Abstract
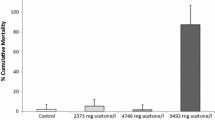
Behavioral endpoints are important parameters to assess the effects of toxicants and other stressors on natural ecosystems. The relevance of these parameters has caused a rise in their use in aquatic ecotoxicology. However, abiotic and biotic parameters may interact causing changes in the behavioral responses. Among those parameters, starvation of animals is a factor that is usually applied in ecotoxicological short-term bioassays. This could alter animal behavior, along with the toxicant. Therefore, the study of the effects of starvation on baseline behaviors of invertebrates is a relevant issue. This study assessed the behavior of the aquatic snail Potamopyrgus antipodarum under a combination of four treatments: (1) animals normally fed (control treatment), (2) starved animals, (3) animals normally fed and exposed to a high conductivity, and (4) starved animals exposed to a high conductivity. The behavior activity of snails was monitored for 14 days. Results show that animals of the second treatment (starved animals) increased their activity. On the contrary, animals of the third and fourth treatments reduced their activity. Animals from the control treatment showed an activity in between starved animals and animals exposed to high conductivity (both starved and normally fed). These results show that starvation increases the snail activity, but under another environmental stressor (i.e., high conductivity), this trend was reversed. The influence of starvation on behavior should be taken into account in the development of behavioral bioassays.


Similar content being viewed by others
References
Alonso, A., & Camargo, J. A. (2009). Long-term effects of ammonia on the behavioral activity of the aquatic snail Potamopyrgus antipodarum (Hydrobiidae, Mollusca). Archives of Environmental Contamination and Toxicology, 56, 796–802.
Alonso, A., & Camargo, J. A. (2013). Nitrate causes deleterious effects on the behaviour and reproduction of the aquatic snail Potamopyrgus antipodarum (Hydrobiidae, Mollusca). Environmental Science and Pollution Research, 20, 5388–5396. https://doi.org/10.1007/s11356-013-1544-x.
Alonso, A., & Valle-Torres, G. (2018). Feeding behavior of an aquatic snail as a simple endpoint to assess the exposure to cadmium. Bulletin of Environmental Contamination and Toxicology, 100, 82–88. https://doi.org/10.1007/s00128-017-2230-3.
Alonso, A., De Lange, H. J., & Peeters, E. T. H. M. (2009). Development of a feeding behavioural bioassay using the freshwater amphipod Gammarus pulex and the multispecies freshwater biomonitor. Chemosphere, 75, 341–346. https://doi.org/10.1016/j.chemosphere.2008.12.031.
Alonso, A., Garcia-Johansson, V., De Lange, H. J., & Peeters, E. T. H. M. (2010). Effects of animal starvation on the sensitivity of the freshwater amphipod Gammarus pulex to cadmium. Chemistry and Ecology, 26, 233–242. https://doi.org/10.1080/02757541003785866.
Alonso, A., Garcia-Perinan, E., & Camargo, J. A. (2016). Development of a low-cost ecotoxicological bioassay based on the feeding behaviour of the aquatic snail Potamopyrgus antipodarum (Hydrobiidae, Mollusca). Archives of Environmental Contamination and Toxicology, 71, 553–560. https://doi.org/10.1007/s00244-016-0316-2.
Bhuyan, K., & Anirudha, G. (2020). Changes in behavior, cardiac rate and acetylcholinesterase activity in tadpoles of Fejervarya limnocharis (Anura, Dicroglossidae) in response to sub-lethal exposure to fluoranthene. Research journal of biotechnology, 15, 91–98.
Bignami, S., Sponaugle, S., Hauff, M., & Cowen, R. K. (2017). Combined effects of elevated pCO2, temperature, and starvation stress on larvae of a large tropical marine fish. ICES Journal of Marine Science, 74, 1220–1229. https://doi.org/10.1093/icesjms/fsw216.
Chiu, J. M. Y., Ng, T. Y. T., Wang, W. X., Thiyagarajan, V., & Qian, P. Y. (2007). Latent effects of larval food limitation on filtration rate, carbon assimilation and growth in juvenile gastropod Crepidula onyx. Marine Ecology Progress Series, 343, 173–182.
Chiu, J. M. Y., Wang, H., Thiyagarajan, V., & Qian, P. Y. (2008). Differential timing of larval starvation effects on filtration rate and growth in juvenile Crepidula onyx. Marine Biology, 154, 91–98. https://doi.org/10.1007/s00227-007-0902-y.
Cloyed, C. S., & Dell, A. I. (2019). Resource distribution and internal factors interact to govern movement of a freshwater snail. Proceedings of the Royal Society B, 286, 20191610. https://doi.org/10.1098/rspb.2019.1610.
Core Team, R. (2019). R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing https://www.R-project.org/.
Curtis, D. L., & McGaw, I. J. (2012). Salinity and thermal preference of Dungeness crabs in the lab and in the field: Effects of food availability and starvation. Journal of Experimental Marine Biology and Ecology, 413, 113–120. https://doi.org/10.1016/j.jembe.2011.12.005.
Dell’Omo, G. (Ed.). (2002). Behavioural ecotoxicology. West Sussex: Wiley.
Denoel, M., Libon, S., Kestemont, P., Brasseur, C., Focant, J., & De Pauw, E. (2013). Effects of a sublethal pesticide exposure on locomotor behavior: A video-tracking analysis in larval amphibians. Chemosphere, 90, 945–951. https://doi.org/10.1016/j.chemosphere.2012.06.037.
Duft, M., Schulte-Oehlmann, U., Tillmann, M., Markert, B., & Oehlmann, J. (2003). Toxicity of triphenyltin and tributyltin to the freshwater mudsnail Potamopyrgus antipodarum in a new sediment biotest. Environmental Toxicology and Chemistry, 22, 145–152.
Faimali, M., Gambardella, C., Costa, E., Piazza, V., Morgana, S., Estevez-Calvar, N., & Garaventa, F. (2017). Old model organisms and new behavioral end-points: Swimming alteration as an ecotoxicological response. Marine Environmental Research, 128, 36–45. https://doi.org/10.1016/j.marenvres.2016.05.006.
Fan, X., Hou, T., Sun, T., Zhu, L., Zhang, S., Tang, K., & Wang, Z. (2019). Starvation stress affects the maternal development and larval fitness in zebrafish (Danio rerio). Science of the Total Environment, 695, 1–13. https://doi.org/10.1016/j.scitotenv.2019.133897.
Feiner, M., Laforsch, C., Letzel, T., & Geist, J. (2014). Sublethal effects of the beta-blocker sotalol at environmentally relevant concentrations on the New Zealand Mudsnail Potamopyrgus antipodarum. Environmental Toxicology and Chemistry, 33, 2510–2515. https://doi.org/10.1002/etc.2699.
Figueroa-Sánchez, M. A., Nandini, S., Castellanos-Páez, M. E., & Sarma, S. S. S. (2019). Effect of temperature, food quality and quantity on the feeding behavior of Simocephalus mixtus and Hyalella azteca: implications for biomanipulation. Wetlands Ecology and Management, 27, 353–361. https://doi.org/10.1007/s11273-019-09664-5.
Freitas, J. S., Girotto, L., Goulart, B. V., Alho, G., de Oliveira, L., Gebara, R. C., Montagner, C. C., Schiesari, L., & Gaeta Espindola, E. L. (2019). Effects of 2,4-D-based herbicide (DMA (R) 806) on sensitivity, respiration rates, energy reserves and behavior of tadpoles. Ecotoxicology and Environmental Safety, 182(UNSP), 109446. https://doi.org/10.1016/j.ecoenv.2019.109446.
French, C. G., & Wahl, D. H. (2018). Influences of dissolved oxygen on juvenile largemouth bass foraging behaviour. Ecology of Freshwater Fish, 27, 559–569. https://doi.org/10.1111/eff.12370.
Gerhardt, A., Schmidt, S., & Höss, S. (2002). Measurement of movement patterns of Caenorhabditis elegans (Nematoda) with the multispecies freshwater biomonitor (MFB)—A potential new method to study a behavioral toxicity parameter of nematodes in sediments. Environmental Pollution, 120, 513–516.
Hansen, M. J., Schaerf, T. M., & Ward, A. J. W. (2015). The effect of hunger on the exploratory behaviour of shoals of mosquitofish Gambusia holbrooki. Behaviour, 152, 1657–1683. https://doi.org/10.1163/1568539X-00003298.
Hellou, J. (2011). Behavioural ecotoxicology, an “early warning” signal to assess environmental quality. Environmental Science and Pollution Research, 18, 1–11. https://doi.org/10.1007/s11356-010-0367-2.
Hervant, F., Mathieu, J., Barré, H., Simon, K., & Pino, C. (1997). Comparative study on the behavioral, ventilatory, and respiratory responses of hypogean and epigean crustaceans to long-term starvation and subsequent feeding. Comparative Biochemistry and Physiology - Part A, 118, 1277–1283.
Heugens, E., Hendriks, A. J., Dekker, T., van Straalen, N. M., & Admiraal, W. (2001). A review of the effects of multiple stressors on aquatic organisms and analysis of uncertainty factors for use in risk assessment. Critical Reviews in Toxicology, 31, 247–284. https://doi.org/10.1080/20014091111695.
Heye, K., Graumnitz, S., Rybicki, M., Schuer, C., Voelker, J., Wick, A., Oehlmann, J., Jungmann, D., & Oetken, M. (2019). Laboratory-to-field extrapolation: Increase in carbamazepine toxicity in a higher tier, multiple-stress experiment. Ecotoxicology and Environmental Safety 183:UNSP 109481. https://doi.org/10.1016/j.ecoenv.2019.109481.
Hong, X., & Zha, J. (2019). Fish behavior: A promising model for aquatic toxicology research. Science of the Total Environment, 686, 311–321. https://doi.org/10.1016/j.scitotenv.2019.06.028.
Janssens de Bisthoven, L., Gerhardt, A., et al. (2006). Behavioral changes and acute toxicity to the freshwater shrimp Atyaephyra desmaresti Millet (Decapoda: Natantia) from exposure to acid mine drainage. Ecotoxicology, 15, 215–227.
Kirkpatrick, A. J., Gerhardt, A., Dick, J. T. A., Laming, P., & Berges, J. A. (2006). Suitability of Crangonyx pseudogracilis (Crustacea: Amphipoda) as an early warning indicator in the multispecies freshwater biomonitor. Environmental Science and Pollution Research, 13, 242–250.
Lecomte, V., Noury, P., Tutundjian, R., Buronfosse, T., Garric, J., & Gust, M. (2013). Organic solvents impair life-traits and biomarkers in the New Zealand mudsnail Potamopyrgus antipodarum (Gray) at concentrations below OECD recommendations. Aquatic Toxicology, 140, 196–203. https://doi.org/10.1016/j.aquatox.2013.06.006.
Little, E. E., & Finger, S. E. (1990). Swimming behavior as an indicator of sublethal toxicity in fish. Environmental Toxicology and Chemistry, 9, 13–19. https://doi.org/10.1002/etc.5620090103.
Mahabir, S., & Gerlai, R. (2017). The importance of holding water: Salinity and chemosensory cues affect Zebrafish behavior. Zebrafish, 14, 444–458. https://doi.org/10.1089/zeb.2017.1472.
Melvin, S. D., & Wilson, S. P. (2013). The utility of behavioral studies for aquatic toxicology testing: A meta-analysis. Chemosphere, 93, 2217–2223. https://doi.org/10.1016/j.chemosphere.2013.07.036.
Miyazaki, T., Masuda, R., Furuta, S., & Tsukamoto, K. (2000). Feeding behaviour of hatchery-reared juveniles of the Japanese flounder following a period of starvation. Aquaculture, 190, 129–138. https://doi.org/10.1016/S0044-8486(00)00385-9.
Naslund, J., & Johnsson, J. I. (2016). State-dependent behavior and alternative behavioral strategies in brown trout (Salmo trutta L.) fry. Behavioral Ecology and Sociobiology, 70, 2111–2125. https://doi.org/10.1007/s00265-016-2215-y.
Nielsen, M. E., & Roslev, P. (2018). Behavioral responses and starvation survival of Daphnia magna exposed to fluoxetine and propranolol. Chemosphere, 211, 978–985.
Nørum, U., Frederiksen, M. A. T., & Bjerregaard, P. (2011). Locomotory behaviour in the freshwater amphipod Gammarus pulex exposed to the pyrethroid cypermethrin. Chemistry and Ecology, 27, 569–577.
OECD (2011) Test No. 235. Chironomus sp., Acute immobilisation test. OECD Guidelines for the Testing of Chemicals, Paris.
OECD (2016) Test No. 242: Potamopyrgus antipodarum reproduction test, OECD Guidelines for the Testing of Chemicals, Section 2, OECD Publishing, Paris, https://doi.org/10.1787/9789264264311-en.
OECD (2019) Test No. 203. Fish, acute toxicity testing. OECD Guidelines for the Testing of Chemicals, Paris.
Peeters, E. T. H. M., de Lange, H. J., & Lürling, M. (2009). Variation in the behavior of the amphipod Gammarus pulex. Human and Ecological Risk Assessment, 15, 41–52.
Roozen, F., & Lurling, M. (2001). Behavioural response of Daphnia to olfactory cues from food, competitors and predators. Journal of Plankton Research, 23, 797–808. https://doi.org/10.1093/plankt/23.8.797.
Ruchin, A. B. (2019). Effect of illumination on growth and behavior of two carp fish species (Carassius gibelio and C. carassius). Periodico Tche Quimica, 16, 8–17.
Ruppert, K., Geiss, C., Ostermann, S., Theis, C., & Oehlmann, J. (2016). Comparative sensitivity of juvenile and adult Potamopyrgus antipodarum (Mollusca: Hydrobiidae) under chronic exposure to cadmium and tributyltin. Journal of Environmental Science and Health, Part A, 51, 736–743. https://doi.org/10.1080/10934529.2016.1170443.
Schmitt, C., Vogt, C., Machala, M., & de Deckere, E. (2011). Sediment contact test with Potamopyrgus antipodarum in effect-directed analyses-challenges and opportunities. Environmental Science and Pollution Research, 18, 1398–1404. https://doi.org/10.1007/s11356-011-0497-1.
Snider, S. B., & Gilliam, J. F. (2008). Movement ecology: Size-specific behavioral response of an invasive snail to food availability. Ecology, 89, 1961–1971. https://doi.org/10.1890/07-0715.1.
Suski, C. D., Philipp, M. A., & Hasler, C. T. (2019). Influence of nutritional status on carbon dioxide tolerance and avoidance behavior in a freshwater teleost. Transactions of the American Fisheries Society, 148, 914–925. https://doi.org/10.1002/tafs.10179.
USEPA (2002a) Short-term methods for estimating the chronic toxicity of effluents and receiving waters to freshwater organisms. Environmental Protection Agency, 821-R-02–013. 4th edn, USEPA, Washington, DC
USEPA (2002b) Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms. Environmental Protection Agency, 821-R-02–012. 1st ed, USEPA, Washington, DC
Veltman, K., Huijbregts, M. A. J., van Kolck, M., Wang, W.-X., & Hendriks, A. J. (2008). Metal bioaccumulation in aquatic species: quantification of uptake and elimination rate constants using physicochemical properties of metals and physiological characteristics of species. Environmental Science & Technology, 42, 852–858.
Wyeth RC (2019) Olfactory navigation in aquatic gastropods. Journal of Experimental Biology 222:UNSP doi: https://doi.org/10.1242/jeb.185843.
Acknowledgements
Many thanks to Pilar Castro for the review of English usage.
Availability of Data and Material
All data are available from the corresponding author on request.
Code Availability
Rcode is available from the corresponding author on request.
Funding
Funds for this research came from the University of Alcalá (research projects CCG2013/EXP-054, CCG2016/EXP-054, and CCG2018/EXP-074), and the projects CGL2011˗16388/BOS, CGL2015-65346R, INTERTOX RTI2018-096046-B-C21 (MCIU/AEI/FEDER, UE), and EXARBIN RTI2018-093504-B-I00 (MCIU/AEI/FEDER, UE) of the Ministerio de Economía y Competitividad of Spain and POII10˗0179˗4700 of the Junta de Comunidades de Castilla˗La Mancha.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
The author consents to participate in the publication.
Consent for Publication
The author provides consent for publication.
Conflict of Interest
The author declares no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alonso, Á. To Eat or Not to Eat: the Importance of Starvation on Behavioral Bioassays. Water Air Soil Pollut 232, 153 (2021). https://doi.org/10.1007/s11270-021-05111-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05111-5