Abstract
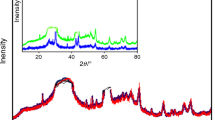
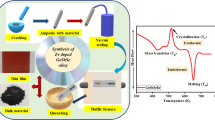
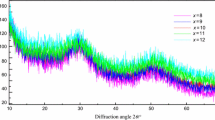
The current paper explains the kinetic variables of crystallization kinetics. Se82−xTe18Gex (0 ≤ x ≤ 12) amorphous glassy materials were synthesized by the melt quench technique. XRD, SEM, and EDX characterizations were done to analyze the structure and elemental composition of the prepared amorphous glassy alloys. DSC measurements were taken to study the phase transition of the amorphous glassy alloys under identical conditions. For the crystallization kinetics, Kissinger, Ozawa, Tang, and Starink iso-conversional methods were used. The order of dimensional growth and frequency factors were examined with the help of the Matusita–Sakka and Augis–Bennett methods, respectively. Also, the compositional dependence of kinetic parameters was studied for the prepared Se-Te-Ge (STG) glassy alloys.












Similar content being viewed by others
References
D Lencer, M Salinga and M Wuttig Adv. Mater. 23 2030 (2011).
A A. Dubkov and B Spagnolo Eur. Phys. J. B 65 361 (2008).
B Spagnolo et al. Chaos, Solitons and Fractals 000 (2015)
D Valenti, L Magazz`u, P Caldara and B Spagnolo Physical Review B 91 235412 (2015).
B Spagnolo, C Guarcello, L Magazzù and A Carollo Dominique persano adorno and davide valenti Entropy 19 20 (2017).
A V Yakimov et al. Appl. Phys. Lett. 114 253506 (2019).
H Fritzsche Annu. Rev. Mat. Sci. 2 697 (1972).
V Srivastava and P Mishra Indian J. Phys. 94 1 (2020).
I H Afify, M S Abo-Ghazala, M M El-Zaidia and E M Farag Indian J. Phys. 94 447 (2020).
V Rao and D K Dwivedi J. Mat. Sci. Elect. 28 6208 (2017).
V Rao, N Mehta, N Chandel and D K Dwivedi Phase Trans. 91 490 (2018).
V Rao, N Chandel, N Mehta and D K Dwivedi J. Therm. Anal. Calorim. (2018)
P K Singh and D K Dwivedi Ferroelectrics 520 265 (2017).
B S Patial and S K Tripathi J. Therm. Anal. Calorim. 106 845 (2011).
P Lucas, Z Yang, M K Fah, T Luo, S Jiang and C Boussard-Pledel Opt. Mater Express. 3 1049 (2013).
S Sen, T Edwards, J Y Cho and Y C Joo Phys. Rev. Lett. 108 195506 (2012).
S Cui et al. Adv. Appl. Ceram. 114 S42 (2015).
H Adachi and K C Kao J. Appl. Phys 51 6326 (1980).
Z Cernosek, E Cernoskova, M Hejdova and J Holubora J. Non-Cryst. Solid 460 169 (2017).
R Svoboda, D Brandova and J Malek J. Alloys Comput. 680 427 (2016).
A Cheruvalath, I Sebastian, M Sebastian, VPN Nampoori 72 265 (2017)
J Zavadil, P Kostka, J Pedlikova, K Zdansky et al J. Non-Cryst. Solid. 355 2083 (2009).
M Mohamed, S Moustafa, A M A Elnaiem and M M A Rahim J. Alloys Comput. 647 771 (2015).
Z U Borisova Glassy Semiconductors. (New York: Plenum) (1981)
W A Jonnson and K F Mehl Trans. Am. Inst. Mining Met. Engns. 12 135 (1981).
M Avrami J. Chem Phys. 7 1103 (1939).
M Avrami J. Chem. Phys. 8 212 (1940).
M Avrami and J Granulation J. Chem. Phys. 9 177 (1941).
S Glasstone Textbook of Physical chemistry, 2nd edn. (New York: Macmillan India Ltd.) (1991)
K Matusita, T K Konotsu and R Yorota J. Mater. Sci. 38 741 (1980).
T Wanjun and C Donghua Thermochim. Acta 443 72 (2005).
M J Starink Thermochim Acta 288 97 (1996).
C D Doyle J. Appl. Polym. Sci. 5 285 (1961).
J A Augis and J E Bennett J. Therm. Anal. Calor. 13 283 (1978).
W Zhang, R Mazzarello, M Wuttig and E Ma Nature Reviews Materials 4 150 (2019).
B Chen, G H T Brink, G Palasantzas and B J Kooi The Journal of Physical Chemistry C 121 8569 (2017).
Acknowledgements
The authors are thankful to ACMS, Indian Institute of Technology, Kanpur, India, for providing the experimental facilities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rao, V., Singh, P.K., Lohia, P. et al. Non-isothermal crystallization kinetics of Se82−xTe18Gex (0 ≤ x ≤ 12) for memory applications. Indian J Phys 96, 1075–1085 (2022). https://doi.org/10.1007/s12648-021-02036-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12648-021-02036-x