Abstract
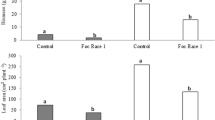
Previous research has shown that application of iron chelates to soil reduces Fusarium wilt in several crop species. The aim of this work was to test the effect for bananas grown in tropical soils. Disease severity and plant characteristics were measured in banana plants (cv. Ducasse, Musa ABB) grown in pots inoculated with Fusarium oxysporum f. sp. cubense, Race 1 in two experiments. Experiment 1 compared amendment with iron chelates (with ligands of differing iron binding stability) with water, plus an uninoculated unamended control, in two tropical Australian soils. Experiment 2 examined the effect of Fe-HBED application rate with high or low calcium addition. In Experiment 1, iron-saturated chelate application did not significantly affect disease severity or plant tissue iron concentration, irrespective of the iron binding stability of the chelate. In Experiment 2, disease severity was not affected by Fe-HBED or calcium application rate. The concentrations of iron and aluminium in plants were both somewhat affected by the addition of Fe-HBED, calcium and their interaction. Fusarium wilt of bananas was not affected by addition of iron chelates to these soils. The lack of effect was likely due to high iron availability in the soils overwhelming the capacity of the treatments to alter iron availability to the host plant and pathogen. Application of strong chelating ligands increased the concentration of aluminium and decreased the concentration of manganese in plant tissue, with possible detrimental effects.






Similar content being viewed by others
Data availability
Data is attached as an appendix.
References
ABGC (2017) The Australian Banana industry. Australia Banana Grower's Council. https://abgc.org.au/our-industry/key-facts/. Accessed 31 Jul 2017
Aznar A et al (2014) Scavenging iron: a novel mechanism of plant immunity activation by microbial siderophores. Plant Physiol 164:2167–2183. https://doi.org/10.1104/pp.113.233585
Bowen A, Orr R, McBeath AV, Pattison A, Nelson PN (2019) Suppressiveness or conduciveness to Fusarium wilt of bananas differs between key Australian soils. Soil Res. https://doi.org/10.1071/SR18159
Dita M, Barquero M, Heck D, Mizubuti ESG, Staver CP (2018) Fusarium wilt of banana: current knowledge on epidemiology and research needs toward sustainable disease management. Front Plant Sci 9. https://doi.org/10.3389/fpls.2018.01468
Dong X, Wang M, Ling N, Shen Q, Guo S (2016) Effects of iron and boron combinations on the suppression of Fusarium wilt in banana. Sci Rep 6:38944. https://doi.org/10.1038/srep38944
Duijff BJ, Bakker PAHM, Schippers B (1994) Suppression of Fusarium Wilt of Carnation by Pseudomonas putida WCS358 at Different Levels of Disease Incidence and Iron Availability. Biocontrol Sci Technol 4:279–288. https://doi.org/10.1080/09583159409355336
Elmer WH (2002) Influence of Inoculum Density of Fusarium oxysporum f. sp. cyclaminis and Sodium Chloride on Cyclamen and the Development of Fusarium Wilt. Plant Dis 86:389–393. https://doi.org/10.1094/pdis.2002.86.4.389
FAO (2018) Banana. Food and Agriculture Organization of the United Nations. http://www.fao.org/land-water/databases-and-software/crop-information/banana/en/. Accessed 30 Jan 2019
Jones JP, Woltz SS (1970) Fusarium wilt of tomato: interaction of soil liming and micronutrient amendments on disease development. Phytopathology 60:812–813. https://doi.org/10.1094/Phyto-60-812
Leeman M, Ouden FMd, Pelt JAv, Dirkx FPM, Steijl H, Bakker PAHM, Schippers B (1996) Iron availability affects induction of systemic resistance to Fusarium wilt of radish by Pseudomonas fluorescens. Phytopathology 86:149–155
Lemanceau P, Alabouvette C, Couteaudier Y (1988) Studies on the disease suppressiveness of soils. XIV. Modification of the receptivity level of a suppressive and a conducive soil to fusarium wilt in response to the supply of iron or of glucose. Agronomie 8:155–162. https://doi.org/10.1051/agro:19880209
López-Berges MS et al (2012) HapX-mediated iron homeostasis is essential for rhizosphere competence and virulence of the soilborne pathogen fusarium oxysporum. Plant Cell 24:3805–3822. https://doi.org/10.1105/tpc.112.098624
Marschner H, Römheld V (1995) Strategies of plants for acquisition of iron. In: Abadía J (ed) Iron Nutrition in Soils and Plants: Proceedings of the Seventh International Symposium on Iron Nutrition and Interactions in Plants, June 27–July 2, 1993, Zaragoza, Spain. Springer Netherlands, Dordrecht, pp 375–388. https://doi.org/10.1007/978-94-011-0503-3_52
Norvell W (1991) Reactions of metal chelates in soils and nutrient solutions. In: Mortvedt JJ (ed) Micronutrients in agriculture. SSSA Book Series. pp 187–227. https://doi.org/10.2136/sssabookser4.2ed.c7
O’Neill WT et al (2016) Detection of Fusarium oxysporum f. sp. cubense tropical race 4 strain in Northern Queensland. Australas Plant Dis Notes 11:33. https://doi.org/10.1007/s13314-016-0218-1
Orr R, Hocking RK, Pattison A, Nelson PN (2020) Extraction of metals from mildly acidic tropical soils: Interactions between chelating ligand, pH and soil type. Chemosphere 248:126060. https://doi.org/10.1016/j.chemosphere.2020.126060
Orr R, Nelson PN (2018) Impacts of soil abiotic attributes on Fusarium wilt, focusing on bananas. Appl Soil Ecol 132:20–33. https://doi.org/10.1016/j.apsoil.2018.06.019
Orr R, Pattison A, East D, Warman N, O’Neill W, Czislowski E, Nelson PN (2019) Image-based quantification of Fusarium wilt severity in banana. Australas Plant Dis Notes 14:14. https://doi.org/10.1007/s13314-019-0344-7
Peng HX, Sivasithamparam K, Turner DW (1999) Chlamydospore germination and Fusarium wilt of banana plantlets in suppressive and conducive soils are affected by physical and chemical factors. Soil Biol Biochem 31:1363–1374. https://doi.org/10.1016/S0038-0717(99)00045-0
D Reuter, JB Robinson (1997) Plant analysis: an interpretation manual. CSIRO Publishing Melbourne.https://doi.org/10.1071/9780643101265
Saikia R, Srivastava AK, Singh K, Arora DK, Lee M-W (2005) Effect of iron availability on induction of systemic resistance to Fusarium wilt of chickpea by Pseudomonas spp. Mycobiology 33:35–40. https://doi.org/10.4489/MYCO.2005.33.1.035
Schenkeveld WDC, Reichwein AM, Temminghoff EJM, van Riemsdijk WH (2007) The behaviour of EDDHA isomers in soils as influenced by soil properties. Plant Soil 290:85–102. https://doi.org/10.1007/s11104-006-9135-y
Schenkeveld WDC, Reichwein AM, Temminghoff EJM, van Riemsdijk WH (2014) Considerations on the shuttle mechanism of FeEDDHA chelates at the soil-root interface in case of Fe deficiency. Plant Soil 379:373–387. https://doi.org/10.1007/s11104-014-2057-1
Scher FM, Baker R (1982) Effect of Pseudomonas putida and a synthetic iron chelator on induction of soil suppressiveness to fusarium wilt pathogens. Ecol Epidemiol 72:1567–1573. https://doi.org/10.1094/Phyto-72-1567
Segarra G, Casanova E, Avilés M, Trillas I (2010) Trichoderma asperellum strain T34 controls Fusarium wilt disease in tomato plants in soilless culture through competition for iron. Micro Ecol 59:141–149. https://doi.org/10.1007/s00248-009-9545-5
Simeoni LA, Lindsay W, Baker R (1987) Critical iron level associated with biological control of Fusarium wilt. Phytopathology 77:1057–1061. https://doi.org/10.1094/PHYTO-77-1057
Weinert M, Simpson M (2016) Sub tropical banana nutrition - matching nutrition requirements to growth demands. NSW Department of Primary Industries, Wollongbar, NSW
Wickham H (2016) ggplot2: Elegeant graphics for data analysis. Springer-Verlag, New York. http://ggplot2.org
Ye F, Albarouki E, Lingam B, Deising HB, von Wirén N (2014) An adequate Fe nutritional status of maize suppresses infection and biotrophic growth of Colletotrichum graminicola. Physiol Plant 151:280–292. https://doi.org/10.1111/ppl.12166
Acknowledgements
We thank Wayne O’Neill from the Queensland Department of Agriculture and Fisheries for providing advice and inoculum. We also thank Daniel Browne, Harriet Allen, Kelly Sissons, Eldon O’Regan and Cecilie Brandsvoll for their help in the field, greenhouse and laboratory. Finally, we would like to thank Dr. Philippe Lemanceau for help at the start of this project.
This project was funded by Hort Innovation, using the Hort Innovation banana research and development levy, co-investment from Queensland Government and contributions from the Australian Government. Hort Innovation is the grower-owned, not-for-profit research and development corporation for Australian horticulture.
Funding
This project was funded by Hort Innovation, using the Hort Innovation banana research and development levy, co-investment from Queensland Government and contributions from the Australian Government. Hort Innovation is the grower-owned, not-for-profit research and development corporation for Australian horticulture.
Author information
Authors and Affiliations
Contributions
Conceptualization: All authors; Methodology: Ryan Orr, Paul Nelson; Formal analysis and investigation: Ryan Orr, Paul Nelson; Writing—original draft preparation: Ryan Orr; Writing—review and editing: Paul Nelson, Tobin Northfield, Anthony Pattison; Funding acquisition: Anthony Pattison, Paul Nelson; Supervision: Paul Nelson, Tobin Northfield.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Orr, R., Pattison, A., Northfield, T. et al. Iron chelates have little to no effect on the severity of Fusarium wilt of bananas in soils of the humid tropics. J Plant Pathol 103, 595–604 (2021). https://doi.org/10.1007/s42161-021-00816-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-021-00816-2