Abstract
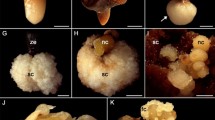
Present study was conducted to achieve intergeneric hybridization in Juglandaceae for the first time. Due to embryo abortion possibility, the embryo rescue technique was conducted via callus induction from immature hybrid embryo culture. For this reason, embryos were sampled at 20, 30, 40 and 60 days after pollination and cultured on two enrichment media (WPM and DKW). The developmental stage of immature embryo, kind of medium and addition of growth regulators to the induction medium played the role in promoting better callus and plantlet formation. The best quality and quantity of callus induction were achieved in 60 days’ explant. Maximum induction was in WPM and there was no callus induction in PGRs free medium. Browning was the most obstacle to conquer in callus proliferation. Germination of remained mature hybrid embryo until fruit harvest time was induced on three media (MS, WPM, and DKW). There were no significant differences between medium and male parents effect on germination parameters. But, MS and WPM performed better for hybrid embryo germination. The first germination was 7 days in MS and WPM and about 10 days for DKW after culture. In all medium the roots were robust and more developed than the leaves. The microplant transferred to soil after hardening.


Similar content being viewed by others
Notes
6‑Benzylaminopurin.
kinetine.
Indol 3‑butiric acid.
Gibberellic acid.
Abbreviations
- PGRs:
-
Plant Growth Regulators
References
Aftab F (2012) Progress and prospects for efficient micropropagation of woody plants. In: Ashraf M, Öztürk M, Ahmad MSA, Aksoy A (eds) Crop production for agricultural improvement. Springer, Dordrecht, pp 363–377
Breton C, Cornu D, Chriqui D, Sauvanet A, Capelli P, Germain E, Jay-Allemand C (2004) Somatic embryogenesis, micropropagation and plant regeneration of “Early Mature” walnut trees (Juglans regia) that flower in vitro. Tree Physiol 24(4):425–435. https://doi.org/10.1093/treephys/24.4.425
Cai X, Wang G, Cao W (2013) In vitro induction and proliferation of callus from immature cotyledons and embryos of Juglans regia cv. ‘Xiangling. Notulae Bot Hortic Agrobot 41(2):378–384. https://doi.org/10.15835/nbha4129134
Chumakov MI, Moiseeva EM (2012) Technologies of agrobacterium plant transformation in planta. Appl Biochem Biotech 48(8):657–666. https://doi.org/10.1134/S0003683812080017
Cornu D, Jay-Allemand C (1989) Micropropagation of Hybrid Walnut trees (Juglans nigra*Juglans regia) through culture and multiplication of embryos. Annu Sci For 46:113–116 (https://www.afs-journal.org/articles/forest/pdf/1989/05/AFS_0003-4312_1989_46_Suppl_ART0023.pdf)
Corte-Olivares J, Phillips GC, Butler-Nance SA (1990) Somatic embryogenesis from Pecan zygotic embryo explants. Hort Sci (Prague) 25(8):983
Davey MR, Anthony P, Power JB, Lowe KC (2005) Plant protoplasts: status and biotechnological perspectives. Biotechnol Adv 23(2):131–171. https://doi.org/10.1016/j.biotechadv.2004.09.008
Farsi M, Vahdati K, Lotfi M, Mirmasoumi M (2012) Open the windows toward somatic embryogenesis of leaf explants of persian walnut (Juglans regia L.). Int J Nuts Relat Sci 3(1):13–22 (https://www.cabdirect.org/cabdirect/abstract/20123239887)
Fatima A, Kamili AN, Shah AM (2006) Plantlet regeneration from excised embryonal axes, shoot apices and nodal segments of Juglans regia L. Acta Hortic 705:387–392. https://doi.org/10.17660/ActaHortic.2005.705.55
Fernández H, Pérez C, Sánchez-Tamés R (2000) Modulation of the morphogenic potential of the embryonic axis of Juglans regia by cultural conditions. Plant Growth Regul 30(2):125–131. https://doi.org/10.1023/A:1006329111046
Grauke LJ, O’Barr RD (1996) Initial survival of pecan grafts on seedling rootstock of pecan, water hickory, and their interspecific hybrid. Hort Technol 6(1):45–48
Haroon A (2010) Propagation of pecan (Carya illinoensis) using in vitro techniques. Lahore. PhD Thesis, department of Botany, University of the Punjab, p 222 (http://prr.hec.gov.pk/jspui/handle/123456789//1309)
Heile-Sudholt C, Huetteman CA, Preece JE, Van Sambeek JW, Gaffney GR (1986) In vitro embryonic axis and seedling shoot tip culture of Juglans nigra L. Plant Cell Tiss Organ Cult 6:189–197. https://doi.org/10.1007/BF00180804
Kaur R, Sharma N, Kumar K, Sharma DR, Sharma SD (2006) In vitro germination of walnut (Juglans regia L.) embryos. Sci Hortic 109:385–388. https://doi.org/10.1016/j.scienta.2006.05.012
Kornova K, Stephanova A, Terzijsky D (1993) In vitro culture of immature embryos and cotyledons of Juglans regia L. Morphological and anatomical analysis of some regenerants. Acta Hortic 311:125–133. https://doi.org/10.1080/13102818.1993.10818737
Long LM, Preece JE, Van Sambeek W (1995) Adventitious regeneration of Juglans nigra L. (Eastern black walnut). Plant Cell Rep 14:799–803. https://doi.org/10.1007/BF00232926
Mapelli S, Brambilla I, Bertani A (2001) Free amino acids in walnut kernels and young seedlings. Tree Physiol 21:1299–1302. https://doi.org/10.1093/treephys/21.17.1299
McGranahan GH, Tulecke W, Arulsekar S, Hansen JJ (1986) Intergeneric hybridization in the Juglandaceae: Pterocarya sp.* Juglans regia. J Am Soc Hortic Sci 111:627–630 (http://agris.fao.org/agris-search/search.do?recordID=US8706048)
Neuman MC, Preece JE, Van Sambeek JW, Gaffney GR (1993) Somatic embryogenesis and callus production from cotyledon explants of Eastern black walnut. Plant Cell Tiss Organ Cult 32:9–18. https://doi.org/10.1007/BF00040110
Payghamzadeh K, Kazemitabar SK (2010) In vitro germination of Pecan (Carya illinoinensis) embryo. Biharean Biol 4(1):37–43 (http://biologie-oradea.xhost.ro/BihBiol/index.html)
Payghamzadeh K, Kazemitabar SK, Amiri A (2011) Effects of two type of medium cultures, Gibberellic acid and some physical factors on Persian walnut (Juglans regia L.) embryos germination. J Hortic Sci 25(1):45–54. https://doi.org/10.5897/AJB10.324
Rodriguez APM, Wetzstein HY (1994) The effect of auxin type and concentration on pecan (Carya illinoinensis) somatic embryo morphology and subsequent conversion into plants. Plant Cell Rep 13:607–611. https://doi.org/10.1007/BF00232930
Rodriguez APM, Wetzstein HY (1998) A morphological and histological comparison of the initiation and development of Pecan (Carya illinoensis) somatic embryogenic cultures induced with naphthaleneacetic acid or 2,4-dichlorophenoxyacetic acid. Protoplasma 204(1–2):71–83. https://doi.org/10.1007/BF01282295
Saadat YA, Hennerty MJ (2002) Factors affecting the shoot multiplication of Persian walnut (Juglans regia L.). Sci Hortic 95:251–260. https://doi.org/10.1016/S0304-4238(02)00003-1
Sahijram L, Madhusudhana Rao B (2015) Plant genomics and biotechnology. Hybrid embryo rescue in crop improvement. Plant biology and biotechnology, vol 2. Springer India, , pp 363–384
Sahijram L, Soneji JR, Naren A, Madhusudhana Rao B (2013) Hybrid embryo rescue: a non-conventional breeding strategy in horticultural crops. J Hortic Sci 8(1):1–20 (http://www.sphindia.org/index.php/jhs/article/view/93/93)
San B, Dumanoglu H (2006) Somatic embryogenesis from immature cotyledons of apomictic and non-apomictic seeds in walnut (Juglans regia L.). Turk J Agric For 30(2):111–117 (http://journals.tubitak.gov.tr/agriculture/abstract.htm?id=8169)
Sanchez-Zamora MA, Cos-Terrer J, Frutos-Tomas D, Garcia-Lopez R (2006) Embryo germination and proliferation in vitro of Juglans regia L. Sci Hortic 108(3):317–321. https://doi.org/10.1016/j.scienta.2006.01.041
Shen X, Gmitter FG Jr, Grosser JW (2011) Immature embryo rescue and culture. In: Thorpe TA, Yeung EC (eds) Plant embryo culture: methods and protocols, methods in molecular biology, vol 710. Springer, , pp 75–92
Stone DE, Adrouny GA, Flake RH (1969) New world Juglandaceae. II. Hickory nut oils, phenetic similarities, and evolutionary implications in the genus Carya. Am J Bot 56(8):928–935. https://doi.org/10.1002/j.1537-2197.1969.tb09743.x
Tang W, Newton RJ (2005) Peroxidase and catalase activities are involved in direct adventitious shoot formation induced by thidiazuron in eastern white pine (Pinus strobus L.) zygotic embryos. Plant Physiol Biochem 43(8):760–769. https://doi.org/10.1016/j.plaphy.2005.05.008
Tang H, Zhenglong R, Gabi K (2000) Improvement of English walnut somatic embryo germination and conversion by desiccation treatments and plantlet development by lower medium salt. Vitro Cell Dev Biol Plant 36:47–50. https://doi.org/10.1007/s11627-000-0011-9
Toosi S, Dilmaghani K, Hikmatshoar H (2010) Proliferation of Juglans regia L. by in vitro embryo culture. Middle East J Sci Res 6(1):1–7 (https://academicjournals.org/journal/JCBG/article-full-text-pdf/797C9B1839)
Tulecke W, McGranahan GH (1985) Somatic embryogenesis and plant regeneration from cotyledons of walnut, Juglans regia L. Plant Sci 40:57–63. https://doi.org/10.1016/0168-9452(85)90163-3
Tulecke W, McGranahan G, Ahmadi H (1988) Regeneration by somatic embryogenesis of triploid plants from endosperm of walnut, Juglans regia L. cv Manregian. Plant Cell Rep 7(5):301–304. https://doi.org/10.1007/BF00269923
Vahdati K, Jariteh M, Niknam V, Mirmasoumi M, Ebrahimzadeh H (2006) Somatic embryogenesis and embryo maturation in Persian walnut. Acta Hortic 705:199–205. https://doi.org/10.17660/ActaHortic.2005.705.24
Von Arnold S, Sabala I, Bozhkov P, Dyachok J, Filonova L (2002) Developmental pathways of somatic embryogenesis. Plant Cell Tiss Organ Cult 69(3):233–249. https://doi.org/10.1023/A:1015673200621
Wetzstein HY, Merckle SA, Ault JR (1990) Factors influencing somatic embryogenesis and plantlet regeneration in Pecan, Carya illinoensis. Acta Hortic. https://doi.org/10.17660/ActaHortic.1990.280.9
Woeste K (2002) Heartwood production in a 35-year old Black walnut progeny test. Can J Forest Res 32:177–181. https://doi.org/10.1139/x06-130
Yates IE, Reilly CC (1990) Hort Sci (Prague) 25(5):573–576
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. Ghazaeian, G. H. Davarynejad, K. G. Bezdi and H. Nemati declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Ghazaeian, M., Davarynejad, G.H., Bezdi, K.G. et al. Embryo Culture of New Hybrid of Pecan (Carya Illinoensis) with Walnut (J. Regia and J. Nigra). Erwerbs-Obstbau 63, 185–192 (2021). https://doi.org/10.1007/s10341-021-00556-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-021-00556-8