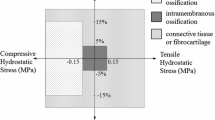
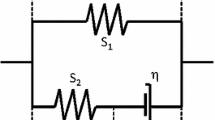
Abstract
The role of the growth plate reserve zone is not well understood. It has been proposed to serve as a source of stem cells and to produce morphogens that control the alignment of clones in preparation for the transition into the proliferative zone. We hypothesized that if such a role exists, there are likely to be mechanoregulatory stimuli in cellular response through the depth of the reserve zone. A poroelastic multiscale finite element model of bone/growth-plate/bone was developed for examining the reserve zone cell transient response when compressed to 5% of the cartilage thickness at strain rates of 0.18%/s, 5%/s, 50%/s, and 200%/s. Chondrocyte maximum principal strains, height-, width-, and membrane-strains were found to be highly dependent on reserve zone tissue depth and strain rate. Cell-level strains and fluid transmembrane outflow from the cell were influenced by the permeability of the calcified cartilage between subchondral bone plate and reserve zone and by the applied strain rate. Cell strain levels in the lower reserve zone were less sensitive to epiphyseal permeability than in the upper reserve zone. In contrast, the intracellular fluid pressures were relatively uniform with reserve zone tissue depth and less sensitive to epiphyseal permeability. Fluid shear stress, induced by fluid flow over the cell surface, provided mechanoregulatory signals potentially sufficient to stimulate reserve zone chondrocytes near the subchondral bone plate interface. These results suggest that the strain rate and tissue depth dependence of cell-level strains and cell surface fluid shear stress may provide mechanoregulatory cues in the reserve zone.








Similar content being viewed by others
References
Abad V, Meyers JL, Weise M, Gafni RI, Barnes KM, Nilsson O, Baron J (2002) The role of the resting zone in growth plate chondrogenesis. Endocrinology 143(5):1851–1857. https://doi.org/10.1210/endo.143.5.8776
Accadbled F, Laffosse JM, Ambard D, Gomez-Brouchet A, De Gauzy JS, Swider P (2008) Influence of location, fluid flow direction, and tissue maturity on the macroscopic permeability of vertebral end plates. Spine 33(6):612–619. https://doi.org/10.1097/BRS.0b013e318166e0d7
Alexopoulos LG, Setton LA, Guilak F (2005) The biomechanical role of the chondrocyte pericellular matrix in articular cartilage. Acta Biomater 1(3):317–325. https://doi.org/10.1016/j.actbio.2005.02.001
Argote PF, Kaplan JT, Poon A, Xu X, Cai L, Emery NC, Neu CP (2019) Chondrocyte viability is lost during high-rate impact loading by transfer of amplified strain, but not stress, to pericellular and cellular regions. Osteoarthritis Cartilage 27(12):1822–1830. https://doi.org/10.1016/j.joca.2019.07.018
Ateshian GA, Costa KD, Hung CT (2007) A theoretical analysis of water transport through chondrocytes. Biomech Model Mechanobiol 6:91–10
Berteau JP, Oyen M, Shefelbine SJ (2016) Permeability and shear modulus of articular cartilage in growing mice. Biomech Model Mechanobiol 15(1):205–212. https://doi.org/10.1007/s10237-015-0671-3
Bries AD, Weiner DS, Jacquet R, Adamczyk MJ, Morscher MA, Lowder E, Askew MJ, Steiner RP, Horne WI, Landis WJ (2012) A study in vivo of the effects of a static compressive load on the proximal tibial physis in rabbits. J Bone Joint Surg 94(15):e111. https://doi.org/10.2106/JBJS.K.00340
Cancel M, Grimard G, Thuillard-Crisinel D, Moldovan F, Villemure I (2009) Effects of in vivo static compressive loading on aggrecan and type II and X collagens in the rat growth plate extracellular matrix. Bone 44(2):306–315. https://doi.org/10.1016/j.bone.2008.09.005
Carter DR, Wong M (1988) Mechanical stresses and endochondral ossification in the chondroepiphysis. J Orthop Res 6(1):148–154. https://doi.org/10.1002/jor.1100060120
Carter DR, Mikić B, Padian K (1998) Epigenetic mechanical factors in the evolution of long bone epiphyses. Zool J Linn Soc 123(2):163–178. https://doi.org/10.1111/j.1096-3642.1998.tb01298.x
Chen T, Buckley M, Cohen I, Bonassar L, Awad HA (2012) Insights into interstitial flow, shear stress, and mass transport effects on ECM heterogeneity in bioreactor-cultivated engineered cartilage hydrogels. Biomech Model Mechanobiol 11(5):689–702. https://doi.org/10.1007/s10237-011-0343-x
Cohen B, Chorney GS, Phillips DP, Dick HM, Mow VC (1994) Compressive stress-relaxation behavior of bovine growth plate may be described by the nonlinear biphasic theory. J Orthop Res 12(6):804–813. https://doi.org/10.1002/jor.1100120608
Cohen B, Lai WM, Mow VC (1998) A transversely isotropic biphasic model for unconfined compression of growth plate and chondroepiphysis. ASME J Biomech Eng 120(4):491–496. https://doi.org/10.1115/1.2798019
De Andrea CE, Wiweger M, Prins F, Bovée JV, Romeo S, Hogendoorn PC (2010) Primary cilia organization reflects polarity in the growth plate and implies loss of polarity and mosaicism in osteochondroma. Lab Invest 90(7):1091–1101. https://doi.org/10.1038/labinvest.2010.81
Eberhardt AW, Keer LM, Lewis JL, Vithoontien V (1990) An analytical model of joint contact. ASME J Biomech Eng 112(4):407–413. https://doi.org/10.1115/1.2891204
Fan C, Cool JC, Scherer MA, Foster BK, Shandala T, Tapp H, Xian CJ (2009) Damaging effects of chronic low-dose methotrexate usage on primary bone formation in young rats and potential protective effects of folinic acid supplementary treatment. Bone 44(1):61–70. https://doi.org/10.1016/j.bone.2008.09.014
Farzaneh S, Paseta O, Gómez-Benito MJ (2015) Multi-scale finite element model of growth plate damage during the development of slipped capital femoral epiphysis. Biomech Model Mechanobiol 14(2):371–385. https://doi.org/10.1007/s10237-014-0610-8
Ficklin TP, Davol A, Klisch SM (2009) Simulating the growth of articular cartilage explants in a permeation bioreactor to aid in experimental protocol design. ASME J Biomech Eng 131(4):041008. https://doi.org/10.1115/1.3049856
Frost HM (1990) Skeletal structural adaptations to mechanical usage (SATMU): 3. The hyaline cartilage modeling problem. Anatom Rec 226(4):423–432. https://doi.org/10.1002/ar.1092260404
Gao J, Williams JL, Roan E (2014) On the state of stress in the growth plate under physiologic compressive loading. Open Journal of Biophysics 4(1):13–21. https://doi.org/10.4236/ojbiphy.2014.41003
Gao J, Roan E, Williams JL (2015) Regional variations in growth plate chondrocyte deformation as predicted by three-dimensional multi-scale simulations. PLoSONE 10(4):e0124862. https://doi.org/10.1371/journal.pone.0124862
Gao J, Williams JL, Roan E (2017) Multiscale modeling of growth plate cartilage mechanobiology. Biomech Model Mechanobiol 16(2):667–679. https://doi.org/10.1007/s10237-016-0844-8
Gemmiti CV, Guldberg RE (2009) Shear stress magnitude and duration modulates matrix composition and tensile mechanical properties in engineered cartilaginous tissue. Biotechnol Bioeng 104(4):809–820. https://doi.org/10.1002/bit.22440
Grimm MJ, Williams JL (1997) Measurements of permeability in human calcaneal trabecular bone. J Biomech 30(7):743–745. https://doi.org/10.1016/S0021-9290(97)00016-X
Guharay F, Sachs F (1984) Stretch-activated single ion channel currents in tissue-cultured embryonic chick skeletal muscle. J Physiol 352(1):685–701. https://doi.org/10.1113/jphysiol.1984.sp015317
Guilak F, Mow VC (2000) The mechanical environment of the chondrocyte: a biphasic finite element model of cell–matrix interactions in articular cartilage. J Biomech 33(12):1663–1673. https://doi.org/10.1016/S0021-9290(00)00105-6
Gupta T, Donahue TLH (2006) Role of cell location and morphology in the mechanical environment around meniscal cells. Acta Biomater 2(5):483–492. https://doi.org/10.1016/j.actbio.2006.05.009
Hallett SA, Ono W, Ono N (2019) Growth plate chondrocytes: skeletal development, growth and beyond. Int J Mol Sci 20(23):6009. https://doi.org/10.3390/ijms20236009
Hwang J, Bae WC, Shieu W, Lewis CW, Bugbee WD, Sah RL (2008) Increased hydraulic conductance of human articular cartilage and subchondral bone plate with progression of osteoarthritis. Arthritis Rheumat Off J Am College Rheumatol 58(12):3831–3842. https://doi.org/10.1002/art.24069
Kazemi M, Williams JL On the role of the reserve zone and mechano-regulatory stimuli in the development and maturation of the growth plate: Observations and models. Manuscript submitted for publication.
Kazemi M, Williams JL (2018) Elemental and histological study of the growth plate reserve zone–subchondral bone interface. Paper presented at: Orthopaedic Research Society Annual Meeting (ORS), 2018; New Orleans, LA.
Kazemi M, Williams JL (2019) Chondrocyte and pericellular matrix deformation and strain in the growth plate cartilage reserve zone under compressive loading. In: International Symposium on Computer Methods in Biomechanics and Biomedical Engineering (pp. 526–538). Springer, Cham. https://doi.org/10.1007/978-3-030-43195-2_43.
Kazemi M, Williams JL (2020) Properties of cartilage-subchondral bone junctions: a narrative review with specific focus on the growth plate. Cartilage. https://doi.org/10.1177/1947603520924776
Komeili A, Otoo BS, Abusara Z, Sibole S, Federico S, Herzog W (2020) Chondrocyte deformations under mild dynamic loading conditions. Ann Biomed Eng. https://doi.org/10.1007/s10439-020-02615-9
Lacroix D, Prendergast PJ, Li G, Marsh D (2002) Biomechanical model to simulate tissue differentiation and bone regeneration: application to fracture healing. Med Biol Eng Compu 40(1):14–21. https://doi.org/10.1007/BF02347690
Lai WM, Mow VC (1980) Drag-induced compression of articular cartilage during a permeation experiment. Biorheology 17(1–2):111–123. https://doi.org/10.3233/BIR-1980-171-213
Lai WM, Mow VC, Roth V (1981) Effects of nonlinear strain-dependent permeability and rate of compression on the stress behavior of articular cartilage. ASME J Biomech Eng 103(2):61–66. https://doi.org/10.1115/1.3138261
Lavagnino M, Arnoczky SP, Kepich E, Caballero O, Haut RC (2008) A finite element model predicts the mechanotransduction response of tendon cells to cyclic tensile loading. Biomech Model Mechanobiol 7(5):405–416. https://doi.org/10.1007/s10237-007-0104-z
Leipzig ND, Athanasiou KA (2008) Static compression of single chondrocytes catabolically modifies single-cell gene expression. Biophys J 94(6):2412–2422. https://doi.org/10.1529/biophysj.107.114207
Lui JC (2020) Home for a rest: stem cell niche of the postnatal growth plate. J Endocrinol 246(1):R1–R11. https://doi.org/10.1530/JOE-20-0045
Maroudas A, Bullough P, Swanson SAV, Freeman MAR (1968) The permeability of articular cartilage. The Journal of bone and joint surgery. 50(1):166–177. https://doi.org/10.1302/0301-620X.50B1.166 (British volume)
Matsushita Y, Ono W, Ono. (2020) Growth plate skeletal stem cells and their transition from cartilage to bone. Bone 136:115359. https://doi.org/10.1016/j.bone.2020.115359
Moo EK, Herzog W (2017) Unfolding of membrane ruffles of in situ chondrocytes under compressive loads. J Orthop Res 35(2):304–310
Moo EK, Herzog W, Han SK, Osman N, Pingguan-Murphy B, Federico S (2012) Mechanical behavior of in-situ chondrocytes subjected to different loading rates: a finite element study. Biomech Model Mechanobiol 11(7):983–993. https://doi.org/10.1007/s10237-011-0367-2
Moo EK, Amrein M, Epstein M, Duvall M, Osman NAA, Pingguan-Murphy B, Herzog W (2013) The properties of chondrocyte membrane reservoirs and their role in impact-induced cell death. Biophys J 105(7):1590–1600. https://doi.org/10.1016/j.bpj.2013.08.035
Moo EK, Han SK, Federico S, Sibole SC, Jinha A, Osman NAA, Herzog W (2014) Extracellular matrix integrity affects the mechanical behavior of in-situ chondrocytes under compression. J Biomech 47(5):1004–1013. https://doi.org/10.1016/j.jbiomech.2014.01.003
Morris CE, Sigurdson W (1989) Stretch-inactivated ion channels coexist with stretch-activated ion channels. Science 243(4892):807–809. https://doi.org/10.1126/science.2536958
Mow VC, Guo XE (2002) Mechano-electrochemical properties of articular cartilage: their inhomogeneities and anisotropies. Annu Rev Biomed Eng 4(1):175–209. https://doi.org/10.1146/annurev.bioeng.4.110701.120309
Mow VC, Kuei SC, Lai WM, Armstrong CG (1980) Biphasic creep and stress relaxation of articular cartilage in compression: theory and experiments. ASME J Biomech Eng 102(1):73–84. https://doi.org/10.1115/1.3138202
Mow VC, Hou JS, Owens JM, Ratcliffe A (1990) Biphasic and quasilinear viscoelastic theories for hydrated soft tissues. In: Biomechanics of diarthrodial joints (pp. 215–260). Springer, New York, NY. https://doi.org/10.1007/978-1-4612-3448-7_8.
Nauman EA, Fong KE, Keaveny TM (1999) Dependence of intertrabecular permeability on flow direction and anatomic site. Ann Biomed Eng 27(4):517–524. https://doi.org/10.1114/1.195
Nguyen BV, Wang QG, Kuiper NJ, El Haj AJ, Thomas CR, Zhang Z (2010) Biomechanical properties of single chondrocytes and chondrons determined by micromanipulation and finite-element modelling. J R Soc Interface 7(53):1723–1733. https://doi.org/10.1098/rsif.2010.0207
Pauwels F (1960) Eine neue Theorie über den Einfluß mechanischer Reize auf die Differenzierung der Stützgewebe. Z Anat Entwicklung 121(6):478–515. https://doi.org/10.1007/BF00523401
Pérez del Palomar A, Doblaré M (2006) On the numerical simulation of the mechanical behavior of articular cartilage. Int J Numer Meth Eng 67(9):1244–1271. https://doi.org/10.1002/nme.1638
Poole CA, Flint MH, Beaumont BW (1987) Chondrons in cartilage: ultrastructural analysis of the pericellular microenvironment in adult human articular cartilages. J Orthop Res 5(4):509–522. https://doi.org/10.1002/jor.1100050406
Radhakrishnan P, Lewis NT, Mao JJ (2004) Zone-specific micromechanical properties of the extracellular matrices of growth plate cartilage. Ann Biomed Eng 32(2):284–291. https://doi.org/10.1023/B:ABME.0000012748.41851.b4
Sakai N, Hagihara Y, Hashimoto C, Komori M, Sawae Y, Murakami T (2015) An estimation of mechanical properties of articular cartilage for biphasic finite element analyses. J Biomech Sci Eng 10(4):15–00228. https://doi.org/10.1299/jbse.15-00228
Seeger-Nukpezah T, Golemis EA (2012) The extracellular matrix and ciliary signaling. Curr Opin Cell Biol 24(5):652–661. https://doi.org/10.1016/j.ceb.2012.06.002
Sergerie K, Lacoursière MO, Lévesque M, Villemure I (2009) Mechanical properties of the porcine growth plate and its three zones from unconfined compression tests. J Biomech 42(4):510–516. https://doi.org/10.1016/j.jbiomech.2008.11.026
Shao YY, Wang L, Welter JF, Ballock RT (2012) Primary cilia modulate Ihh signal transduction in response to hydrostatic loading of growth plate chondrocytes. Bone 50(1):79–84. https://doi.org/10.1016/j.bone.2011.08.033
Shefelbine SJ, Carter DR (2004) Mechanobiological predictions of growth front morphology in developmental hip dysplasia. J Orthop Res 22(2):346–352. https://doi.org/10.1016/j.orthres.2003.08.004
Soltz MA, Ateshian GA (1998) Experimental verification and theoretical prediction of cartilage interstitial fluid pressurization at an impermeable contact interface in confined compression. J Biomech 31(10):927–934. https://doi.org/10.1016/S0021-9290(98)00105-5
Song Y, Lee D, Shin CS, Carter DR, Giori NJ (2013) Physeal cartilage exhibits rapid consolidation and recovery in intact knees that are physiologically loaded. J Biomech 46(9):1516–1523. https://doi.org/10.1016/j.jbiomech.2013.03.026
Speer DP (1982) Collagenous architecture of the growth plate and perichondrial ossification groove. J Bone Joint Surg 64(3):399–407. https://doi.org/10.2106/00004623-198264030-00010 (American volume)
Stender ME, Regueiro RA, Ferguson VL (2017) A poroelastic finite element model of the bone–cartilage unit to determine the effects of changes in permeability with osteoarthritis. Comput Methods Biomech Biomed Engin 20(3):319–331. https://doi.org/10.1080/10255842.2016.1233326
Sylvestre PL, Villemure I, Aubin CE (2007) Finite element modeling of the growth plate in a detailed spine model. Med Biol Eng Compu 45(10):977–988. https://doi.org/10.1007/s11517-007-0220-z
Tutorino, JC, Khubchandani, ZG, Williams, JL, Cobb, CM and Schmidt, TL (2001). Can the epiphyseal growth plate be injured in compression? In 47th Annual Meeting of Orthopaedic Research Society (p. 353).
Williams JL, Do PD, Eick JD, Schmidt TL (2001) Tensile properties of the physis vary with anatomic location, thickness, strain rate and age. J Orthop Res 19(6):1043–1048. https://doi.org/10.1016/S0736-0266(01)00040-7
Wosu R, Sergerie K, Lévesque M, Villemure I (2012) Mechanical properties of the porcine growth plate vary with developmental stage. Biomech Model Mechanobiol 11(3–4):303–312. https://doi.org/10.1007/s10237-011-0310-6
Wu JZ, Herzog W (2000) Finite element simulation of location-and time-dependent mechanical behavior of chondrocytes in unconfined compression tests. Ann Biomed Eng 28(3):318–330. https://doi.org/10.1114/1.271
Wu JZ, Herzog W, Epstein M (1997) Evaluation of the finite element software ABAQUS for biomechanical modelling of biphasic tissues. J Biomech 31(2):165–169. https://doi.org/10.1016/S0021-9290(97)00117-6
Yellowley CE, Jacobs CR, Li Z, Zhou Z, Donahue HJ (1997) Effects of fluid flow on intracellular calcium in bovine articular chondrocytes. Am J Physiol Cell Physiol 273(1):C30–C36. https://doi.org/10.1152/ajpcell.1997.273.1.C30
Acknowledgements
We thank Dr. Eng Kuan Moo at the University of Calgary for his help with computational implementation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kazemi, M., Williams, J.L. Depth and strain rate-dependent mechanical response of chondrocytes in reserve zone cartilage subjected to compressive loading. Biomech Model Mechanobiol 20, 1477–1493 (2021). https://doi.org/10.1007/s10237-021-01457-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-021-01457-1