Abstract
Purpose
To investigate whether abnormal retinal microcirculation correlates with retinal neuronal changes in untreated diabetic eyes without macular edema.
Methods
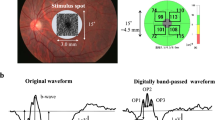
This study enrolled 29 diabetic patients without diabetic retinopathy (DR), 18 patients with mild non-proliferative diabetic retinopathy (NPDR), 15 patients with moderate NPDR, 14 patients with severe NPDR, 27 patients with proliferative diabetic retinopathy (PDR), and 25 healthy control subjects. Pattern electroretinography (PERG) and optical coherence tomography angiography (OCT-A) tests were performed.
Results
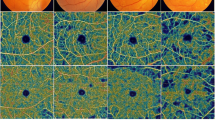
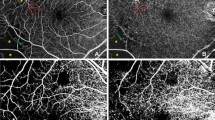
Differences in the mean values for the area, acircularity index, and perimeter of foveal avascular zone were statistically significant between the healthy control group and the diabetic patients (P < 0.05 for all). P50 and N95 amplitudes were statistically significantly lower in the PDR group compared to diabetic patients without DR, control, and moderate NPDR groups (P < 0.05 for all). The whole retina vessel densities in superficial and deep capillary plexus were lower in the PDR group compared to the diabetic patients without DR and control group (P < 0.05 for all). There were statistically significant positive correlations between the amplitudes of the P50 and N95 waves with the vessel densities.
Conclusion
The existence of significant correlations between PERG and OCT-A parameters in diabetic patients has shown that vascular and neuronal changes in the macula affect each other in diabetic patients.


Similar content being viewed by others
References
Fong DS, Aiello LP, Ferris FL 3rd, Klein R (2004) Diabetic retinopathy. Diabetes Care 27:2540–2553
Ling R, Ramsewak V, Taylor D, Jacob J (2002) Longitudinal study of a cohort of people with diabetes screened by the Exeter Diabetic Retinopathy Screening Programme. Eye (Lond) 16:140–145
Lutty GA (2013) Effects of diabetes on the eye. Invest Ophthalmol Vis Sci 54:81–87
Chhablani J, Sharma A, Goud A, Peguda HK, Rao HL, Begum VU, Barteselli G (2015) Neurodegeneration in type 2 diabetes: evidence from spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci 56:6333–6338
Sohn EH, van Dijk HW, Jiao C, Kok PH, Jeong W, Demirkaya N, Garmager A, Wit F, Kucukevcilioglu M, van Velthoven ME, DeVries JH, Mullins RF, Kuehn MH, Schlingemann RO, Sonka M, Verbraak FD, Abràmoff MD (2016) Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc Natl Acad Sci USA 113:E2655-2664
El-Fayoumi D, Badr Eldine NM, Esmael AF, Ghalwash D, Soliman HM (2016) Retinal nerve fiber layer and ganglion cell complex thicknesses are reduced in children with type 1 diabetes with no evidence of vascular retinopathy. Invest Ophthalmol Vis Sci 57:5355–5360
Shoji T, Sakurai Y, Sato H, Chihara E, Takeuchi M (2011) Do type 2 diabetes patients without diabetic retinopathy or subjects with impaired fasting glucose have impaired colour vision? The Okubo Color Study Report. Diabet Med 28:865–871
Sokol S, Moskowitz A, Skarf B, Evans R, Molitch M, Senior B (1985) Contrastsensitivity in diabetics with and without backgroundretinopathy. Arch Ophthalmol 103:51–54
Adhikari P, Marasini S, Sah RP, Joshi SN, Shrestha JK (2014) Multifocal electroretinogram responses in Nepalese diabetic patients without retinopathy. Doc Ophthalmol 129:39–46
Mermeklieva EA (2019) Pattern electroretinography and retinal changes in patients with diabetes mellitus type 2. Neurophysiol Clin 49:209–215
Prager TC, Garcia CA, Mincher CA, Mishra J, Chu HH (1990) The pattern electroretinogram in diabetes. Am J Ophthalmol 109:279–284
Shin MK, Kim SI, Park SW, Byon IS, Kim HW, Lee JE (2015) Evaluation of macular function using pattern electroretinogram in idiopathic epiretinal membrane. Asia Pac J Ophthalmol (Phila) 4:267–272
Spaide RF, Fujimoto JG, Waheed NK, Sadda SR, Staurenghi G (2018) Optical coherence tomography angiography. Prog Retin Eye Res 64:1–55
de Carlo TE, Chin AT, Bonini Filho MA, Adhi M, Branchini L, Salz DA, Baumal CR, Crawford C, Reichel E, Witkin AJ, Duker JS, Waheed NK (2015) Detection of microvascular changes in eyes of patients with diabetes but not clinical diabetic retinopathy using optical coherence tomography angiography. Retina 35:2364–2370
Li L, Almansoob S, Zhang P, Zhou YD, Tan Y, Gao L (2019) Quantitative analysis of retinal and choroid capillary ischaemia using optical coherence tomography angiography in type 2 diabetes. Acta Ophthalmol 97:240–246
Dimitrova G, ChiharaE TH, Amano H, Okazaki K (2017) Quantitative retinal optical coherence tomography angiography in patients with diabetes without diabetic retinopathy. Invest Ophthalmol Vis Sci 58:190–196
Early Treatment Diabetic Retinopathy Study Research Group (1991) Grading diabetic retinopathy from stereoscopic color fundus photographs-an extension of the modified Airlie House classification. ETDRS report number 10. Ophthalmology 98:786–806
Bach M, Brigell MG, Hawlina M, Holder GE, Johnson MA, McCulloch DL, Meigen T, Viswanathan S (2013) ISCEV standard for clinical pattern electroretinography (PERG): 2012 update. Doc Ophthalmol 126:1–7
American Clinical Neurophysiology Society (2006) Guideline 5: guidelines for standard electrode position nomenclature. J Clin Neurophysiol 23:107–110
Tam J, Dhamdhere KP, Tiruveedhula P, Manzanera S, Barez S, Bearse MA Jr, Adams AJ, Roorda A (2011) Disruption of the retinal parafoveal capillary network in type 2 diabetes before the onset of diabetic retinopathy. Invest Ophthalmol Vis Sci 52:9257–9266
Mo S, Krawitz B, Efstathiadis E, Geyman L, Weitz R, Chui TY, Carroll J, Dubra A, Rosen RB (2016) Imaging foveal microvasculature: optical coherence tomography angiography versus adaptive optics scanning light ophthalmoscope fluorescein angiography. Invest Ophthalmol Vis Sci 57:130–140
Kern TS, Barber AJ (2008) Retinal ganglion cells in diabetes. J Physiol 586:4401–4408
Barber AJ, Lieth E, Khin SA, Antonetti DA, Buchanan AG, Gardner TW (1998) Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest 102:783–791
Lorenzi M, Gerhardinger C (2001) Early cellular and molecular changes induced by diabetes in the retina. Diabetologia 44:791–804
Lieth E, Gardner TW, Barber AJ, Antonetti DA; Penn State Retina Research Group (2000) Retinal neurodegeneration: early pathology in diabetes. Clin Exper Ophthalmol 28:3–8
Stem MS, Gardner TW (2013) Neurodegeneration in the pathogenesis of diabetic retinopathy: molecular mechanisms and therapeutic implications. Curr Med Chem 20:3241–3250
Roy S, Trudeau K, Roy S, Tien T, Barrette KF (2013) Mitochondrial dysfunction and endoplasmic reticulum stress in diabetic retinopathy: mechanistic insights into high glucose-induced retinal cell death. Curr Clin Pharmacol 8:278–284
Rosa MD, Distefano G, Gagliano C, Rusciano D, Malaguarnera L (2016) Autophagy in diabetic retinopathy. Curr Neuropharmacol 14:810–825
Kowluru RA, Mishra M (2015) Oxidative stress, mitochondrial damage and diabetic retinopathy. Biochim Biophys Acta 1852:2474–2483
Fernyhough P, McGavock J (2014) Mechanisms of disease: Mitochondrial dysfunction in sensory neuropathy and other complications in diabetes. Handb Clin Neurol 126:353–377
Bek T (2017) Mitochondrial dysfunction and diabetic retinopathy. Mitochondrion 36:4–6
Barber AJ, Baccouche B (2017) Neurodegeneration in diabetic retinopathy: potential for novel therapies. Vision Res 139:82–92
Van Dijk HW, Verbraak FD, Stehouwer M, Kok PH, Garvin MK, Sonka M, DeVries JH, Schlingemann RO, Abràmoff MD (2011) Association of visualfunction and ganglion cell layer thickness in patients with diabetes mellitus type 1 and no or minimal diabetic retinopathy. Vis Res 51:224–228
Rossino MG, Dal Monte M, Casini G (2019) Relationships between neurodegeneration and vascular damage in diabetic retinopathy. Front Neurosci 13:1172
Saint-Geniez M, Maharaj AS, Walshe TE, Tucker BA, Sekiyama E, Kurihara T, Darland DC, Young MJ, D’Amore PA (2008) Endogenous VEGF is required for visual function: evidence for a survival role on müller cells and photoreceptors. PLoS ONE 3:e3554
Romano MR, Biagioni F, Besozzi G, Carrizzo A, Vecchione C, Fornai F, Lograno MD (2012) Effects of bevacizumab on neuronal viability of retinal ganglion cells in rats. Brain Res 1478:55–63
Beazley-Long N, Hua J, Jehle T, Hulse RP, Dersch R, Lehrling C, Bevan H, Qiu Y, Lagrèze WA, Wynick D, Churchill AJ, Kehoe P, Harper SJ, Bates DO, Donaldson LF (2013) VEGF-A165b is an endogenous neuroprotective splice isoform of vascular endothelial growth factor A in vivo and in vitro. Am J Pathol 183:918–929
Amato R, Rossino MG, Cammalleri M, Locri F, Pucci L, Dal Monte M, Casini G (2018) Lisosan G protects the retina from neurovascular damage in experimental diabetic retinopathy. Nutrients 10:1932
Chen Y, Meng J, Li H, Wei H, Bi F, Liu S, Tang K, Guo H, Liu W (2019) Resveratrol exhibits an effect on attenuating retina inflammatory condition and damage of diabetic retinopathy via PON1. Exp Eye Res 181:356–366
Orhan C, Akdemir F, Tuzcu M, Sahin N, Yilmaz I, Deshpande J, Juturu V, Sahin K (2016) Mesozeaxanthin protects retina from oxidative stress in a rat model. J Ocul Pharmacol Ther 32:631–637
He M, Long P, Yan W, Chen T, Guo L, Zhang Z, Wang S (2018) ALDH2 attenuates early-stage STZ-induced aged diabetic rats retinas damage via Sirt1/Nrf2 pathway. Life Sci 215:227–235
Liu Q, Zhang X, Cheng R, Ma JX, Yi J, Li J (2019) Salutary effect of fenofibrate on type 1 diabetic retinopathy via inhibiting oxidative stress-mediated Wnt/β-catenin pathway activation. Cell Tissue Res 376:165–177
Ozkiris A (2010) Pattern electroretinogram changes after intravitreal bevacizumab injection for diabetic macular edema. Doc Ophthalmol 120:243–250
Ozkiris A, Evereklioglu C, Oner A, Erkiliç K (2004) Pattern electroretinogram for monitoring the efficacy of intravitreal triamcinolone injection in diabetic macular edema. Doc Ophthalmol 109:139–145
Carnevali A, Sacconi R, Corbelli E, Tomasso L, Querques L, Zerbini G, Scorcia V, Bandello F, Querques G (2017) Optical coherence tomography angiography analysis of retinal vascular plexuses and choriocapillaris in patients with type 1 diabetes without diabetic retinopathy. Acta Diabetol 54:695–702
Tan B, Chua J, Lin E, Cheng J, Gan A, Yao X, Wong DWK, Sabanayagam C, Wong D, Chan CM, Wong TY, Schmetterer L, Tan GS (2020) Quantitative microvascular analysis with wide-field optical coherence tomography angiography in eyes with diabetic retinopathy. JAMA Netw Open 3:e1919469
Conti FF, Song W, Rodrigues EB, Singh RP (2019) Changes in retinal and choriocapillaris density in diabetic patients receiving anti-vascular endothelial growth factor treatment using optical coherence tomography angiography. Int J Retina Vitreous 5:41
Dastiridou A, Karathanou K, Riga P, Anagnostopoulou S, Balasubramanian S, Mataftsi A, Brazitikos P, Ziakas N, Androudi S (2020) OCT angiography study of the macula in patients with diabetic macular edema treated with intravitreal aflibercept. OculImmunol Inflamm. https://doi.org/10.1080/09273948.2019.1704028
Vujosevic S, Muraca A, Gatti V, Masoero L, Brambilla M, Cannillo B, Villani E, Nucci P, De Cillà S (2018) Peripapillary microvascular and neural changes in diabetes mellitus: an OCT-angiography study. Invest Ophthalmol Vis Sci 59:5074–5081
Acknowledgements
A part from the authors, no individuals or organizations have made substantial contributions to the study.
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Ankara Numune Training and Research Hospital and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Koçer, A.M., Şekeroğlu, M.A. Evaluation of the neuronal and microvascular components of the macula in patients with diabetic retinopathy. Doc Ophthalmol 143, 193–205 (2021). https://doi.org/10.1007/s10633-021-09834-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-021-09834-y