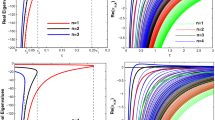
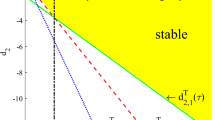
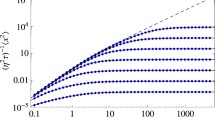
Abstract
Diffusion has been widely applied to model animal movement that follows Brownian motion. However, animals typically move in non-Brownian ways due to their perceptual judgment. Spatial memory and cognition recently have received much attention in characterizing complicated animal movement behaviours. Explicit spatial memory is modeled via a distributed delayed diffusion term in this paper. The distributed time represents the memory growth and decay over time, and the spatial nonlocality reflects the dependence of spatial memory on location. When the temporal delay kernel is weak under the assumption that animals can immediately acquire knowledge and memory decays over time, the equation is equivalent to a Keller–Segel chemotaxis model. For the strong kernel with learning and memory decay stages, rich spatiotemporal dynamics, such as Turing and checker-board patterns, appear via spatially non-homogeneous steady-state and Hopf bifurcations.




Similar content being viewed by others
References
Abrahms B, Hazen EL, Aikens EO, Savoca MS, Goldbogen JA, Bograd SJ, Jacox MG, Irvine LM, Palacios DM, Mate BR (2019) Memory and resource tracking drive blue whale migrations. Proc Natl Acad Sci 116(12):5582–5587
Amann H (1991) Hopf bifurcation in quasilinear reaction-diffusion systems. In: Delay differential equations and dynamical systems (Claremont, CA, 1990), volume 1475 of Lecture Notes in Mathematics. Springer, Berlin, pp 53–63
Bellomo N, Bellouquid A, Tao Y, Winkler M (2015) Toward a mathematical theory of Keller–Segel models of pattern formation in biological tissues. Math Models Methods Appl Sci 25(9):1663–1763
Britton NF (1990) Spatial structures and periodic travelling waves in an integro-differential reaction–diffusion population model. SIAM J Appl Math 50(6):1663–1688
Chen S, Yu J (2016) Stability analysis of a reaction–diffusion equation with spatiotemporal delay and Dirichlet boundary condition. J Dyn Differ Equ 28(3–4):857–866
Cooke KL, Grossman Z (1982) Discrete delay, distributed delay and stability switches. J Math Anal Appl 86(2):592–627
Crandall MG, Rabinowitz PH (1971) Bifurcation from simple eigenvalues. J Funct. Anal. 8:321–340
Crandall MG, Rabinowitz PH (1973) Bifurcation, perturbation of simple eigenvalues and linearized stability. Arch Ration Mech Anal 52:161–180
Ducrot A, Fu X, Magal P (2018) Turing and Turing–Hopf bifurcations for a reaction diffusion equation with nonlocal advection. J Nonlinear Sci 28(5):1959–1997
Fagan WF (2019) Migrating whales depend on memory to exploit reliable resources. Proc Natl Acad Sci 116(12):5217–5219
Fagan WF, Lewis MA, Auger-Méthé M, Avgar T, Benhamou S, Breed G, LaDage L, Schlägel UE, Tang WW, Papastamatiou YP, Forester J, Mueller T (2013) Spatial memory and animal movement. Ecol Lett 16(10):1316–1329
Fagan WF, Gurarie E, Bewick S, Howard A, Cantrell RS, Cosner C (2017) Perceptual ranges, information gathering, and foraging success in dynamic landscapes. Am Nat 189(5):474–489
Foss-Grant AP (2017) Quantitative challenges in ecology: competition, migration, and social learning. Ph.d thesis, University of Maryland
Golledge R (1998) Wayfinding behavior: cognitive mapping and other spatial processes. Johns Hopkins University Press, Baltimore
Gourley SA, Ruan S (2000) Dynamics of the diffusive Nicholson’s blowflies equation with distributed delay. Proc R Soc Edinburgh Sect A 130(6):1275–1291
Gourley SA, So JW-H (2002) Dynamics of a food-limited population model incorporating nonlocal delays on a finite domain. J Math Biol 44(1):49–78
Hillen T, Buttenschön A (2020) Nonlocal adhesion models for microorganisms on bounded domains. SIAM J Appl Math 80(1):382–401
Hillen T, Painter KJ (2009) A user’s guide to PDE models for chemotaxis. J Math Biol 58(1–2):183–217
Jin J, Shi J, Wei J, Yi F (2013) Bifurcations of patterned solutions in the diffusive Lengyel–Epstein system of CIMA chemical reactions. Rocky Mt J Math 43(5):1637–1674
Kappeler P (2010) Animal behaviour: evolution and mechanisms. Springer, Berlin, Heidelberg
Kareiva P, Odell G (1987) Swarms of predators exhibit preytaxis if individual predators use area-restricted search. Am Nat 130(2):233–270
Keller EF, Segel LA (1970) Initiation of slime mold aggregation viewed as an instability. J Theoret Biol 26(3):399–415
Kuto K, Osaki K, Sakurai T, Tsujikawa T (2012) Spatial pattern formation in a chemotaxis-diffusion-growth model. Phys D 241(19):1629–1639
Lee JM, Hillen T, Lewis MA (2009) Pattern formation in prey-taxis systems. J Biol Dyn 3(6):551–573
Lewis MA, Murray JD (1993) Modelling territoriality and wolf–deer interactions. Nature 366(6457):738–740
Liu P, Shi J (2018) Bifurcation of positive solutions to scalar reaction–diffusion equations with nonlinear boundary condition. J Differ Equ 264(1):425–454
Liu P, Shi J, Wang Z (2013) Pattern formation of the attraction-repulsion Keller–Segel system. Discrete Contin Dyn Syst Ser B 18(10):2597–2625
Ma M, Wang Z (2015) Global bifurcation and stability of steady states for a reaction–diffusion-chemotaxis model with volume-filling effect. Nonlinearity 28(8):2639–2660
Macdonald N (1987) Time lags in biological models: lecture notes in biomathematics, vol 27. Springer, Berlin
Mimura M, Tsujikawa T (1996) Aggregating pattern dynamics in a chemotaxis model including growth. Physica A 230(3–4):499–543
Moorcroft PR, Lewis MA (2006) Mechanistic home range analysis. Princeton University Press, Princeton
Moorcroft PR, Lewis MA, Crabtree RL (1999) Home range analysis using a mechanistic home range model. Ecology 80(5):1656–1665
Morales JM, Moorcroft PR, Matthiopoulos J, Frair JL, Kie JG, Powell RA, Merrill EH, Haydon DT (2010) Building the bridge between animal movement and population dynamics. Philos Trans R Soc B Biol Sci 365(1550):2289–2301
O’Keefe J, Nadel L (1978) The Hippocampus as a cognitive map. Oxford University Press, Oxford
Painter KJ, Hillen T (2011) Spatio-temporal chaos in a chemotaxis model. Phys D 240(4–5):363–375
Potts JR, Lewis MA (2016) How memory of direct animal interactions can lead to territorial pattern formation. J R Soc Interface 13(118):20160059
Potts JR, Lewis MA (2019) Spatial memory and taxis-driven pattern formation in model ecosystems. Bull Math Biol 81(7):2725–2747
Schlägel UE, Lewis MA (2014) Detecting effects of spatial memory and dynamic information on animal movement decisions. Methods Ecol Evol 5(11):1236–1246
Shi J (1999) Persistence and bifurcation of degenerate solutions. J Funct Anal 169(2):494–531
Shi J, Wang C, Wang H (2019) Diffusive spatial movement with memory and maturation delays. Nonlinearity 32(9):3188–3208
Shi J, Wang C, Wang H, Yan X (2020) Diffusive spatial movement with memory. J Dyn Differ Equ 32(2):979–1002
Shi J, Wang X (2009) On global bifurcation for quasilinear elliptic systems on bounded domains. J Differ Equ 246(7):2788–2812
Tao Y (2010) Global existence of classical solutions to a predator-prey model with nonlinear prey-taxis. Nonlinear Anal Real World Appl 11(3):2056–2064
Tao Y (2013) Global dynamics in a higher-dimensional repulsion chemotaxis model with nonlinear sensitivity. Discrete Contin Dyn Syst Ser B 18(10):2705–2722
Tello JI, Winkler M (2007) A chemotaxis system with logistic source. Commun Partial Differ. Equ. 32(4–6):849–877
Turing AM (1952) The chemical basis of morphogenesis. Philos Trans R Soc Lond Ser B 237(641):37–72
van Schaik CP (2010) Social learning and culture in animals. Springer, Berlin, pp 623–653
Wang Z, Zhao K (2013) Global dynamics and diffusion limit of a one-dimensional repulsive chemotaxis model. Commun Pure Appl Anal 12(6):3027–3046
Winkler M (2010) Boundedness in the higher-dimensional parabolic-parabolic chemotaxis system with logistic source. Commun Partial Differ Equ 35(8):1516–1537
Winkler M (2014a) Global asymptotic stability of constant equilibria in a fully parabolic chemotaxis system with strong logistic dampening. J Differ Equ 257(4):1056–1077
Winkler M (2014b) How far can chemotactic cross-diffusion enforce exceeding carrying capacities? J Nonlinear Sci 24(5):809–855
Winkler M (2017) Emergence of large population densities despite logistic growth restrictions in fully parabolic chemotaxis systems. Discrete Contin Dyn Syst Ser B 22(7):2777–2793
Wu S, Shi J, Wu B (2016) Global existence of solutions and uniform persistence of a diffusive predator-prey model with prey-taxis. J Differ Equ 260(7):5847–5874
Zuo W, Shi J (2021) Existence and stability of steady-state solutions of reaction–diffusion equations with nonlocal delay effect. Z Angew Math Phys 72(2):43
Zuo W, Song Y (2015) Stability and bifurcation analysis of a reaction–diffusion equation with spatio-temporal delay. J Math Anal Appl 430(1):243–261
Zuo W, Song Y Stability and double-Hopf bifurcations of a Gause–Kolmogorov-type predator-prey system with indirect prey-taxis. J Dyn Differ Equ 1–41 (to appear)
Acknowledgements
We would like to thank the editor and three anonymous reviewers for helpful comments which improve the manuscript. This work was done when the first author visited Department of Mathematics, William & Mary during the academic year 2016–2018, and she would like to thank William & Mary for their support and kind hospitality.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Partially supported by a Grant from China Scholarship Council, NSFC Grant-12001240, Natural Science Foundation of Jiangsu Province (No.BK20200589), US-NSF Grant DMS-1715651, NSERC Discovery Grant RGPIN-2020-03911 and Accelerator Grant RGPAS-2020-00090.
Appendix
Appendix
Proof of Theorem 3
We apply Theorem 1.7 of Crandall and Rabinowitz (1971) for the local bifurcation. Fixing \(d_1,\tau >0\), we define a nonlinear mapping \(F:{\mathbb R}^+\times X^2\rightarrow Y^2\) by
It is clear that \(F(d_2,\theta ,\theta )=0\) for any \(d_2>0\). The Fréchet derivative of F with respect to (u, v) is
Step 1. First we determine the null space of L. From Lemma 4, we have \(D_n(d_{2,s}^w)=0\) so \(\mu =0\) is an eigenvalue of \(J_n^w\) defined in (3.3) thus also an eigenvalue of (3.2) and there exists \(q=(\varphi ,\psi )^T=(1,h_n)\phi _n\in \mathcal {N}(L)\). Moreover as \(T_n(d_{2,s}^w)>0\), \(\mu =0\) is a simple eigenvalue of \(J_n^w\); and since \(\lambda _n\) is a simple eigenvalue of (1.4), and \(d_{2,n}^w\ne d_{2,k}^w\) for any \(k\in {\mathbb N}\) and \(k\ne n\), then \(\mu =0\) is as simple eigenvalue of L and
with \(h_n=1/(d_1\lambda _n\tau +1)\), thus \(\dim \left( {\mathcal {N}(L)}\right) =1\).
Step 2. We next consider the range space \(\mathcal {R}(L)\) of L. We can verify that \(\mathcal {R}(L)\) is given by \(\{(f_1,f_2)\in Y^2: \langle q^*, (f_1,f_2)\rangle =0\}\) where \(q^*\in \mathcal {N}(L^*)\) and \(L^*\) is the adjoint operator of L and defined by
Since \(\mathcal {N}(L^*)=\text {Span}\left\{ q^*=\left( 1,r_n\right) ^T\phi _n\right\} \), where \(r_n=\tau (d_1\lambda _n-f'(\theta ))\). We obtain
and \(\text {codim}\left( \mathcal {R}(L)\right) =1\).
Step 3. We show that \(F_{d_2(u,v)}(d_{2,n}^w,\theta ,\theta )[q]\not \in \mathcal {R}(L)\). From (7.1), we have
Since
thus \(F_{d_2(u,v)}\left( d_{2,n}^w,\theta ,\theta \right) [q]\not \in \mathcal {R}(L)\) by the definition of \(\mathcal {R}(L)\). From Step 1, 2 and 3, now we can apply Theorem 1.7 of Crandall and Rabinowitz (1971) to obtain part (i).
Step 4. Now we consider the bifurcation direction and stability of the bifurcating solutions in \(\Gamma _n\). To obtain more detailed information of the bifurcation, we use one-dimensional domain \(\Omega =(0,l\pi )\). In this case, it is known that \(\phi _n=\cos (nx/l)\) and \(\lambda _n=n^2/l^2\), so that \(q=(1,h_n)^{T}\cos (nx/l)\). From Shi (1999), we have
where \(l\in Y\) satisfies \(\mathcal {N}(l)=\mathcal {R}(L)\) and can be calculated as
By (7.4) and the definition of l, we have
From (7.1), it can be obtained that
This implies that
and thus
Therefore \(d'_{2,n}(0)=0\).
Next we calculate \(d''_{2,n}(0)\) to determine the bifurcation direction by modifying the calculation in Jin et al. (2013). From Shi (1999), \(d''_{2,n}(0)\) takes the form:
where \(\Theta =(\Theta _1,\Theta _2)\) is the unique solution of
From (7.5), we have
thus
In the following, we show the calculation of \(\left\langle l,\ F_{(u,v)(u,v)}\left( d_{2,n}^{w},\theta ,\theta \right) [q,\Theta ]\right\rangle \). By (7.6) and (7.7), we may assume \(\Theta =(\Theta _1,\Theta _2)\) has the following form
since \(F_{(u,v)(u,v)}\left( d_{2,n}^{w},\theta ,\theta \right) \) consists of only constant and \(\cos \left( \frac{2nx}{l}\right) \) terms. Substituting (7.9) into (7.7), we obtain
Form Eq. (7.10), we can solve \(\Theta \) as in (3.10). Thus, we obtain
Using all above we obtain \(d_{2,n}''(0)\) in Eq. (3.9).
Step 5. By applying Corollary 1.13 and Theorem 1.16 of Crandall and Rabinowitz (1973) or Theorem 5.4 of Liu and Shi (2018), the stability of the bifurcating non-constant steady states can be determined by the sign of \(\mu (s)\) which satisfies
where \(m(d_2)\) and \(\mu (s)\) are the eigenvalues defined as
with \(K:X\rightarrow Y\) is the inclusion map \(K(u)=u\), \(m\left( d_{2,n}^{w}\right) =\mu (0)=0\) and \(\left( \varphi \left( d_{2,n}^{w}\right) ,\psi \left( d_{2,n}^{w}\right) \right) =(\Lambda (0),\Phi (0))=(1,h_n)\cos \left( \frac{nx}{l}\right) \).
Now consider the bifurcation at \(d_2=d_{2,N}^w=d_2^*\). From Lemma 4, \((\theta ,\theta )\) is stable and \(m(d_2)<0\) when \(d_2>d_{2,N}^{w}\), and it is unstable and \(m(d_2)>0\) when \(d_2<d_{2,N}^{w}\). One can calculate that
where \(T_N,D_N\) are defined in (3.5), and this implies that \(m'(d_{2,N}^{w})=-\theta \lambda _N/(T_N\tau )<0\). If \(d''_{2,N}(0)<0\), then \(d'_{2,N}(s)<0\) for \(s\in (0,\delta )\) and \(d'_{2,N}(s)>0\) for \(s\in (-\delta ,0)\). Hence \(-sd'_2(s)m'(d_{2,N}^{w})<0\) for \(s\in (-\delta ,\delta )\backslash \{0\}\), and consequently \(\mu (s)<0\) by(7.11) and the bifurcating solutions are locally asymptotically stable. Similarly when \(d''_{2,N}(0)>0\), the bifurcating solutions are unstable. For any other bifurcation at \(d_2=d_{2,n}^w<d_2^*\), the trivial solution \((\theta ,\theta )\) is already unstable at the bifurcation point, hence all bifurcating solutions are also unstable.
Rights and permissions
About this article
Cite this article
Shi, Q., Shi, J. & Wang, H. Spatial movement with distributed memory. J. Math. Biol. 82, 33 (2021). https://doi.org/10.1007/s00285-021-01588-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00285-021-01588-0
Keywords
- Spatial memory
- Reaction–diffusion equation
- Distributed delayed diffusion
- Pattern formation
- Spatially non-homogeneous time periodic solution
- Hopf bifurcation
- Turing bifurcation