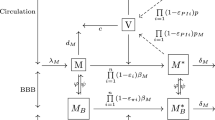
Abstract
Combination antiretroviral therapy (cART) has greatly increased life expectancy for human immunodeficiency virus-1 (HIV-1)-infected patients. Even given the remarkable success of cART, the virus persists in many different cells and tissues. The presence of viral reservoirs represents a major obstacle to HIV-1 eradication. These viral reservoirs contain latently infected long-lived cells. The “Shock and Kill” therapeutic strategy aims to reactivate latently infected cells by latency reversing agents (LRAs) and kill these reactivated cells by strategies involving the host immune system. The brain is a natural anatomical reservoir for HIV-1 infection. Brain macrophages, including microglia and perivascular macrophages, display productive HIV-1 infection. A mathematical model was used to analyze the dynamics of latently and productively infected brain macrophages during viral infection and this mathematical model enabled prediction of the effects of LRAs applied to the “Shock and Kill” strategy in the brain. The model was calibrated using reported data from simian immunodeficiency virus (SIV) studies. Our model produces the overarching observation that effective cART can suppress productively infected brain macrophages but leaves a residual latent viral reservoir in brain macrophages. In addition, our model demonstrates that there exists a parameter regime wherein the “Shock and Kill” strategy can be safe and effective for SIV infection in the brain. The results indicate that the “Shock and Kill” strategy can restrict brain viral RNA burden associated with severe neuroinflammation and can lead to the eradication of the latent reservoir of brain macrophages.







Similar content being viewed by others
References
Althaus CL, Joos B, Perelson AS, Günthard HF (2014) Quantifying the turnover of transcriptional subclasses of hiv-1-infected cells. PLoS Comput Biol 10(10):e1003871
Annamalai L, Bhaskar V, Pauley DR, Knight H, Williams K, Lentz M, Ratai E, Westmoreland SV, González RG, O’Neil SP (2010) Impact of short-term combined antiretroviral therapy on brain virus burden in simian immunodeficiency virus-infected and cd8+ lymphocyte-depleted rhesus macaques. Am J Pathol 177(2):777–791. https://doi.org/10.2353/ajpath.2010.091248
Asahchop EL, Meziane O, Mamik MK, Chan WF, Branton WG, Resch L, Gill MJ, Haddad E, Guimond JV, Wainberg MA, Baker GB, Cohen EA, Power C (2017) Reduced antiretroviral drug efficacy and concentration in hiv-infected microglia contributes to viral persistence in brain. Retrovirology. https://doi.org/10.1186/s12977-017-0370-5
Askew K, Li K, Olmos-Alonso A, Garcia-Moreno F, Liang Y, Richardson P, Tipton T, Chapman MA, Riecken K, Beccari S, Sierra A, Molnár Z, Cragg MS, Garaschuk O, Perry VH, Gomez-Nicola D (2017) Coupled proliferation and apoptosis maintain the rapid turnover of microglia in the adult brain. Cell Rep 18(2):391–405. https://doi.org/10.1016/j.celrep.2016.12.041
Avalos CR, Price SL, Forsyth ER, Pin JN, Shirk EN, Bullock BT, Queen SE, Li M, Gellerup D, O’Connor SL, Zink MC, Mankowski JL, Gama L, Clements JE (2016) Quantitation of productively infected monocytes and macrophages of simian immunodeficiency virus-infected macaques. J Virol 90(12):5643–5656. https://doi.org/10.1128/JVI.00290-16
Azevedo FAC, Carvalho LRB, Grinberg LT, Farfel JM, Ferretti REL, Leite REP et al (2009) Equal numbers of neuronal and nonneuronal cells make the human brain an isometrically scaled-up primate brain. J Comp Neurol 513:532–541. https://doi.org/10.1002/cne.21974
Babas T, Muñoz D, Mankowski JL, Tarwater PM, Clements JE, Zink MC (2003) Role of microglial cells in selective replication of simian immunodeficiency virus genotypes in the brain. J Virol 77(1):208–216. https://doi.org/10.1128/jvi.77.1.208-216.2003
Barber SA, Gama L, Dudaronek JM, Voelker T, Tarwater PM, Clements JE (2006) Mechanism for the establishment of transcriptional hiv latency in the brain in a simian immunodeficiency virus-macaque model. J Infect Dis 193(7):963–970. https://doi.org/10.1086/500983
Bracq L, Xie M, Benichou S, Jérôme Bouchet (2018) Mechanisms for cell-to-cell transmission of hiv-1. Front Immunol. https://doi.org/10.3389/fimmu.2018.00260
Campbell JH, Ratai EM, Autissier P, Nolan DJ, Tse S, Miller AD, González RG, Salemi M, Burdo TH, Williams KC (2014) Anti-\(\alpha \)4 antibody treatment blocks virus traffic to the brain and gut early, and stabilizes cns injury late in infection. PLoS Pathog 10(12):1–15. https://doi.org/10.1371/journal.ppat.1004533
Castillo-Chavez C, Thieme HR (1994) Asymptotically autonomous epidemic models. Technical report (Cornell University. Mathematical Sciences Institute), 94(38)
Cenker JJ, Stultz RD, McDonald D (2017) Brain microglial cells are highly susceptible to hiv-1 infection and spread. AIDS Res Hum Retrov 33(11):1155–1165. https://doi.org/10.1089/aid.2017.0004
Cifuentes-Munoza N, El Najjarb F, Ellis Dutch R (2020) Viral cell-to-cell spread: conventional and non-conventional ways. Adv Virus Res 108:85–125. https://doi.org/10.1016/bs.aivir.2020.09.002
Clements JE, Babas T, Mankowski JL, Suryanarayana K, Piatak M Jr, Tarwater PM, Lifson JD, Zink MC (2002) The central nervous system as a reservoir for simian immunodeficiency virus (siv): steady-state levels of siv dna in brain from acute through asymptomatic infection. J Infect Dis 186:905–913. https://doi.org/10.1086/343768
Clements JE, Li M, Gama L, Bullock B, Carruth LM, Mankowski JL, Zink MC (2005) The central nervous system is a viral reservoir in simian immunodeficiency virus-infected macaques on combined antiretroviral therapy: A model for human immunodeficiency virus patients on highly active antiretroviral therapy. J Neurovirol 11(2):180–9. https://doi.org/10.1080/13550280590922748-1
Deeks SG, Overbaugh J, Phillips A, Buchbinder S (2015) Hiv infection. Nat Rev Dis Primers 1:1–22. https://doi.org/10.1038/nrdp.2015.35
Deeks SG, Lewin SR, Bekker LG (2017) The end of hiv: still a very long way to go, but progress continues. PLoS Med 14(11):e1002466. https://doi.org/10.1371/journal.pmed.1002466
Diekmann O, Heesterbeek JAP, Metz JAJ (1990) On the definition and the computation of the basic reproduction ratio r0 in models for infectious diseases in heterogeneous populations. J Math Biol 28:365–382. https://doi.org/10.1007/bf00178324
Dupont M, James Sattentau Q (2020) Macrophage cell-cell interactions promoting hiv-1 infection. Viruses. https://doi.org/10.3390/v12050492
Eisele E, Siliciano RF (2012) Redefining the viral reservoirs that prevent hiv-1 eradication. Immunity 37(3):377–88. https://doi.org/10.1016/j.immuni.2012.08.010
Gama L, Abreu CM, Shirk EN, Price SL, Li M, Laird GM, Pate KA, Wietgrefe SW, O’Connor SL, Pianowski L, Haase AT, Van Lint C, Siliciano RF, Clements JE (2017) Reactivation of simian immunodeficiency virus reservoirs in the brain of virally suppressed macaques. AIDS 31(1):5–14. https://doi.org/10.1097/QAD.0000000000001267
Gavegnano C, Schinazi RF (2009) Antiretroviral therapy in macrophages: implication for hiv eradication. Antivir Chem Chemother 20(2):63–78. https://doi.org/10.3851/IMP1374
Gelman A, Brooks SP (1998) General methods for monitoring convergence of iterative simulations. J Comp Graph Stat 7(4):434–455
Gelman BB, Lisinicchia JG, Morgello S, Masliah E, Commins D, Achim CL, Fox HS, Kolson DL, Grant I, Singer E, Yiannoutsos CT, Sherman S, Gensler G, Moore DJ, Chen T, Soukup VM (2013) Neurovirological correlation with hiv-associated neurocognitive disorders and encephalitis in a haart-era cohort. J Acquir Immune Defic Syndr 62(5):487–495. https://doi.org/10.1097/QAI.0b013e31827f1bdb
Gerngross L, Fischer T (2015) Evidence for cfms signaling in hiv production by brain macrophages and microglia. J Neurovirol 21(3):249–56. https://doi.org/10.1007/s13365-014-0270-6
González-Scarano F, Martin-Garcia J (2005) The neuropathogenesis of aids. Nat Rev Immunol 5:69–81. https://doi.org/10.1038/nri1527
Graham DR, Gama L, Queen SE, Li M, Brice AK, Kelly KM, Mankowski JL, Clements JE, Zink MC (2011) Initiation of haart during acute simian immunodeficiency virus infection rapidly controls virus replication in the cns by enhancing immune activity and preserving protective immune responses. J Neurovirol 17:120–130. https://doi.org/10.1007/s13365-010-0005-2
Grinsted A (2015) Markov chain monte carlo sampling of posterior distribution. http://www.mathworks.com/matlabcentral/fileexchange/49820-grinsted-gwmcmc
Gupta V, Dixit NM (2018) Trade-off between synergy and efficacy in combinations of hiv-1 latency-reversing agents. PLoS Comput Biol 14(2):e1006004. https://doi.org/10.1371/journal.pcbi.1006004
Hartmann P, Ramseier A, Gudat F, Mihatsch MJ, Polasek W (1994) Normal weight of the brain in adults in relation to age, sex, body height and weight. Pathologe 15(3):165–170. https://doi.org/10.1007/s002920050040
Helke KL, Queen SE, Tarwater PM, Turchan-Cholewo J, Nath A, Zink MC, Irani DN, Mankowski JL (2005) 14–3-3 protein in csf: an early predictor of siv cns disease. J Neuropathol Exp Neurol 64(3):202–208. https://doi.org/10.1093/jnen/64.3.202
Henderson LJ, Reoma LB, Kovacs JA, Nath A (2020) Advances toward curing hiv-1 infection in tissue reservoirs. J Virol 94(3):e00375-19
Herculano-Houzel S (2014) The glia/neuron ratio: how it varies uniformly across brain structures and species and what that means for brain physiology and evolution. Glia 62:1377–1391
Hernandez-Vargas EA (2020) Modeling kick-kill strategies toward hiv cure. Front Immunol 8:995. https://doi.org/10.3389/fimmu.2017.00995
The MathWorks Inc. Matlab version 9.7.0.1261785 Natick, Massachusetts., (2019)
Kim Y, Anderson JL, Lewin SR (2018) Getting the “kill” into “shock and kill”: Strategies to eliminate latent hiv. Cell Host Microbe 23(1):14–26. https://doi.org/10.1016/j.chom.2017.12.004
Kirschner DE, Perelson AS (1995) A model for the immune system response to hiv: Azt treatment studies. In: Arino O, Axelrod DE, Kimmel M, Langlais M (eds) Mathematical population dynamics: analysis of heterogeneity theory of epidemics, vol 1. Wuerz Publishing Ltd, Winnipeg, pp 295–310
Lindl KA, Marks DR, Kolson DL (2010) Hiv-associated neurocognitive disorder: pathogenesis and therapeutic opportunities. J Neuroimmune Pharmacol 5:294–309. https://doi.org/10.1007/s11481-010-9205-z
Loucif H, Gouard S, Dagenais-Lussier X, Murira A, Van Grevenynghe J, Stager S, Tremblay C (2018) Deciphering natural control of hiv-1: A valuable strategy to achieve antiretroviral therapy termination. Cytokine Growth Factor Rev 40:90–98. https://doi.org/10.1016/j.cytogfr.2018.03.010
Lutgen V, Narasipura SD, Barbian HJ, Richards M, Wallace J, Razmpour R, Buzhdygan T, Ramirez SH, Prevedel L, Eugenin EA, Al-Harthi L (2020) Hiv infects astrocytes in vivo and egresses from the brain to the periphery. PLoS Pathog 16(6):e1008381
Meulendyke KA, Queen SE, Engle EL, Shirk EN, Liu J, Steiner JP, Nath A, Tarwater PM, Graham DR, Mankowski JL, Zink MC (2014) Combination fluconazole/paroxetine treatment is neuroprotective despite ongoing neuroinflammation and viral replication in an siv model of hiv neurological disease. J Neurovirol 20(6):591–602. https://doi.org/10.1007/s13365-014-0283-1
Nowak MA, May RM (2000) Virus dynamics. Mathematical principles of immunology and virology. Oxford University Press, Oxford
Patrick MK, Johnston JB, Power C (2002) Lentiviral neuropathogenesis: comparative neuroinvasion, neurotropism, neurovirulence, and host neurosusceptibility. J Virol 76(16):7923–7931. https://doi.org/10.1128/JVI.76.16.7923-7931.2002
Roda WC (2020) Bayesian inference for dynamical systems. Infect Dis Model 5:221–232. https://doi.org/10.1016/j.idm.2019.12.007
Roda WC, Li MY, Akinwumi MS, Asahchop EL, Gelman BB, Witwer KW, Power C (2017) Modeling brain lentiviral infections during antiretroviral therapy in aids. J Neurovirol 23(4):577–586. https://doi.org/10.1007/s13365-017-0530-3
Singer EJ, Nemanim NM (2017) The persistence of hiv-associated neurocognitive disorder (hand) in the era of combined antiretroviral therapy (cart). In: Shapshak P, Levine A, Foley B, Somboonwit C, Singer E, Chiappelli F, Sinnott JT (eds) Global Virology II - HIV and NeuroAIDS. Springer, LLC, New York, pp 375–403
Suspène R, Meyerhans A (2012) Quantification of unintegrated hiv-1 dna at the single cell level in vivo. PLoS One 7(5):e36246
Thieme HR (1994) Asymptotically autonomous differential equations in the plane. Rocky Mt J Math 24(1):351–380. https://doi.org/10.1216/rmjm/1181072470
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48. https://doi.org/10.1016/S0025-5564(02)00108-6
van Marle G, Church DL, van der Meer F, Gill MJ (2018) Combating the hiv reservoirs. Biotechnol Genet Eng Rev 34(1):76–89. https://doi.org/10.1080/02648725.2018.1471641
Vivithanaporn P, Heo G, Gamble J, Krentz HB, Hoke A, Gill MJ, Power C (2010) Neurologic disease burden in treated hiv/aids predicts survival. Neurology 75:1150–1158. https://doi.org/10.1212/WNL.0b013e3181f4d5bb
Wallet C, De Rovere M, Van Assche J, Daouad F, De Wit S, Gautier V, Mallon PW, Marcello A, Van Lint C, Rohr O, Schwartz C (2019) Microglial cells: the main hiv-1 reservoir in the brain. Front Cell Infect Microbiol. https://doi.org/10.3389/fcimb.2019.00362
Walsh JG, Reinke SN, Mamik MK, McKenzie BA, Maingat F, Branton WG, Broadhurst DI, Power C (2014) Rapid inflammasome activation in microglia contributes to brain disease in hiv/aids. Retrovirology. https://doi.org/10.1186/1742-4690-11-35
Wandeler G, Johnson LF, Egger M (2016) Trends in life expectancy of hiv-positive adults on art across the globe: comparisons with general population. Curr Opin HIV AIDS 11(5):492–500. https://doi.org/10.1097/COH.0000000000000298
Wang Y, Liu M, Lu Q, Farrell M, Lappin JM, Shi J, Lu L, Bao Y (2020) Global prevalence and burden of hiv-associated neurocognitive disorder: a meta-analysis. Neurology 95(19):e2610–e2621. https://doi.org/10.1212/WNL.0000000000010752
Witwer KW, Gama L, Li M, Bartizal CM, Queen SE, Varrone JJ, Brice AK, Graham DR, Tarwater PM, Mankowski JL, Zink MC, Clements JE (2009) Coordinated regulation of siv replication and immune responses in the cns. PLoS One 4(12):e8129. https://doi.org/10.1371/journal.pone.0008129
Zink MC, Suryanarayana K, Mankowski JL, Shen A, Piatak M Jr, Spelman JP, Carter DL, Adams RJ, Lifson JD, Clements JE (1999) High viral load in the cerebrospinal fluid and brain correlates with severity of simian immunodeficiency virus encephalitis. J Virol 73(12):10480–10488
Zink MC, Uhrlaub J, DeWitt J, Voelker T, Bullock B, Mankowski J, Tarwater P, Clements J, Barber S (2005) Neuroprotective and anti-human immunodeficiency virus activity of minocycline. JAMA 293(16):2003–2011. https://doi.org/10.1001/jama.293.16.2003
Zink MC, Brice AK, Kelly KM, Queen SE, Gama L, Li M, Adams RJ, Bartizal C, Varrone J, Rabi SA, Graham DR, Tarwater PM, Mankowski JL, Clements JE (2010) Simian immunodeficiency virus-infected macaques treated with highly active antiretroviral therapy have reduced central nervous system viral replication and inflammation but persistence of viral dna. J Infect Dis 202(1):161–170. https://doi.org/10.1086/653213
Acknowledgements
Research for MYL is supported in part by grants from the Natural Science and Engineering Research Council of Canada (NSERC) and Canada Foundation for Innovation (CFI).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A: Mathematical Results and Proofs
Using the theory of asymptotically autonomous differential equations (Thieme 1994; Castillo-Chavez and Thieme 1994), the asymptotic behaviors of system (1) are the same as the following two-dimensional system of differential equations in the bounded feasible region \(D=\{ (x,y)\in {\mathbb {R}}^{2}_{+}: 0 \le x+y \le \frac{\lambda }{k} \}\):
The Jacobian matrix of system (8) is
Theorem 1
-
1.
If the control reproduction number \(R_c < 1\), then the disease-free equilibrium \(P_0\) is locally asymptotically stable. If \(R_c > 1\), then \(P_0\) is unstable.
-
2.
If \(R_c > 1\) and \(R_{c1} > R_{c2}\), then the productive equilibrium \(P_1\) is locally asymptotically stable.
-
3.
If \(R_c > 1\) and \(R_{c1} < R_{c2}\), then the latent equilibrium \(P_2\) is locally asymptotically stable.
Proof
The Jacobian matrix evaluated at \(P_0\) is
Since the matrix (10) is upper triangular, the eigenvalues of the matrix \(J(P_0)\) are \(\mu _1 = -k + \beta _2 \frac{\lambda }{k}\) and \(\mu _2 = p (1 - \epsilon ) \beta _1 \frac{\lambda }{k} - (k + \epsilon \gamma )\). If \(R_c < 1\), then \(\mu _1, \mu _2 < 0\) and the disease-free equilibrium \(P_0\) is locally asymptotically stable. If \(R_c > 1\), then max\(\{\mu _1, \mu _2 \} > 0\) and \(P_0\) is unstable.
The Jacobian matrix evaluated at \(P_1\) is
since \(p (1 - \epsilon ) \beta _1 x_1 - k - \epsilon \gamma = \frac{(p(1 - \epsilon ) \beta _1)(k + \epsilon \gamma )}{p(1 - \epsilon ) \beta _1} - (k + \epsilon \gamma )\) = 0. If \(R_c > 1\) and \(R_{c1} > R_{c2}\), then \(R_{c1}> 1 > \frac{R_{c2}}{R_{c1}}\) and by using the productive equilibrium \(P_1\),
and
By the Routh–Hurwitz condition, \(P_1\) is locally asymptotically stable.
The Jacobian matrix evaluated at \(P_2\) is
Since the matrix (14) is upper triangular, the eigenvalues of the matrix 14 are \(\mu _1 = k - \frac{\lambda }{k} \beta _2\) and \(\mu _2 = p (1 - \epsilon ) \beta _1 \frac{k}{\beta 2} - k - \epsilon \gamma \). If \(R_c > 1\) and \(R_{c1} < R_{c2}\), then \(\mu _1, \mu _2 < 0\) and the latent equilibrium \(P_2\) is locally asymptotically stable.
\(\square \)
Theorem 2
If the control reproduction number \(0 < R_c \le 1\), then the disease-free equilibrium \(P_0\) is globally asymptotically stable in \(\varGamma \).
Proof
When \(0 < R_c \le 1\), \(P_0\) is the only equilibrium.
Consider the Lyapunov function \(L = y\). Then
The maximal invariant set in \(\{(x, y, l) \in \varGamma : {\dot{L}} = 0\}\) is the singleton \(P_0\). By LaSalle’s invariance principle, all limit points of solutions belong to the largest invariant set in \(\{(x, y, l) \in \varGamma : {\dot{L}} = 0\}\). Therefore, all solutions in \(\varGamma \) converge to \(P_0\) and \(P_0\) is globally asymptotically stable in \(\varGamma \).
\(\square \)
Theorem 3
If \(R_c > 1\) and \(R_{c1} > R_{c2}\), then the productive equilibrium \(P_1\) is globally asymptotically stable in \(\mathring{\varGamma }\). If \(R_c > 1\) and \(R_{c1} < R_{c2}\), then the latent equilibrium \(P_2\) is globally asymptotically stable in \(\varGamma \).
Proof
The asymptotic behaviors of system (1) in \(\varGamma \) are the same as the asymptotic behaviors of system (8) in D. In system (8), let \(\mathbf{f } (x, y) = (g(x, y), h(x, y))\).
Consider a scalar-valued function \(\alpha (x, y) = \frac{1}{x y (\frac{\lambda }{k} - x - y)}\),
where \((x, y) \in \mathring{D} = \{(x, y): 0< x + y < \frac{\lambda }{k} \}\). For all \((x, y) \in \mathring{D}\),
Therefore, by Dulac’s criteria, system (8) has no closed orbit lying entirely in \(\mathring{D}\). So, no periodic solutions can exist.
By the Poincare–Bendixson theorem, if \(R_c > 1\) and \(R_{c1} > R_{c2}\), all solutions with initial condition in \(\mathring{D}\) must have \(P_1\) as an \(\omega \)-limit point. Since \(P_1\) is locally asymptotically stable, solutions that get close to \(P_1\) must converge to \(P_1\) and all \(\omega \)-limit sets in \(\mathring{D}\) are equal to the singleton \(\{ P_1 \}\). Therefore, \(P_1\) is globally stable in \(\mathring{D}\) when \(R_c > 1\) and \(R_{c1} > R_{c2}\).
Similarly, by using the Poincare–Bendixson theorem, if \(R_c > 1\) and \(R_{c1} < R_{c2}\), then \(P_2\) is globally stable in \(\mathring{D}\).
\(\square \)
Appendix B: Data Conversion
SIV brain viral DNA was measured in three different units (Clements et al. 2002; Zink et al. 2010; Graham et al. 2011; Clements et al. 2005): \(\text {log}_{10}\) SIV DNA copy equivalents per 2 micrograms of total DNA, \(\text {log}_{10}\) SIV DNA copy equivalents per 10,000 cells, and SIV DNA copy equivalents per microgram of total DNA.
The first and second SIV brain viral DNA units were converted to integrated SIV DNA per gram of brain tissue in the same fashion as described in the supplementary material of our previous modeling study (Roda et al. 2017). Since each SIV-infected brain macrophage is assumed to contain a single copy of integrated viral DNA, the integrated SIV DNA per gram of brain tissue is equal to the number of stably infected brain macrophages per gram of brain tissue.
For the third SIV brain viral DNA unit, these data were converted to integrated SIV DNA per gram of brain tissue in a similar way to the first SIV brain viral DNA unit. The SIV DNA copy equivalents per microgram of total DNA were converted to SIV DNA copy equivalents per gram of brain tissue by using the conversion that each gram of brain tissue contains 4 micrograms of total host genomic DNA. By using the ratio of integrated proviral DNA to total viral DNA (1:86) (Suspène and Meyerhans 2012) and our assumption that each SIV-infected brain macrophage is assumed to contain a single copy of integrated viral DNA, we obtain the number of stably infected brain macrophages per gram of brain tissue.
SIV brain viral RNA was measured in five different units (Avalos et al. 2016; Babas et al. 2003; Barber et al. 2006; Clements et al. 2002; Graham et al. 2011; Helke et al. 2005; Meulendyke et al. 2014; Witwer et al. 2009; Zink et al. 1999, 2005, 2010): SIV RNA copy equivalents per microgram of total RNA, \(\text {log}_{10}\) SIV RNA copy equivalents per microgram of total RNA, SIV RNA copy equivalents per 2 micrograms of total RNA, \(\text {log}_{10}\) SIV RNA copy equivalents per 2 micrograms of total RNA, and \(10^6\) SIV RNA copy equivalents per microgram of total RNA. When the SIV brain viral RNA unit is not already in the form of SIV RNA copy equivalents per microgram of total RNA, they are converted to SIV RNA copy equivalents per microgram of total RNA by exponentiation. The SIV RNA copy equivalents per microgram of total RNA were converted to SIV RNA copy equivalents per gram of brain tissue by using the conversion that each gram of brain tissue contains 25 micrograms of total host genomic RNA.
Some SIV studies measured SIV brain viral DNA and RNA in multiple regions of the brain (Avalos et al. 2016; Clements et al. 2002; Graham et al. 2011; Helke et al. 2005; Witwer et al. 2009; Zink et al. 1999; Clements et al. 2005). For these SIV studies, the average SIV brain viral DNA and RNA across the brain regions was used. This was done so that the fitted system (1) and prediction using system (5) would be projecting the average viral dynamics per gram of brain tissue.
After these conversions are completed, the data for SIV RNA copies per gram of brain tissue and the estimated number of stably infected brain macrophages per gram of brain tissue are given in Table 5.
Prior Distributions for Bayesian Inference
The prior distributions are listed in Table 6. The parameters \(\beta _1\), \(\beta _2\), k, and \(x_0\) used the same uniform prior distributions as described in (Roda et al. 2017) and they are the following:
\(\beta _1 \sim U(1 \times 10^{-8}, 1 \times 10^{-4})\), \(\beta _2 \sim U(1 \times 10^{-8}, 1 \times 10^{-4})\), \(k \sim U(0.5, 10.22)\), and \(x_0 \sim U(2.07 \times 10^{6}, 3.94 \times 10^{6})\). The constraint \(\beta _2 < p \beta _1\) was used for the assumption that infection due to productively infected brain macrophages, \(\beta _2\), is sufficiently larger than that due to latently infected brain macrophages, \(\beta _1\). Since \(x_0 = \frac{\lambda }{k}\), \(\lambda \) is determined by \(x_0\) and k. Simple linear regression is used to estimate, \(\text {log}_{10} (y_0)\), by fitting the following linear model to the \(\text {log}_{10}\) SIV DNA copies per gram data for untreated infection (row 1 in Table 2):
where \(\text {log}_{10} (y)\) is the \(\text {log}_{10}\) SIV DNA copies per gram data for untreated infection, \(\text {log}_{10} (y_0)\) is the intercept, \(m_y\) is the slope, and \(t_y\) is the independent variable time. The intercept of this regression model, \(\text {log}_{10} (y_0)\), is estimated to be 1.045, and consequently, \(y_0\) is estimated to be 11.1 and the chosen prior distribution is \(y_0 \sim U(1, 11.1)\). It is assumed that \(l_0 = 0\). The initial viral load is given by \(v_0 = N y_0\) and consequently, \(N = \frac{v_0}{y_0}\). Simple linear regression is used to estimate, \(\text {log}_{10} (v_0)\), by fitting the following linear model to the \(\text {log}_{10}\) SIV RNA copies per gram data for untreated infection (row 2 in Table 2):
where \(\text {log}_{10} (v)\) is the \(\text {log}_{10}\) SIV RNA copies per gram data for untreated infection, \(\text {log}_{10} (v_0)\) is the intercept, \(m_v\) is the slope, and \(t_v\) is the independent variable time. The intercept of this regression model, \(\text {log}_{10} (v_0)\), is estimated to be 3.676 and hence \(v_0\) is estimated to be \(4.74 \times 10^3\). Since \(v_0\) is estimated to be \(4.74 \times 10^3\) and \(y_0 \sim U(1, 11.1)\), \(N \sim U(427, 4.74 \times 10^3)\). The cART effectiveness, \(\epsilon \), varies between 0 and 1 and the prior distribution is \(\epsilon \sim U(0.01, 1)\). The rate productively infected brain macrophages are deactivated once cART is applied, \(\epsilon \gamma \), is unknown and a broad range is chosen for \(\gamma \sim U(0.01, 200)\). The proportion of newly infected susceptible brain macrophages by productively infected brain macrophages that enter the productively infected population is assumed to be \(p = 0.5\).
Model Fitting and Prediction Using Bayesian Inference
Bayesian inference is used to fit system (1) simultaneously to the SIV brain viral data for untreated and cART-exposed animals (data in the first eight rows of Table 2). Let \(D_j = \{ d^{j}_1, \dots , d^{j}_{n_j} \}\) and \(T_j = \{ t^{j}_1, \dots , t^{j}_{n_j} \}\) be the SIV brain viral data and times corresponding to the \(j^{\text {th}}\) row of Table 2. Let \(D = \{ D_1, \dots , D_8 \}\) and \(T = \{ T_1, \dots , T_8 \}\).
System (1) was solved numerically by using the MATLAB function ode45 with the option setting odeset(“nonnegative,” 2) to ensure that numerically solved solutions have nonnegative values (Inc. 2019). The function ode45 is based on an explicit Runge–Kutta (4,5) formula.
There are four different scenarios being considered when system (1) is being fit simultaneously to the SIV brain viral data for untreated and cART-exposed animals: untreated SIV infection, SIV infection with cART initiated at 4 days p.i., SIV infection with cART initiated at 12 days p.i., and SIV infection with cART initiated at 42 days p.i. For untreated SIV infection, \(\epsilon \) is set to zero in system (1). For the scenarios when cART is initiated at a certain time p.i., \(\epsilon \) is time dependent in system (1):
where the constant \(\epsilon \) and is to be estimated. The parameter vector to be estimated in system (1) is \(\varvec{\nu } = \langle \beta _1, \beta _2, k, x_0, y_0, N, \epsilon , \gamma \rangle \). For untreated infection, let the model solution vector over time for stably infected brain macrophages per gram of brain tissue and SIV RNA copy equivalents per gram of brain tissue be given by \(s_u(\varvec{\nu }, t)\) and \(v_u(\varvec{\nu }, t)\), respectively. The dataset \(D_1\) will be described by the model solution vector over time \(s_u(\varvec{\nu }, t)\). The dataset \(D_2\) will be described by the model solution vector over time \(v_u(\varvec{\nu }, t)\). Similarly, for the scenarios when cART is initiated at a certain time p.i., \(D_3\) and \(D_4\) will be described by \(s_g(\varvec{\nu }, t)\) and \(v_g(\varvec{\nu }, t)\), respectively, \(D_5\) and \(D_6\) will be described by \(s_z(\varvec{\nu }, t)\) and \(v_z(\varvec{\nu }, t)\), respectively, and \(D_7\) and \(D_8\) will be described by \(s_c(\varvec{\nu }, t)\) and \(v_c(\varvec{\nu }, t)\), respectively. The subscript letters g, z, and c are inspired by the last name of the first author on the SIV studies (Graham et al. 2011; Zink et al. 2010; Clements et al. 2005) in order to quickly keep track of the corresponding model solution vector for each of the datasets \(D_3\) through \(D_8\).
Since each of the observed SIV brain viral datasets \(D_1\) and \(D_3\) through \(D_8\) is overdispersed count data (variance of the data is larger than the mean of the data), a negative binomial distribution is chosen to describe each of the datasets \(D_1\) and \(D_3\) through \(D_8\). The observed SIV brain viral RNA dataset for untreated animals \(D_2\) is also overdispersed count data. However, many of the counts in the dataset \(D_2\) are very large positive numbers. For this reason, a normal distribution is used to describe the \(\text {log}_{10}\) of the data \(D_2\).
Hence, for \(j = 1, 3, \dots , 8\), the probability of observing \(d_i^j\) is given by the negative binomial distribution:
where \(r_i^j = \frac{(p^j) (\mu _i^j)}{1 - (p^j)} \iff \mu _i^j = \frac{(r_i^j) (1 - p^j)}{p^j}\) changes depending on the time \(t_i^j\), and \(0< p^j < 1\) is specific to the \(j^{\text {th}}\) dataset \(D_j\) and determines the most likely shape of the negative binomial distribution given the data \(D_j\). Hence, the variance, \(Var[ D_i^j ] = \frac{\mu _i^j}{p^j}\), also changes over time. Here \(\mu _i^1 = s_u(\varvec{\nu }, t_i^1)\), \(\mu _i^3 = s_g(\varvec{\nu }, t_i^1)\), \(\mu _i^4 = v_g(\varvec{\nu }, t_i^1)\), \(\mu _i^5 = s_z(\varvec{\nu }, t_i^1)\), \(\mu _i^6 = v_z(\varvec{\nu }, t_i^1)\), \(\mu _i^7 = s_c(\varvec{\nu }, t_i^1)\), and \(\mu _i^8 = v_c(\varvec{\nu }, t_i^1)\).
Based on the datasets \(D_1, D_3, \dots , D_8\), the following uniform prior distributions are chosen for \(p^1, p^3, \dots , p^8\):
\(p^1 \sim U(1 \times 10^{-3}, 1)\), \(p^3 \sim U(1 \times 10^{-4}, 1)\), \(p^4 \sim U(1 \times 10^{-4}, 1)\),
\(p^5 \sim U(1 \times 10^{-2}, 1)\), \(p^6 \sim U(1 \times 10^{-1}, 1)\), \(p^7 \sim U(1 \times 10^{-1}, 1)\), and
\(p^8 \sim U(1 \times 10^{-1}, 1)\).
For \(j = 2\), the probability of observing \(\text {log}_{10} (d_i^2)\) is given by the normal distribution:
where the mean \(\mu _i^2\) changes depending on the time, \(t_i^2\), and the variance \(\frac{1}{\tau }\) is specific to the dataset \(D_2\). Here \(\mu _i^2 = \text {log}_{10} (v_u(\varvec{\nu }, t_i^2))\).
Based on the dataset \(D_2\), the uniform prior distribution for \(\tau \) is chosen to be \(U(1 \times 10^{-2}, 100)\).
Let \(\varvec{\phi } = \langle p^1, \tau , p^3, p^4, p^5, p^6, p^7, p^8 \rangle \). Given the extra parameters in vector \(\varvec{\phi }\), we want to estimate the vector \(\varvec{\theta } = \langle \varvec{\nu }, \varvec{\phi } \rangle \).
The probability model for datasets \(D_1, D_3, \dots , D_8\) is
and the probability model for dataset \(D_2\) is
The likelihood function for \(\varvec{\theta }\) is given by
where C is any positive constant not depending on \(\varvec{\theta }\) used to simplify the likelihood function. For more information about Bayesian inference for dynamical systems and combining probability models, we refer the reader to the recent lecture notes (Roda 2020).
The prior distribution for \(\varvec{\theta }\) is equal to the product of the uniform distributions specified for the parameters in \(\varvec{\theta }\).
The fitting was completed using an affine invariant ensemble Markov Chain Monte Carlo (MCMC) sampling program from the MATLAB Central File Exchange (Grinsted 2015). The MCMC program sampled from the following unnormalized posterior distribution, \(\pi (\varvec{\theta } | D)\):
where \(\varvec{\theta }\) is the vector of unknown parameters, \(D = \{ D_1, \dots , D_8 \}\) is the observed data, \(L(\varvec{\theta })\) is the likelihood function, and \(P(\varvec{\theta })\) is the prior distribution.
The parameters to be estimated in system (1) are \(\varvec{\nu } = \langle \beta _1, \beta _2, k, x_0, y_0, N, \epsilon , \gamma \rangle \).
The affine invariant ensemble MCMC sampler was used with \(T = 375,000\) iterations and \(K = 32\) walkers making a total of \(K T = 12,000,000\) samples. Every \(10^{\text {th}}\) sample is thinned from each walker during the MCMC algorithm to decrease autocorrelation. After the iterations of the MCMC sampler are completed, a burn-in of 10,000 is used for each walker and the number of pooled samples after the burn-in is \(H = 880,000\). Convergence of the MCMC sampling to the estimated posterior distribution for each parameter in \(\varvec{\theta }\) was determined by using a general univariate comparison method (Gelman and Brooks 1998). The general univariate comparison method uses the distance of the empirical \(100 (1 - \alpha )\%\) interval for the pooled samples, S, and divides this distance by the average of the distances of the empirical \(100 (1 - \alpha )\%\) interval for each of the K walkers, \(s_i\), to receive the potential scale reduction factor, r (Gelman and Brooks 1998). Here the empirical 95% interval (\(\alpha = 0.05\)) is used for the general univariate comparison method. Table 7 contains the estimated parameters in \(\varvec{\theta }\) with the 95% credible intervals and the potential scale reduction factors, r, for each parameter. The potential scale reduction factors, r, are close to 1 and this indicates that the sampler converged to the posterior distribution.
The 95% prediction intervals for each dataset \(D_j\), \(j = 1, \dots , 8\), are determined by the posterior predictive distribution as described in (Roda 2020).
For the scenario when cART is initiated at a certain time p.i. and LRA drugs are initiated at a later time p.i., \(\omega \) is set to zero in system (5) and \(\epsilon \) and \(\alpha \) are time dependent in system (5). The time-dependent \(\epsilon \) is given by (17) and \(\alpha (t)\) is given by
where \(\alpha \) is a constant.
For the model prediction of the LRA drug SIV experiment (data \(D_9\)), the vector \(\varvec{\nu }\) in the pooled samples of \(\varvec{\theta } = \langle \varvec{\nu }, \varvec{\phi } \rangle \) in H are used in system (5) with \(\omega = 0\) and the reactivation rate, \(\alpha \), in (24) is varied from 0.01 to 100 to determine which predicted solutions pass near the points in data \(D_9\). The mean of these predicted solutions is displayed in Fig. 5.
Likewise, for the scenario when cART is initiated at a certain time p.i. and the “Shock and Kill” strategy is initiated at a later time p.i., \(\epsilon \), \(\alpha \), and \(\omega \) are time dependent in system (5). The time-dependent \(\epsilon \) is given by (17) and \(\alpha (t)\) is given by
where \(\alpha \) is a constant, and
where \(\omega \) is a constant.
Similarly, for the prediction of the “Shock and Kill” strategy, the vector \(\varvec{\nu }\) in the pooled samples of \(\varvec{\theta } = \langle \varvec{\nu }, \varvec{\phi } \rangle \) in H are used. For each vector \(\varvec{\nu }\), system (5) is solved and the control reproduction number, \({\overline{R}}_{c}\), is calculated as the reactivation rate, \(\alpha \), in (25) and additional kill rate, \(\omega \), in (26) are varied from 0 to 40. The mean control reproduction number, mean \({\overline{R}}_{c}\), is shown in Fig. 6 and the mean of the predicted model solutions within the safe and effective region (turquoise region in Fig. 6) are shown in Fig. 7.
Rights and permissions
About this article
Cite this article
Roda, W.C., Liu, S., Power, C. et al. Modeling the Effects of Latency Reversing Drugs During HIV-1 and SIV Brain Infection with Implications for the “Shock and Kill” Strategy. Bull Math Biol 83, 39 (2021). https://doi.org/10.1007/s11538-021-00875-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-021-00875-7