Abstract
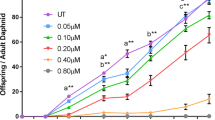
Cytostatics are compounds used in chemotherapy, known to be genotoxic, mutagenic, and teratogenic at low concentrations. The amount of cytostatic drugs prescribed increases every year as does their release into the aquatic ecosystems, which possibly is a major concern for the health of aquatic organisms. This study aimed to evaluate the putative toxicity of five cytostatics to fathead minnow (Pimephales promelas) larvae: tamoxifen, capecitabine, methotrexate, cyclophosphamide, and ifosfamide. Eggs collected post-fertilization were exposed for 6 days to a range of concentrations, including one above environmental level. At all environmental concentrations, no significant difference in mortality, hatching time, length, heart rate, and presence of malformations were found. Altogether, these cytostatics do not seem embryotoxic to fish. Although, an increased proportion of complete swim bladder were found after ifosfamide’s exposure, suggesting an interaction with the thyroid axis, involved in swim bladder development. Complementary work should address other endpoints, such as behavioral changes, reproductive success, and transgenerational effects.
Similar content being viewed by others
References
Alsaadi F, Madison BN, Brown RS, Hodson PV, Langlois VS (2018) Morphological and molecular effects of two diluted bitumens on developing fathead minnow (Pimephales promelas). Aq Tox. 204:107–116. https://doi.org/10.1016/j.aquatox.2018.09.003
Alsop D, Vijayan M (2009) The zebrafish stress axis: molecular fallout from the teleost-specific genome duplication event. Gen Comp Endocrinol 161:62–66 https://doi.org/10.1016/j.ecoenv.2015.05.036
Brun GL, Bernier M, Losier R, Doe K, Jackman P, Lee HB (2006) Pharmaceutical active compounds in Atlantic Canadian sewage treatment plant effluents and receiving waters, and potential for environmental effects as measured by acute and chronic aquatic toxicity. Environ Toxicol Chem 25(8):2163–2176. https://doi.org/10.1016/j.ygcen.2008.09.011
Borgatta M, Waridel P, Decosterd LA, Buclin T, Chèvre N (2016) Multigenerational effects of the anticancer drug tamoxifen and its metabolite 4-hydroxy-tamoxifen on Daphnia pulex. Sci Total Environ 545–546:21–29. https://doi.org/10.1016/j.scitotenv.2015.11.155
Cavallin JE, Ankley GT, Blackwell BR et al (2017) Impaired swim bladder inflation in early life stage fathead minnows exposed to a deiodinase inhibitor, iopanoic acid. Environ Toxicol Chem 36(11):2942–2952. https://doi.org/10.1002/etc.3855
Delvin EW, Brammer JD, Puycar RL, McKim JM (1996) Prehatching development of the Fatead Minnow Pimephales Promelas Rafinesque. U.S. Environmental Protection agency (EPA), Office of Research Development, National Health and Environmental Effects Research Laboratory.
DellaGreca M, Iesce MR, Isidori M, Nardelli A, Previtera L, Rubino M (2007) Phototransformation products of tamoxifen by sunlight in water. Toxicity of the drug and its derivatives on aquatic orgnisms. Chemosphere 67:1933–1939. https://doi.org/10.1016/j.chemosphere.2006.12.001
FASS (2011) The Swedish medicines information engine. Läkemedelsfakta: Miljöinformation Access. Stockholm (Sweden): FASS. Accessed 22 Oct 2020 www.fass.se/LIF/produktfakta/artikel_produkt.jsp?NplID=19860829000074&DocTypeID=78#IDE4POEFUA6FBVERT1
FDA (2012) Highlights of prescribing information – IFEX. Access on dec 2020. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/019763s017lbl.pdf
Ferrando-Climent L, Rodriguez-Mozaz S, Barceló D, (2014) Incidence of anticancer drugs in an aquatic urban system: from hospital effluents through urban wastewater to natural environment. Environ Pollut 193:216–223. https://doi.org/10.1016/j.envpol.2014.07.002
Franquet-Griell H, Medina A, Sans C, Lacorte S (2017) Biological and photochemical degradation of cytostatic drugs under laboratory conditions. J. Hazard. Mater. 323(A):319–328 https://doi.org/10.1016/j.jhazmat.2016.06.057
Godfrey A, Hooser B, Abdelmoneim A, Horzmann KA (2017) Thyroid disrupting effects of halogenated and next generation chemicals on the swim bladder development of zebrafish. Aquat Toxicol 193:228–235. https://doi.org/10.1016/j.aquatox.2017.10.024
Henschel KP, Wenzel A, Diedrich M, Fliedner A (1997) Environmental hazars assessment of pharmaceuticals. Regul Toxicol Pharm 25:220–225. https://doi.org/10.1006/rtph.1997.1102
Hoppe-Tichy T, (2010) Current challenges in European oncology pharmacy practice. J Oncol Pharm Pract 16:9–18. https://doi.org/10.1177/1078155209354346
Johnson AC, Oldenkamp R, Dumont E, Sumpter JP (2013) Predicting concentrations of the cytostatic drugs cyclophosphamide, carboplatin, 5-fluorouracil, and capecitabine throughout the sewage effluents and surface waters of Europe. Environ Toxicol Chem 32(9):1954–1961. https://doi.org/10.1002/etc.2311
Kosjek T, Heath E (2011) Occurrence, fate and determination of cytostatic pharmaceuticals in the environment. Trends Anal Chem 30(7):1065–1087. https://doi.org/10.1016/j.trac.2011.04.007
Madison BN, Wallace SJ, Zhang J, Hodson LVS (2020) Transcriptional responses in newly-hatched Japanese medaka (Oryzias latipes) associated with developmental malformations following diluted bitumen exposure. Comp Biochem Physio Part D. https://doi.org/10.1016/j.cbd.2020.100685
Madison BN, Hodson PV, Langlois VS (2017) The effects of Cold Lake Blend diluted bitumen toxicity on the early development of Japanese medaka (Oryzias latipes). Environ Poll 225:579–586. https://doi.org/10.1016/j.envpol.2017.03.025
Negreira N, López de Alda M, Barceló D (2014) Cytostatic drugs and metabolites in muniicpal and hospital wastewaters in Spain: filtration, occurrence, and environmental risk. Sci Total Environ 497:68–77. https://doi.org/10.1016/j.scitotenv.2014.07.101
Nelson KR, Schroeder AL, Ankley GT et al (2016) Impaired anterior swim bladder inflation following exposure to the thyroid peroxidase inhibitor 2-mercaptobenzothiazole par I: fathead minnow. Aquat Toxicol 173:192–203. https://doi.org/10.1016/j.aquatox.2015.12.024
Novak M, Žegura B, Modic B, Heath E, Filipič M (2017) Cytotoxicity and genotoxicity of anticancer drugs residues and their mixtures in experimental model with zebrafish liver cells. Sci Total Environ 601:293–300. https://doi.org/10.1016/j.scitotenv.2017.05.115
Nussbaumer S, Bonnabry P, Veuthey JL, Fleury-Souverain S (2011) Analysis of anticancer drugs: a review. Talanta 85:2265–2289. https://doi.org/10.1016/j.talanta.2011.08.034
Organisation for Economic Cooperation and Development (OECD) (1998) Test No. 212: Fish, Short-term Toxicity Test on Embryo and Sac-Fry Stages, OECD Guidelines for the Testing of Chemicals, Section 2, Éditions OCDE, Paris. https://doi.org/10.1787/9789264070141-en.
Statistics Canada (2020) Table 13-10-0394-01 Leading causes of death, total population, by age group. https://doi.org/10.25318/1310039401-eng
Stephen SM, Alkindi AYA, Waring CP, Brown JA (1997) Corticosteroid and thyroid responses of larval and juvenile turbot exposed to the water-soluble fraction of crude oil. J Fish Biol 50:953–964. https://doi.org/10.1111/j.1095-8649.1997.tb01621.x
Straub JO (2010) Combined environmental risk assessment for 5-fluorouracil and capecitabin in Europe. Integr Environ Assess Manag 6:540–566. https://doi.org/10.1897/IEAM_2009-073.1
Sun L, Zha J, Spear PA, Wang Z (2007) Tamoxifen effects on early life stages and reproduction of Japanese medaka (Oryzias latipes). Environ Toxicol Pharm 24:23–29. https://doi.org/10.1016/j.etap.2007.01.003
Tadic D, Bozovic-Spasojevic I, Tomasevic ZI, Djukic-Dejanovic S (2015) Oral administration of antineoplastic agents: the challenges for healthcare professionals. JBUON 20(3):690–698
Van der Ven LTM, Van den Brandhof EJ, Vos JH, Wester PW (2007) Effects of the estrogen agonist 17β-estradiol and antagonist tamoxifen in a partial life-cycle assay with zebrafish (Danio rerio). Environ Toxicol Chem 26(1):92–99. https://doi.org/10.1897/06-092R1.1
Weigt S, Huebler N, Strecker R, Braunbeck T, Broscherd TH (2011) Zebrafish (Danio rerio) embryos as a model for testing proteratogens. Toxicol 281:25–36. https://doi.org/10.1016/j.tox.2011.01.004
Wester PW, Van der Ven LTM, Van den Brandhof EJ, Vos JH (2003) Identification of endocrine disruptive effects in the aquatic environment: a partial life cycle study in Zebrafish. Report 640920. RIVM, the Netherlands, 112p
Zhang J, Chang VWC, Giannis A, Wang JY (2013) Removal of cytostatic drugs from aquatic environment: a review. Sci Total Environ 445:281–298. https://doi.org/10.1016/j.scitotenv.2012.12.061
Acknowledgments
The authors would like to thanks Tuan Anh To and Scott Hepditch for helping with exposure and sampling. This project was funded by the St. Lawrence Action Plan to NP and GTB, by the Network of Ecotoxicologists of Quebec (EcotoQ) to VSL and NP, and by a Canada Research Chair (CRC) to VSL (#950-232235).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lefebvre-Raine, M., Paquet, N., Triffault-Bouchet, G. et al. Embryotoxicity of Five Cytostatics in Fathead Minnow (Pimephales promelas) Larvae. Bull Environ Contam Toxicol 106, 747–752 (2021). https://doi.org/10.1007/s00128-021-03146-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-021-03146-2