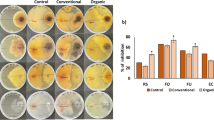
Abstract
Monosporascus root rot and vine decline of melon (MRRVD) is a destructive disease complex mainly occurring in semiarid cultivation areas. In the last decade, in the melon producing area of Sardinia (Italy), yield reductions up to 100% were recorded due to the occurrence of MRRVD. The present study aimed to undertake a two-year survey of fungal pathogens associated with MRRVD grown as a monocrop in two locations in Central Sardinia, Sinis, and Sassu, and to investigate the possible role of soil fungal diversity in the disease development. Melon plants were affected by similar symptoms but colonized by a set of soil-borne fungal pathogens different between surveyed sites and cropping seasons, including Plectosphaerella melonis, P. cucumerina, Fusarium solani, Macrophomina phaseolina and Monosporascus cannonballus. Olpidium bornovanus and O. virulentus were isolated using bait plants and detected by NGS analysis. Bait plants had a general decrease in biomass and yellowing of foliage. The presence of most of the isolated pathogens was also confirmed by the NGS analysis of the soil microbiome. Our results confirm that among the fungal complex implicated in the occurrence of RRVD in Sardinia, O. bornovanus along with O. virulentus likely assume a key role in the development of the disease alone and possibly in association with other pathogens. Differences in the pathogenic fungal spectrum here recorded in symptomatic roots may be associated with agricultural practices, soil physicochemical characteristics, and fungal community composition and function profile.



Similar content being viewed by others
References
Aleandri MP, Martignoni D, Reda R, Alfaro-Fernandez A, Font MI, Armengol J, Chilosi G (2014) First report of Olpidium bornovanus and O. virulentus on melon in Italy. Plant Dis 98:997. https://doi.org/10.1094/PDIS-10-13-1041-PDN
Aleandri MP, Martignoni D, Reda R, Chilosi G (2015a) Effects of preconditioning through mycorrhizal inoculation on the control of melon root rot and vine decline caused by Monosporascus cannonballus. J Phytopathol 163:898–907. https://doi.org/10.1111/jph.12389
Aleandri MP, Chilosi G, Bruni N, Vettraino TA, AM, Vannini A, (2015b) Use of nursery potting mixes amended with local Trichoderma strains with multiple complementary mechanisms to control soil-borne diseases. Crop Prot 67:269–278. https://doi.org/10.1016/j.cropro.2014.10.023
Aleandri MP, Martignoni D, Reda R, Alfaro-Fernandez A, Font MI, Armengol J, Chilosi G (2017) Involvement of Olpidium bornovanus and O. virulentus in the occurrence of melon root rot and vine decline caused by Monosporascus cannonballus in Central Italy. J Plant Pathol 99:169–176. https://doi.org/10.4454/jpp.v99i1.3787
Alfaro-Garcìa A, Armengol J, Bruton BD, Gams W, Garcìa-Jimenez J, Martinez-Ferrer G (1996) The taxonomic position of the causal agent of Acremonium collapse of muskmelon. Mycologia 88:804–808. https://doi.org/10.1080/00275514.1996.12026718
Balmas V, Infantino A, Chilosi G, Siddu G, Murru S (2016) Collasso delle cucurbitacee, diffusione e difesa in Sardegna. L’Informatore Agrario 18:53–56
Beale RE, Pitt D (1995) The antifungal properties of Minimedusa polyspora. Mycol Res 99:337–342. https://doi.org/10.1016/S0953-7562(09)80910-6
Beltrán R, Vicent A, Garcia-Jiménez J, Armengol J (2007) Quantification of Monosporascus cannonballus ascospores in muskmelon fields in Eastern Spain. J Phytopathol 155:248–250. https://doi.org/10.1111/j.1439-0434.2007.01215.x
Beltrán R, Vicent A, García-Jiménez J, Armengol J (2008) Comparative epidemiology of Monosporascus root rot and vine decline in muskmelon, watermelon, and grafted watermelon crops. Plant Dis 92:158–163. https://doi.org/10.1094/PDIS-92-1-0158
Ben Salem I, Correia KC, Boughalleb N, Michereff SJ, León M, Abad-Campos P, García-Jiménez J, Armengol J (2013) Monosporascus eutypoides, a cause of root rot and vine decline in Tunisia, and evidence that M. cannonballus and M. eutypoides are distinct species. Plant Dis 97:737–743. https://doi.org/10.1094/PDIS-05-12-0464-RE
Berendsen RL, Pieterse CM, Bakker PA (2012) The rhizosphere microbiome and plant health. Trends in Plant Sci 17:478–486. https://doi.org/10.1016/j.tplants.2012.04.001
Boughalleb N, Salem IB, Beltrán R, Vicent A, Sierra AP, Abad-Campos P, Garcìa-Imenez J, Armengol J (2010) Occurrence of Monosporascus cannonballus in watermelon fields in Tunisia and factors associated with ascospore density in soil. J Phytopathol 158:137–142. https://doi.org/10.1111/j.1439-0434.2009.01591.x
Bruton BD, Garcia-Jimenez J, Armengol J, Popham TW (2000) Assessment of virulence of Acremonium cucurbitacearum and Monosporascus cannonballus on Cucumis melo. Plant Dis 84:907–913. https://doi.org/10.1094/PDIS.2000.84.8.907
Carlucci A, Raimondo ML, Frisullo S (2006) Different morphotypes of Plectosporium tabacinum associated with collapse of cucurbit in Apulia. J Plant Pathol 88, S36
Carlucci A, Raimondo ML, Santos J, Phillips AJL (2012) Plectosphaerella species associated with root and collar rots of horticultural crops in southern Italy. Persoonia: Molecular Phylogeny and Evolution of Fungi 28:34. https://doi.org/10.3767/003158512X638251
Chiellini C, Cardelli V, De Feudis M, Corti G, Cocco S, Agnelli A, Massaccesi L, Donato Alessi G, Mengoni A, Mocali S (2019) Exploring the links between bacterial communities and magnetic susceptibility in bulk soil and rhizosphere of beech (Fagus sylvatica L.). App Soil Ecol 138:69–79. https://doi.org/10.1016/j.apsoil.2019.02.008
Chilosi G, Reda R, Aleandri MP, Camele I, Altieri L, Montuschi C, Languisco L, Rossi V, Agosteo GE, Macrì C, Carlucci A, Lops F, Mucci M, Raimondo ML, Frisullo S (2008) Fungi associated with root rot and collapse of melon in Italy. EPPO Bull 38:147–154. https://doi.org/10.1111/j.1365-2338.2008.01200.x
Clement JA, Dombrowski AD, Sigmund JM, Goetz MA (2016) Occurrence of Podosporin B In Podospora decipiens and Sordaria araneosa. Planta Med 82:PC25. https://doi.org/10.1055/s-0036-1578727
Cohen R, Pivonia S, Burger Y, Edelstein M, Gamliel A, Katan J (2000) Toward integrated management of Monosporascus wilt of melons in Israel. Plant Dis 84:496–505. https://doi.org/10.1094/PDIS.2000.84.5.496
Cohen R, Pivonia S, Crosby KM, Martyn RD (2012) Advances in the biology and management of Monosporascus vine decline and wilt of melons and other cucurbits. Hortic Rev 39:77–120. https://doi.org/10.1002/9781118100592.ch2
Crecchio C, Mimmo T, Bulgarelli D, Pertot I, Pii Y, Perazzolli M, Scagliola M, Cesco S (2018) Beneficial Soil Microbiome for Sustainable Agriculture Production. In: Lichtfouse E (ed) Sustainable Agriculture Reviews 31. Springer, Cham, pp 443–481. https://doi.org/10.1007/978-3-319-94232-2_9
De Cara M, López V, Córdoba MC, Santo M, Jordá C, Tello JC (2008) Association of Olpidium bornovanus and melon necrotic spot virus with vine decline of melon in Guatemala. Plant Dis 92:709–713. https://doi.org/10.1094/PDIS-92-5-0709
de Hoog GS, Smith MT, Guého E (1986) A revision of the genus Geotrichum and its teleomorphs. Stud Mycol 29:1–131
El-Desouky SM, El-Wakil AA (2003) Occurrence of Monosporascus root rot and vine decline of cantaloupe and watermelon in Egypt. Egypt J Phytopathol 31:141–150
Falk SP, Gadoury DM, Seem PRC, RC, (1995) Partial control of grape powdery mildew by the mycoparasite Ampelomyces quisqualis. Plant Dis 79:483–490. https://doi.org/10.1094/PD-79-0483
Frąc M, Hannula SE, Bełka M, Jędryczka M (2018) Fungal biodiversity and their role in soil health. Front Microbiol. https://doi.org/10.1007/s42161-019-00314-6
García-Jiménez J, Velázquez MT, Jorda C, Alfaro-Garcia A (1994) Acremonium species as the causal agent of muskmelon collapse in Spain. Plant Dis 78:416–419
García-Jiménez J, Armengol J, Sales R, Jordá C, Bruton BD (2000) Fungal pathogens associated with melon collapse in Spain. EPPO Bull 30:169–173. https://doi.org/10.1111/j.1365-2338.2000.tb00873.x
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118. https://doi.org/10.1111/j.1365-294X.1993.tb00005.x
Herrera-Vásquez JÁ, del Carmen CM, Alfaro-Fernández A, del Carmen C-S, Jordá C (2009) Multiplex PCR assay for the simultaneous detection and differentiation of Olpidium bornovanus, O. brassicae, and O. virulentus. Mycol Res 113:602–610. https://doi.org/10.1016/j.mycres.2009.01.007
Herrera-Vásquez JÁ, Córdoba-Sellés MC, Cebrián M, Rosselló JA, Alfaro-Fernández A, Jordá C (2010) Genetic diversity of Melon necrotic spot virus and Olpidium isolates from different origins. Plant Pathol 59:240–251. https://doi.org/10.1111/j.1365-3059.2009.02208.x
Infantino A, Carlucci A, Pucci N, Ciuffreda G, Montuschi C, Lops F, Uccelletti A, Mucci M, Frisullo S (2004) Funghi che causano marciumi radicali e il collasso delle cucurbitacee in Italia. Petria 14:77–89
ISTAT (Istituto Nazionale di Statistica, Italy) 2020. http://dati.istat.it/Index.aspx?QueryId=33703#
Jager G, Velvis H (1984) Biological control of Rhizoctonia solani on potatoes by antagonists. 2. Sprout protection against soil-borne R. solani through seed inoculation with Verticilium biguttatum. Neth J Plant Pathol 90:29–33. https://doi.org/10.1007/BF02014180
Johnston-Monje D, Loewen S, Lazarovits G (2017) Mycobiomes of tomato plants with vine decline. Can J Plant Pathol 39:184–200. https://doi.org/10.1080/07060661.2017.1325938
Lauber CL, Strickland MS, Bradford MA, Fierer N (2008) The influence of soil properties on the structure of bacterial and fungal communities across land-use types. Soil Biol Biochem 40:2407–2415. https://doi.org/10.1016/j.soilbio.2008.05.021
Lewis JA, Papavizas GC (1993) Stilbella aciculosa: a potential biocontrol fungus against Rhizoctonia solani. Biocontrol Sci Techn 3:3–11. https://doi.org/10.1080/09583159309355253
Libohova Z, Seybold C, Wysocki D, Wills S, Schoeneberger P, Williams C, Lindbo D, Stott D, Owens PR (2018) Reevaluating the effects of soil organic matter and other properties on available water-holding capacity using the National Cooperative Soil Survey Characterization Database. J Soil Water Conserv 73:411–421. https://doi.org/10.2489/jswc.73.4.411
Lieckfeldt E, Kullnig CM, Kubicek CP, Samuels GJ, Börner T (2001) Trichoderma aureoviride: phylogenetic position and characterization. Mycol Res 105:313–322. https://doi.org/10.1017/S0953756201003616
Lo Piccolo S, Alfonzo A, Giambra S, Conigliaro G, Lopez-Llorca LV, Burruano S (2015) Identification of Acremonium isolates from grapevines and evaluation of their antagonism towards Plasmopara viticola. Ann Microbiol 65:2393–2403. https://doi.org/10.1007/s13213-015-1082-5
Kim YK, Kim TS, Shim HS, Park KS, Yeh WH, Hong SJ, Shim C, Kim J, Jong-Ho Park J, Han E, Lee MH, Hyeong-Jin J (2011) First report of sour rot on post-harvest oriental melon, tomato, cucumber, potato, pumpkin and carrot caused by Geotrichum candidum. Res Plant Dis 17:232–234. https://doi.org/10.5423/RPD.2011.17.2.232
Maccarone LD (2013) Relationships between the pathogen Olpidium virulentus and viruses associated with lettuce Big-Vein Disease. Plant Dis 97:700–707. https://doi.org/10.1094/PDIS-10-12-0979-FE
Manici LM, Caputo F, Cerato C (1995) Temperature responses of isolates of Macrophomina phaseolina from different climatic regions of sunflower production in Italy. Plant Dis 79:934–938
Markakis EA, Trantas EA, Lagogianni CS, Mpalantinaki E, Pagoulatou M, Ververidis FN, Goumas DE (2017) First report of root rot and vine decline of melon caused by Monosporascus cannonballus in Greece. Plant Dis 102:1036. https://doi.org/10.1094/PDIS-10-17-1568-PDN
Martyn RD, Miller ME (1996) Monosporascus root rot and vine decline: An emerging disease of melons worldwide. Plant Dis 80:716–725. https://doi.org/10.1094/PD-80-0716
Nilsson RH, Anslan S, Bahram M, Wurzbacher C, Baldrian P, Tedersoo L (2019) Mycobiome diversity: high-throughput sequencing and identification of fungi. Nat Rev Microbiol 17:95–109. https://doi.org/10.1038/s41579-018-0116-y
Pivonia S, Cohen R, Kafkafi U, Ze’ev IB, Katan J (1997) Sudden wilt of melons in southern Israel: fungal agents and relationship with plant development. Plant Dis 81:1264–1268. https://doi.org/10.1094/PDIS.1997.81.11.1264
Rhouma A, Salem IB, M’hamdi M, Boughalleb-M’Hamdi N, (2019) Relationship study among soils physico-chemical properties and Dawn Brooks ascospores densities for cucurbit fields in Tunisia. Eur J Plant Pathol 153:65–78. https://doi.org/10.1007/s10658-018-1541-5
Scervino JM, Papinutti VL, Godoy MS, Rodriguez MA, Della Monica I., Recchi M., Pettinari MJ, Godeas AM (2011) Medium pH, carbon and nitrogen concentrations modulate the phosphate solubilization efficiency of Penicillium purpurogenum through organic acid production. J Appl Microbiol 110:1215–1223
Schianchi N, Oggiano L, Chilosi G, Balmas V (2019) First report of Olpidium bornovanus and O. virulentus on watermelon in Sardinia. Italy J Plant Pathol 101:1253. https://doi.org/10.1007/s42161-019-00314-6
Schianchi N, Oufensou S, Porqueddu G, Chilosi G, Balmas V (2020) First report of Olpidium virulentus, O. bornovanus, O. brassicae on cucumber in Sardinia, Italy. J Plant Pathol 1–1 https://doi.org/10.1007/s42161-020-00605-3
Schumacher T (1993) Ecology and distribution of the genus Scutellinia in Norway. Arct Alp Mycol 3:215–233
Schoch CL, Seifert KA, Huhndorf S, Robert V, Spouge JL, Levesque CA, Chen W (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc Natl Acad Sci 109:6241–6246. https://doi.org/10.1073/pnas.1117018109
Scholler M, Rubner A (1999) Arthrobotrys hertziana sp. nov. from the Canary Islands. Mycol Res 103:764–768. https://doi.org/10.1017/S095375629800759X
Segata N, Izard J, Waldron L, Gevers D, Miropolsky L, Garrett WS, Huttenhower C (2011) Metagenomic biomarker discovery and explanation. Genome Biol 12:R60. https://doi.org/10.1186/gb-2011-12-6-r60
Smalla K, Wieland G, Buchner A, Zock A, Parzy J, Kaiser S, Roskot N, Heuer H, Berg G (2001) Bulk and rhizosphere soil bacterial communities studied by denaturing gradient gel electrophoresis: plant-dependent enrichment and seasonal shifts reveale. Appl Environ Microbiol 67:4742–4751. https://doi.org/10.1128/AEM.67.10.4742-4751.2001
Srivathsan A, Baloğlu Wang W, Tan WT, Bertrand D, Ng AHQ, Boey EJH, Koh JJY, Nagarajan N, Meier R (2018) A MinIONTM-based pipeline for fast and cost-effective DNA barcoding. Mol Eco Resour 18:1035–1049. https://doi.org/10.1111/1755-0998.12890
Stanghellini ME, Rasmussen SL (1992) A quantitative method for the recovery of ascospores of Monosporascus cannonballus from field soil. Phytopathology 82:1115
Stanghellini ME, Mathews DM, Misaghi IJ (2010) Pathogenicity and management of Olpidium bornovanus, a root pathogen of melons. Plant Dis 94:163–166. https://doi.org/10.1094/PDIS-94-2-0163
Stanghellini ME, Misaghi IJ (2011) Olpidium bornovanus-mediated germination of ascospores of Monosporascus cannonballus: a host-specific rhizosphere interaction. Phytopathology 101:794–796. https://doi.org/10.1094/PHYTO-11-10-0313
Stanghellini ME, Mohammadi M, Adaskaveg JE (2014) Effect of soil matric water potentials on germination of ascospores of Monosporascus cannonballus and colonization of melon roots by zoospores of Olpidium bornovanus. Eur J Plant Pathol 139:393–398. https://doi.org/10.1007/s10658-014-0395-8
Thornton CR, Slaughter CD, Davis RM (2010) Detection of the sour-rot pathogen Geotrichum candidum in tomatoes fruit and juice by using a highly specific monoclonal antibody-based ELISA. Int J Food Microbiol 43:166–172. https://doi.org/10.1016/j.ijfoodmicro.2010.08.012
Tian XL, Wang CB, Bao XG, Wang P, Li XF, Yang SC, Ding GC, Christie P, Li L (2019) Crop diversity facilitates soil aggregation in relation to soil microbial community composition driven by intercropping. Plant Soil 436:173–192. https://doi.org/10.1007/s11104-018-03924-8
Tomassoli L, Barba M (2000) Occurrence of Melon Necrotic Spot Carmovirus in Italy. EPPO Bull 30:279–280. https://doi.org/10.1111/j.1365-2338.2000.tb00895.x
Trouvelot A, Kough JL, Gianinazzi-Pearson V (1986) Mesure du taux de mycorhization VA d’un système radiculaire. Recherche de méthodes d’estimation ayant une signification fonctionnelle. In: Gianinazzi-Pearson V, Gianinazzi S (eds) Mycorrhizae: Physiology and Genetics. INRA, Paris, France , pp 217–221
Wang X, Radwan MM, Taráwneh A, Gao J, Wedge DE, Rosa LH, Cutler HG, Cutler SJ (2013) Antifungal activity against plant pathogens of metabolites from the endophytic fungus Cladosporium cladosporioides. J Agr Food Chem 61:4551–4555. https://doi.org/10.1021/jf400212y
White TJ, Brums T, Lee S, Taylor J (1990) Amplification phylogenetics. In: Innis N, Gelfand D, Sninsky J, White T (eds) PCR Protocols: a Guide to Methods and Applications. Accademic Press, San Diego, pp 315–322
Waugh M, Kim DH, Ferrin DM, Stanghellini ME (2003) Reproductive potential of Monosporascus cannonballus. Plant Dis 87:45–50. https://doi.org/10.1094/PDIS.2003.87.1.45
Xiong W, Li R, Ren Y, Liu C, Zhao Q, Wu H, Jousset A, Shen Q (2017) Distinct roles for soil fungal and bacterial communities associated with the suppression of vanilla Fusarium wilt disease. Soil Biol Biochem 107:198–207. https://doi.org/10.1016/j.soilbio.2017.01.010
Acknowledgements
This work was supported by a project grant from Regione Sardegna (delibera della Giunta Regionale n. 52/101). Authors thank Gianfranco Siddu (Op-Ortofrutta cooperativa produttori di Arborea) and Sandro Murru (Agenzia Laore Sardegna) for technical assistance in the on-field trials.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All other authors herewith declare that they have no conflict of interest. This study does not contain studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Infantino, A., Balmas, V., Schianchi, N. et al. Diversity of soil-borne fungal species associated to root rot and vine decline of melon in Sardinia (Italy). J Plant Pathol 103, 421–432 (2021). https://doi.org/10.1007/s42161-021-00774-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-021-00774-9