Abstract
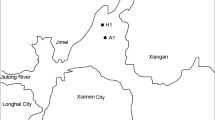
Dinoflagellate blooms currently caused serious environmental problems in different areas of the world. Recent studies revealed close relationship between dinoflagellate blooms and microbial community dynamics, while less attention has been paid on the bacterial culturability change associated with the bloom. Here, we investigated the temporal variation of microbial community composition and culturability during the decline stage of an Akashiwo sanguinea bloom occurred in Shenzhen, China. The daily microbial community phylogenetic structures in water samples collected during a four-day period after the bloom peak were assessed through 16S rRNA gene amplicons sequencing on the MiSeq (Illumina) platform. The environmental parameters, Chlorophyll a concentrations, and total viable and culturable bacterial densities were also measured. Our results showed that Gamma-proteobacteria comprising mostly of Pseudoalteromonadaceae and Vibrionaceae was the predominant microbial class in the post-bloom samples, except for the second day. During that day, the represented groups switched to Alpha-proteobacteria (Rhizobiales) and Beta-proteobacteria (Comamonadaceae), with the microbial culturability decreased. Total viable bacterial densities reached the maximum value on the third day, with Gamma-proteobacteria regained the dominance till the fourth day. The dramatic microbial community succession and culturability variation observed in this study indicated the complication of algae-bacteria interactions during dinoflagellate bloom.







Similar content being viewed by others
References
Badylak S, Philps EJ, Mathews AL (2014) Akashiwo sanguinea (Dinophyceae) blooms in a sub-tropical estuary: an alga for all seasons. Plankton and Benthos. Research 9(3):147–155
Bergmann M, Peters RH (1980) A simple reflectance method for the measurement of particulate pigment in lake water and its application to Phosphorus-Chlorophyll-Seston Relationships. Can J Fish Aquat Sci 37:111–114
Buchan A, LeCleir GR, Gulvik CA, Gonzalez JM (2014) Master recyclers: features and functions of bacteria associated with phytoplankton blooms. Nat Rev Micro 12(10):686–698
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M, Gormley N, Gilbert JA, Smith G, Knight R (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 6:1621–1624
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Lozupone CA, Turnbaugh PJ, Fierer N, Knight R (2011) Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc Natl Acad Sci 108:4516–4522
Chen H, Fu L, Luo L, Lu J, Lindsey WW, Hu Z (2012) Induction and resuscitation of the viable but nonculturable state in a cyanobacteria-lysing bacterium isolated from cyanobacterial bloom. Microb Ecol 63(1):64–73
Chen H, Shen J, Pan G, Liu J, Li J, Hu Z (2015) Correlations between cyanobacterial density and bacterial transformation to the viable but nonculturable (VBNC) state in four freshwater water bodies. Ecotoxicology 24(7-8):1459–1466
Dong K, Pan H, Yang D, Rao L, Zhao L, Wang Y, Liao X (2020) Induction, detection, formation, and resuscitation of viable but non-culturable state microorganisms. Compr Rev Food Sci Food Saf 19(1):149–183
Du X, Peterson W, McCulloch A, Liu G (2011) An unusual bloom of the dinoflagellate Akashiwo sanguinea off the central Oregon, USA, coast in autumn 2009. Harmful Algae 10(6):784–793
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Evans JC, Prepas EE (1997) Relative importance of iron and molybdenum in restricting phytoplankton biomass in high phosphorus saline lakes. Limnol Oceanogr 42(3):461–472
Fandino LB, Riemann L, Steward GF, Long RA, Azam F (2001) Variations in bacterial community structure during a dinoflagellate bloom analyzed by DGGE and 16S rDNA sequencing. Aquat Microb Ecol 23(2):119–130
Fuhrman JA, Cram JA, Needham DM (2015) Marine microbial community dynamics and their ecological interpretation. Nat Rev Micro 13(3):133–146
Green DH, Llewellyn LE, Negri AP, Blackburn SI, Bolch CJS (2004) Phylogenetic and functional diversity of the cultivable bacterial community associated with the paralytic shellfish poisoning dinoflagellate Gymnodinium catenatum. FEMS Microbiol Ecol 47(3):345–357
Grossart HP, Levold F, Allgaier M, Simon M, Brinkhoff T (2005) Marine diatom species harbour distinct bacterial communities. Environ Microbiol 7(6):860–873
Hahnke RL, Bennke CM, Fuchs BM, Mann AJ, Rhiel E, Teeling H, Amann R, Harder J (2015) Dilution cultivation of marine heterotrophic bacteria abundant after a spring phytoplankton bloom in the North Sea. Environ Microbiol 17:3515–3526
Hasegawa Y, Martin JL, Giewat MW, Rooney-Varga JN (2007) Microbial community diversity in the phycosphere of natural populations of the toxic alga, Alexandrium fundyense. Environ Microbiol 9(12):3108–3121
Li S, Chen M, Chen Y, Tong J, Wang L, Xu Y, Hu Z, Chen H (2019) Epibiotic bacterial community composition in red-tide dinoflagellate Akashiwo sanguinea culture under various growth conditions. FEMS Microbiol Ecol 95(5):fiz057
Lu SH, Hodgkiss IJ (2004) Harmful algal bloom causative collected from Hong Kong waters. Hydrobiologia 512:231–238
Luria CM, Amaral-Zettler LA, Ducklow HW, Rich JJ (2016) Seasonal succession of free-living bacterial communities in coastal waters of the western Antarctic peninsula. Front Microbiol 3:7–1731. NoveCollection 2016
Main CR, Salvitti LR, Whereat EB, Coyne KJ (2015) Community-Level and species-specific associations between phytoplankton and particle-associated Vibrio species in Delaware’s Inland Bays. Appl Environ Microbiol 81(17):5703–5713
Manichanh C, Rigottier-Gois L, Bonnaud E, Gloux K, Pelletier E, Frangeul L, Nalin R, Jarrin C, Chardon P, Marteau P, Roca J, Dore J (2006) Reduced diversity of faecal microbiota in Crohn’s disease revealed by a metagenomic approach. Gut 55:205–211
Mayali X, Azam F (2004) Algicidal bacteria in the sea and their impact on algal blooms. J Eukaryot Microbiol 51(2):139–144
Oliver JD (2010) Recent findings on the viable but nonculturable state in pathogenic bacteria. FEMS Microb Rev 34:415–425
Riemann L, Steward GF, Azam F (2000) Dynamics of bacterial community composition and activity during a mesocosm diatom Bloom. Appl Environ Microbiol 66(2):578–587
Schwenk D, Nohynek L, Rischer H (2014) Algae-bacteria association inferred by 16S rDNA similarity in established microalgae cultures. Microbiologyopen 3(3):356–368
Shao Q, Lin Z, Zhou C, Zhu P, Yan X (2020) Succession of bacterioplankton communities over complete Gymnodinium-diatom bloom cycles. Sci Total Environ 709:135951
Shumway SE (1990) A review of the effects of algal blooms on shellfish and aquaculture. J World Aquacul Soc 21:65–104
Sintes E, Witte H, Stodderegger K, Steiner P, Herndl GJ (2013) Temporal dynamics in the free-living bacterial community composition in the coastal North Sea. FEMS Microbiol Ecol 83(2):413–424
Sison-Mangus MP, Jiang S, Kudela RM, Mehic S (2016) Phytoplankton-associated bacterial community composition and succession during toxic diatom bloom and non-bloom events. Front Microbiol 12:7–1433. https://doi.org/10.3389/fmicb.2016.01433. eCollection 2016
Teeling H, Fuchs BM, Becher D et al. (2012) Substrate-controlled succession of marine bacterioplankton populations induced by a phytoplankton bloom. Science 336:608–611
Wang X, Liu J, Liang J, Sun H, Zhang XH (2020) Spatiotemporal dynamics of the total and active spp. populations throughout the Changjiang estuary in China. Environmental Microbiology 22(10):4438–4455
White AE, Watkins-Brandt KS, McKibben SM, Wood AM, Hunter M, Forster Z, Du X, Peterson WT (2014) Large-scale bloom of Akashiwo sanguinea in the Northern California current system in 2009. Harmful Algae 37:38–46
Wu YL, Zhou CX, Zhang YS, Pu XM, Li WH (2001) Evolution and causes of formation of Gymnodinium sanguinea bloom in Yantai Sishili Bay. Oceanol Et Limnol Sin 32(2):159–167. In Chinese
Xu HS, Roberts N, Singleton FL, Attwell RW, Grimes DJ, Colwell RR (1982) Survival and viability of nonculturable Escherichia coli and Vibrio cholerae in the estuarine and marine environment. Microb Ecol 8:313–323
Yang C, Li Y, Zhou Y, Zheng W, Tian Y, Zheng T (2012) Bacterial community dynamics during a bloom caused by Akashiwo sanguinea in the Xiamen sea area, China. Harmful Algae 20:132–141
Yang C, Li Y, Zhou B, Zhou Y, Zheng W, Tian Y, Van Nostrand JD, Wu L, He Z, Zhou J, Zheng T (2015) Illumina sequencing-based analysis of free-living bacterial community dynamics during an Akashiwo sanguine bloom in Xiamen sea. China Sci Rep 5:8476
Yang C, Li Y, Zhou Y, Lei X, Zheng W, Tian Y, Van Nostrand JD, He Z, Wu L, Zhou J, Zheng T (2016) A comprehensive insight into functional profiles of free-living microbial community responses to a toxic Akashiwo sanguinea bloom. Sci Rep 6:34645
Zhang XH, Ahmad W, Zhu X, Chen J, Austin B (2021) Viable but nonculturable bacteria and their resuscitation: implications for cultivating uncultured marine microorganisms. Marine Life Science & Technology 3:189–203
Zhou J, Richlen ML, Sehein TR, Kulis DA, Anderson DM, Cai Z (2018). Microbial Community Structure and Associations During a Marine Dinoflagellate Bloom. Frontiers in Microbiology 9:1201
Acknowledgements
This work was supported by Grant Plan for Demonstration City Project for Marine Economic Development in Shenzhen (No.86), the Guangxi Innovation Drive Development Special Fund (Gui Ke AA18242047) and Shenzhen Basic Research Projects (JCYJ20180507182405562) to ZHU, Shenzhen Grant Plan for Science and Technology (NO. JCYJ20200109105823170) to HC. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, H., JIang, J., Jiang, F. et al. Temporal variability of free-living microbial culturability and community composition after an Akashiwo sanguinea bloom in Shenzhen, China. Ecotoxicology 30, 975–985 (2021). https://doi.org/10.1007/s10646-021-02407-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-021-02407-4