Abstract
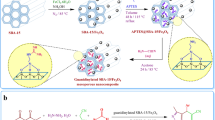
In this work, tris(hydroxymethyl)aminomethane-Zirconium complex supported on modified SBA-15 (SBA-15@n-Pr-THMAM-ZrO) prepared as a novel mesoporous catalyst. The structure of this porous compound was characterized by XRD, nitroge adsorption–desorption, TEM, TGA, EDS, ICP, X-ray mapping, and SEM techniques. The catalytic activity of SBA-15@n-Pr-THMAM-Zr has been investigated for the multicomponent synthesis of polyhydroquinoline and 2,3-dihydroquinazolin-4(1H)-one derivaives.













Similar content being viewed by others
References
M. Kazemi, M. Mohammadi, Appl. Organomet. Chem. 34, e5400 (2020)
B. Maleki, N. Nasiri, R. Tayebee, A. Khojastehnezhad, H.A. Akhlaghi, RSC Adv. 6, 79128 (2016)
A. Ghorbani-Choghamarani, R. Sahraei, Z. Taherinia, M. Mohammadi, J. Iran. Chem. Soc. (2020).
M. Nikoorazm, M. Mohammadi, M. Khanmoradi, Appl. Organomet. Chem. 34, e5704 (2020)
M. Mohammadi, M. Khodamorady, B. Tahmasbi, K. Bahrami, A. Ghorbani-Choghamarani, J. Ind. Eng. Chem. (2021). https://doi.org/10.1016/j.jiec.2021.02.001
M. Mohammadi, A. Ghorbani-Choghamarani, New J. Chem. 44, 2919 (2020)
A. Ghorbani-Choghamarani, M. Mohammadi, Z. Taherinia, J. Iran. Chem. Soc. 16, 411 (2019)
A. Ghorbani-Choghamarani, M. Mohammadi, R.H.E. Hudson, T. Tamoradi, Appl. Organomet. Chem. 33, e4977 (2019)
F. Adibian, A.R. Pourali, B. Maleki, M. Baghayeri, A. Amiri, Polyhedron 175, 114179 (2020)
B. Maleki, H. Alinezhad, H. Atharifar, R. Tayebee, A.V. Mofrad, Org. Prep. Proced. Int. 51, 301 (2019)
H. Firouzabadi, M. Jafarpour, J. Iran. Chem. Soc. 5, 159 (2008)
J. Aboonajmi, M.R. Mousavi, M.T. Maghsoodlou, N. Hazeri, A. Masoumnia, Res. Chem. Intermed. 41, 1925 (2015)
B.F. Mirjalili, M.A. Zolfigol, A. Bamoniri, M.A. Karimi-Zarchi, Z. Zaghaghi, M. Parvaideh, J. Iran. Chem. Soc. 4, 340 (2007)
B. Vijayakumar, G.R. Rao, J. Porous Mater. 19, 491 (2012)
Z. Esam, M. Akhavan, A. Bekhradnia, M. Mohammadi, S. Tourani, Catal. Lett. 150, 3112 (2020)
T. Tamoradi, S. Masoumeh Mousavi, and M. Mohammadi, ChemistrySelect 5, 5077 (2020).
Z. Derikvand, F. Derikvand, Chin. J. Catal. 32, 532 (2011)
A. E. A. A. Said, M. M. M. Abd El-Wahab, and M. Abd El-Aal, Res. Chem. Intermed. 42, 1537 (2016).
R. Pourhasan-Kisomi, F. Shirini, and M. Golshekan, Appl. Organomet. Chem. 32, (2018).
M.A. Zolfigol, P. Salehi, M. Shiri, Z. Tanbakouchian, Catal. Commun. 8, 173 (2007)
E. Kolvari, M.A. Zolfigol, N. Koukabi, M. Gilandust, A.V. Kordi, J. Iran. Chem. Soc. 10, 1183 (2013)
A. Ghorbani-Choghamarani, M. Mohammadi, L. Shiri, Z. Taherinia, Res. Chem. Intermed. 45, 5705 (2019)
M. Nikoorazm, M. Khanmoradi, M. Mohammadi, Appl. Organomet. Chem. 34, e5504 (2020)
H. Filian, A. Kohzadian, M. Mohammadi, A. Ghorbani-Choghamarani, A. Karami, Appl. Organomet. Chem. 34, e5579 (2020)
N. Moeini, M. Ghadermazi, A. Ghorbani-Choghamarani, Polyhedron 170, 278 (2019)
B. Maleki, H. Natheghi, R. Tayebee, H. Alinezhad, A. Amiri, S.A. Hossieni, S.M.M. Nouri, Polycycl. Aromat. Compd. 40, 633 (2020)
B. Maleki, R. Tayebee, M. Kermanian, S.S. Ashrafi, J. Mex. Chem. Soc. 57, 290 (2013)
A. Ghorbani-Choghamarani, H. Aghavandi, M. Mohammadi, Appl. Organomet. Chem. 34, e5804 (2020)
T. Tamoradi, S.M. Mousavi, M. Mohammadi, New J. Chem. 44, 8289 (2020)
M. Lashanizadegan, Z. Zareian, Catal. Lett. 141, 1698 (2011)
I. C. Gerber and P. Serp, Chem. Rev. (2019).
B. Maleki, H. Atharifar, O. Reiser, R. Sabbaghzadeh, Polycycl. Aromat. Compd. 1 (2019).
A.A. Ardakani, H. Kargar, N. Feizi, M.N. Tahir, J. Iran. Chem. Soc. 15, 1495 (2018)
C. Xie, Z. Niu, D. Kim, M. Li, and P. Yang, Chem. Rev. (2019).
Q.R. Fang, T.A. Makal, M.D. Young, H.C. Zhou, Comments Inorg. Chem. 31, 165 (2010)
C.M.A. Parlett, K. Wilson, A.F. Lee, Chem. Soc. Rev. 42, 3876 (2013)
A. Corma, Chem. Rev. 97, 2373 (1997)
I.I. Ivanova, E.E. Knyazeva, Chem. Soc. Rev. 42, 3671 (2013)
D.E. De Vos, M. Dams, B.F. Sels, P.A. Jacobs, Chem. Rev. 102, 3615 (2002)
J. Salonen, A.M. Kaukonen, J. Hirvonen, V.P. Lehto, J. Pharm. Sci. 97, 632 (2008)
N. Mizoshita, T. Tani, S. Inagaki, Chem. Soc. Rev. 40, 789 (2011)
B. Naik, N. Ghosh, Recent Pat. Nanotechnol. 3, 213 (2009)
T. Wagner, S. Haffer, C. Weinberger, D. Klaus, M. Tiemann, Chem. Soc. Rev. 42, 4036 (2013)
B. Melde, B. Johnson, P. Charles, Sensors 8, 5202 (2008)
M. Hasanzadeh, N. Shadjou, M. de la Guardia, M. Eskandani, P. Sheikhzadeh, TrAC Trends Anal. Chem. 33, 117 (2012)
S. Wang, Microporous Mesoporous Mater. 117, 1 (2009)
N. Vadia, S. Rajput, Asian J. Pharm. Clin. Res. 4, 44 (2011)
M. Vallet-Regí, F. Balas, D. Arcos, Angew. Chemie Int. Ed. 46, 7548 (2007)
H. Yu, Q.-Z. Zhai, Microporous Mesoporous Mater. 123, 298 (2009)
F. Qu, G. Zhu, S. Huang, S. Li, J. Sun, D. Zhang, S. Qiu, Microporous Mesoporous Mater. 92, 1 (2006)
A. Ghorbani-Choghamarani, M. Mohammadi, T. Tamoradi, M. Ghadermazi, Polyhedron 158, 25 (2019)
S. Singh, R. Kumar, H.D. Setiabudi, S. Nanda, D.V.N. Vo, Appl. Catal. A 559, 57 (2018)
R. Huirache-Acuña, R. Nava, C. Peza-Ledesma, J. Lara-Romero, G. Alonso-Núez, B. Pawelec, E. Rivera-Muñoz, Materials (Basel). 6, 4139 (2013)
D.P. Sahoo, D. Rath, B. Nanda, K.M. Parida, RSC Adv. 5, 83707 (2015)
T. Tamoradi, M. Ghadermazi, A. Ghorbani-Choghamarani, J. Porous Mater. 26, 121 (2019)
S. Rostamnia, F. Pourhassan, Chinese Chem. Lett. 24, 401 (2013)
Z. Sun, Y. Wang, Z. Zhang, F. Zhu, P. Zhao, G. Li, F. Shao, J. Rui, Res. Chem. Intermed. 45, 3107 (2019)
H. Filian, A. Ghorbani-Choghamarani, E. Tahanpesar, J. Porous Mater. 26, 1091 (2019)
H. Filian, A. Ghorbani-Choghamarani, E. Tahanpesar, J. Iran. Chem. Soc. 16, 2673 (2019)
A. Kohzadian, H. Filian, Z. Kordrostami, A. Zare, A. Ghorbani-Choghamarani, Res. Chem. Intermed. 46, 1941 (2020)
S. Kumar, J. Heterocycl. Chem. 56, 1168 (2019)
M.M. Heravi, N. Nami, M.M. Heravi, N. Ramezanian, J. Iran. Chem. Soc. 5, S21 (2008)
M.A. Zolfigol, M. Yarie, RSC Adv. 5, 103617 (2015)
M. Yarie, M.A. Zolfigol, Y. Bayat, A. Asgari, D.A. Alonso, A. Khoshnood, RSC Adv. 6, 82842 (2016)
M. Dabiri, P. Salehi, M. Baghbanzadeh, M.A. Zolfigol, M. Agheb, S. Heydari, Catal. Commun. 9, 785 (2008)
T. Tamoradi, S.M. Mousavi, M. Mohammadi, New J. Chem. 44, 3012 (2020)
P. Salehi, M. Ali Zolfigol, F. Shirini, and M. Baghbanzadeh, Curr. Org. Chem. 10, 2171 (2006).
B.D. Rupnar, T.R. Kachave, P.D. Jawale, S.U. Shisodia, R.P. Pawar, J. Iran. Chem. Soc. 14, 1853 (2017)
P. V Govardhana Reddy, B. Rajendra Prasad Reddy, M. Venkata Krishna Reddy, K. Raghava Reddy, N. P. Shetti, T. A. Saleh, and T. M. Aminabhavi, J. Environ. Manage. (2020).
M.A. Zolfigol, H. Ghaderi, S. Baghery, L. Mohammadi, J. Iran. Chem. Soc. 14, 121 (2017)
P. Salehi, M. Dabiri, M. A. Zolfigol, and M. Baghbanzadeh, Synlett 1155 (2005).
J. C. García-Martínez, H. A. González Uribe, M. M. González-Brambila, J. A. Colín-Luna, Y. E. Escobedo-García, A. López-Gaona, and L. Alvarado-Perea, Catal. Today 305, 40 (2018).
J.P. Thielemann, F. Girgsdies, R. Schlögl, C. Hess, Beilstein J. Nanotechnol. 2, 110 (2011)
S. Ganji, P. Bukya, V. Vakati, K.S.R. Rao, D.R. Burri, Catal. Sci. Technol. 3, 409 (2013)
M. Prokopowicz, J. Żeglinski, A. Szewczyk, A. Skwira, G. Walker, AAPS PharmSciTech 20, 17 (2019)
T. Tamoradi, M. Ghadermazi, A. Ghorbani-Choghamarani, J. Saudi Chem. Soc. 23, 846 (2019)
H. Ghafuri, N. Goodarzi, A. Rashidizadeh, and M. A. Douzandegi Fard, Res. Chem. Intermed. 45, 5027 (2019).
S.U. Tekale, S.B. Munde, S.S. Kauthale, R.P. Pawar, Org. Prep. Proced. Int. 50, 314 (2018)
M. Hajjami, A. Ghorbani-Choghamarani, R. Ghafouri-Nejad, B. Tahmasbi, New J. Chem. 40, 3066 (2016)
J. Herrero, C. Blanco, M.A. Esteruelas, L.A. Oro, Appl. Organomet. Chem. 4, 157 (1990)
T. Tamoradi, M. Ghadermazi, A. Ghorbani-Choghamarani, Appl. Organomet. Chem. 32, e3974 (2018)
A. Maleki, A.R. Akbarzade, A.R. Bhat, J. Nanostructure Chem. 7, 309 (2017)
H. Alinezhad, M. Tarahomi, B. Maleki, and A. Amiri, Appl. Organomet. Chem. 33, (2019).
N. Taheri, F. Heidarizadeh, A. Kiasat, J. Magn. Magn. Mater. 428, 481 (2017)
Q. Zhang, X.M. Ma, H.X. Wei, X. Zhao, J. Luo, RSC Adv. 7, 53861 (2017)
E. Pourian, S. Javanshir, Z. Dolatkhah, S. Molaei, A. Maleki, ACS Omega 3, 5012 (2018)
T. dos Santos, C. Grundke, T. Lucas, L. Großmann, G.C. Clososki, T. Opatz, European. J. Org. Chem. 2020, 6429 (2020)
M.A. Erfan, B. Akhlaghinia, S.S.E. Ghodsinia, ChemistrySelect 5, 2306 (2020)
Y.-H. Shang, L.-Y. Fan, X.-X. Li, M.-X. Liu, Chinese Chem. Lett. 26, 1355 (2015)
S.B. Bharate, N. Mupparapu, S. Manda, J.B. Bharate, R. Mudududdla, R.R. Yadav, R.A. Vishwakarma, ARKIVOC 2012, 308 (2012)
S. Zhaleh, N. Hazeri, M.T. Maghsoodlou, Res. Chem. Intermed. 42, 6381 (2016)
Acknowledgements
This work was supported by the research facilities of Ilam University, Ilam, Iran, and Bu-Ali Sina University, Hamedan, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest with this research work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghorbani-Choghamarani, A., Aghavandi, H. & Mohammadi, M. Mesoporous SBA-15@n-Pr-THAM-ZrO organic–inorganic hybrid: as a highly efficient reusable nanocatalyst for the synthesis of polyhydroquinolines and 2,3-dihydroquinazolin-4 (1h)-ones. J Porous Mater 28, 1167–1186 (2021). https://doi.org/10.1007/s10934-021-01063-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-021-01063-7