi.org/10.3733/ca.v054n06p22
Molina RMS, Fronza V, Parra JRP (2005) Seleção de Trichogramma spp., para o controle de Ecdytolopha aurantiana, com base na biologia e exigências térmicas. Rev Bras Entomol 49(1):152–158. https://doi.org/10.1590/S0085-56262005000100018
Noldus LP (1989) Semiochemicals, foraging behaviour and quality of entomophagous insects for biological control. J Appl Entomol 108(1-5):425–451. https://doi.org/10.1111/j.1439-0418.1989.tb00478.x
Ozkan C (2007) Effect of food, light and host instar on the egg load of the synovigenic endoparasitoid Venturia canescens (Hymenoptera: Ichneumonidae). J Pest Sci 80:79–83. https://doi.org/10.1007/s10340-006-0155-4
Parra JRP (1997) Técnicas de criação de Anagasta kuehniella, hospedeiro alternativo para produção de Trichogramma. In: JRP P, Zucchi RA (eds) Trichogramma e o Controle Biológico Aplicado. FEALQ, Piracicaba, p 324
Parra JRP (2014) Biological control in Brazil: an overview. Sci Agric 71:345–355. https://doi.org/10.1590/0103-9016-2014-0167
Pereira FF, Barros R, Pratissoli D, Parra JRP (2004) Biologia e exigências térmicas de Trichogramma pretiosum Riley e T. exiguum Pinto & Planter (Hymenoptera: Trichogrammatidae) criados em ovos de Plutella xylostella (L.) (Lepidoptera: Plutellidae). Neotrop Entomol 33(2):231–236. https://doi.org/10.1590/S1519-566X2004000200014
Peres WAA, Corrêa-Ferreira BS (2004) Methodology of mass multiplication of Telenomus podisi Ash. and Trissolcus basalis (Woll.) (Hymenoptera: Scelionidae) on eggs of Euschistus heros (Fab.) (Hemiptera: Pentatomidae). Neotrop Entomol 33(4):457–462. https://doi.org/10.1590/S1519-566X2004000400010
Pinto AS, Parra JRP (2002) Liberações de inimigos naturais. In: Parra JRP, Botelho PSM, Côrrea-Ferreira BS, Bento JMS (eds) Controle biológico no Brasil: parasitoides e predadores. Manole, São Paulo, pp 325–342
Pinto AS, Parra JRP, Oliveira HN, Arrigoni EDB (2003) Comparação de técnicas de liberação de Trichogramma galloi Zucchi (Hymenoptera: Trichogrammatidae) para o controle de Diatraea saccharalis (Fabricius) (Lepidoptera: Crambidae). Neotrop Entomol 32(2):311–318. https://doi.org/10.1590/S1519-566X2003000200018
Pomari AF, Freitas AF, Bueno RCOF, Menezes Junior AO (2012) Biological characteristics and thermal requirements of the biological control agent Telenomus remus (Hymenoptera: Scelionidae) reared on eggs of different species of the genus Spodoptera (Lepidoptera: Noctuidae). Ann Entomol Soc Am 105(1):73–81. https://doi.org/10.1603/AN11115
Prezotti L, Parra JRP, Vencovsky R, Dias CTS, Cruz I, Chagas MCM (2002) Teste de voo como critério de avaliação da qualidade de Trichogramma pretiosumRiley (Hymenoptera: Trichogrammatidae): Adaptação de metodologia. Neotrop Entomol 31(3):411–417. https://doi.org/10.1590/S1519-566X2002000300010
Queiroz AP, Bueno AF, Pomari-Fernandes A, Bortolotto OC, Mikami AY, Olive L (2017a) Influence of host preference, mating, and release density on the parasitism of Telenomus remus (Nixon) (Hymenoptera, Scelionidae). Rev Bras Entomol 61(1):86–90. https://doi.org/10.1016/j.rbe.2016.12.004
Queiroz AP, Bueno AF, Pomari-Fernandes A, Grande MLM, Bortolotto OC, Silva DM (2017b) Low temperature storage of Telenomus remus (Nixon) (Hymenoptera: Platygastridae) and its factitious host Corcyra cephalonica (Stainton) (Lepidoptera: Pyralidae). Neotrop Entomol 46:182–192. https://doi.org/10.1007/s13744-016-0442-6
Querino RB, Zucchi RA (2016) Trichogramma na Amazônia - Visão geral e potencialidades. In: Silva NM et al (eds) Pragas agrícolas e florestais na Amazônia. Embrapa Amapá, Macapá, pp 597–606
Reichardt K (1985) Processos de transferência no sistema solo-planta-atmosfera. 4 ed. Fundação Cargill, Campinas, p 445
Roth CH, Meyer B, Frede HG (1985) A portable rainfall simulator for studying factors affecting runoff, infiltration and soil loss. Catena 12:79–85
Rousse P, Gourdon F, Roubaud M, Chiroleu F, Quilici S (2009) Biotic and abiotic factors affecting the flight activity of Fopius arisanus, an egg-pupal parasitoid of fruit fly pests. Environ Entomol 38(3):896–903. https://doi.org/10.1603/022.038.0344
Sagarra LA, Vincent C, Peters NF, Stewart RK (2000) Effect of host density, temperature, and photoperiod on the fitness of Anagyrus kamali, a parasitoid of the hibiscus mealybug Maconellicoccus hirsutus. Entomol Exp Appl 96(2):141–147. https://doi.org/10.1046/j.1570-7458.2000.00689.x
Santana DRS, Bellon PP, Melo E, Oliveira HN (2013) Influência do Fotoperíodo no Parasitismo de Trichogramma galloi Zucchi (Hymenoptera: Trichogrammatidae) em Ovos de Diatraea saccharalis (Fabricius) (Lepidoptera: Crambidae). EntomoBrasilis 6(2):165–167. https://doi.org/10.12741/ebrasilis.v6i2.263
SAS Institute (2001) SAS/STAT user’s guide: statistics, version 8.2.6. SAS Institute, Cary, NC, 201 pp
Schmitz OJ, Barton BT (2014) Climate change effects on behavioral and physiological ecology of predator–prey interactions: Implications for conservation biological control. Biol Control 75:87–96. https://doi.org/10.1016/j.biocontrol.2013.10.001
Shapiro SS, Wilk MB (1965) An analysys of variance test for normality (complete samples). Biometrika 52:591–611. https://doi.org/10.2307/2333709
Silva CSB (2007) Dispersão do parasitoide de ovos Telenomus remus Nixon (Hymenoptera: Scelionidae) e sua interação com algumas variáveis ambientais em agroecossistemas de algodoeiro (Gossypium hirsutum L.). 160f. 2007. Dissertation. Centro de Ciências Agrárias da Unesp/FCAV
Silva GV, Bueno AF, Neves PMOJ, Favetti BM (2018) Biological characteristics and parasitism capacity of Telenomus podisi (Hymenoptera: Platygastridae) on Eggs of Euschistus heros (Hemiptera: Pentatomidae). J Agric Sci 10:210–220. https://doi.org/10.5539/jas.v10n8p210
Smith SM (1996) Biological control with Trichogramma: advances, successes, and potencial of their use. Annu Rev Entomol 41:375–406. https://doi.org/10.1146/annurev.en.41.010196.002111
Souza DS, Lopes RM, Sarcinelli PN (2015) Educational intervention in exposure to pesticides: an integrative review. Revista Trabalho e Educação 24(2):247–265
Stein CP, Parra JRP (1987) Uso da radiação para inviabilizar ovos de Anagasta kuehniella (Zeller, 1879) visando estudos com Trichogramma spp. An Soc Entomol Bras 16:229–231
Tatsumi E, Takada H (2005) Effects of photoperiod and temperature on adult oligopause of Aphelinus asychis and larval diapause of A. albipodus (Hymenoptera: Aphelinidae). Appl Entomol Zool 40:447–456. https://doi.org/10.1303/aez.2005.447
Thomson LJ, Hoffmann AA (2002) Laboratory fecundity as predictor of field success in Trichogramma carverae (Hymenoptera: Trichogrammatidae). J Econ Entomol 95(5):912–917. https://doi.org/10.1603/0022-0493-95.5.912
Trussell GC, Ewanchuk PJ, Matassa CM (2008) Resource identity modifies the influence of predation risk on ecosystem function. Ecology 89:2798–2807. https://doi.org/10.1890/08-0250.1
Tunçbilek AS, Ayvaz A (2003) Influences of host age, sex ratio, population density, and photoperiod on parasitism by Trichogramma evanescens Westw. (Hym., Trichogrammatidae). AFS 76:176–180. https://doi.org/10.1007/s10340-003-0018-1
Valente ECN, Broglio SMF, Passos EM, Lima AST (2016) Desempenho de Trichogramma galloi sobre ovos de Diatraea spp. (Lepidoptera: Crambidae). Pesq Agropec Bras 51(4):293–300. https://doi.org/10.1590/S0100-204X2016000400001
van Lenteren JC, Bolckmans K, Köhl J, Ravensberg WJ, Urbaneja A (2018) Biological control using invertebrates and microorganisms: plenty of new opportunities. BioControl 63:39–59. https://doi.org/10.1007/s10526-017-9801-4
Wajnberg E, Hassan SA (1994) Biological control with egg parasitoids. British Library, Wallingford, p 286
Zart M, Berardi O, Nunes AM, da Silva Andersson F, Coimbra SM, Busato GR, Garcia MS (2012) Influência do fotoperíodo e da densidade de ovos de Anagasta kuenhiella (Zeller) sobre aspectos biológicos e parasitismo de ovos por Trichogramma pretiosum Riley. EntomoBrasilis 5:115–119. https://doi.org/10.12741/ebrasilis.v5i2.197
Acknowledgements
Thanks are extended to Dr Valmir Antônio Costa (Instituto Biológico de Campinas) for parasitoid identification and to Adair Vicente Carneiro for drawing Figs. 1 and 2. Thanks are also extended to Dra Graziela Moraes de Cesare Barbosa (IDR/Paraná) for allowing the use of the rain simulator in this research.
Funding
The authors thank Embrapa Soja, the Coordination for the Improvement of Higher Education Personnel (CAPES), and the National Council for Scientific and Technological Development (CNPq) (grants 402797/2016-7 and 302645/2018-7) for their financial support and fellowships.
Author information
Authors and Affiliations
Contributions
MLM Grande, A F. Bueno, and MU Ventura contributed to the study conception and design. AP de Queiroz, J Gonçalves, and R Hayashida contributed to the data collection and analysis. MLM Grande wrote the first draft of the manuscript, and all authors commented on previous versions of the manuscript.
Corresponding author
Additional information
Edited by Anne-Nathalie Volkoff
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Grande, M.L.M., de Queiroz, A.P., Gonçalves, J. et al. Impact of Environmental Variables on Parasitism and Emergence of Trichogramma pretiosum, Telenomus remus and Telenomus podisi. Neotrop Entomol 50, 605–614 (2021). https://doi.org/10.1007/s13744-021-00874-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-021-00874-2