Abstract
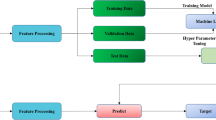
To design a hybrid deep learning system (hDL-system) for discriminating low-grade from high-grade colorectal cancer (CRC) lesions, using immunohistochemically stained biopsy specimens for AIB1 expression. AIB1 has oncogenic function in tumour genesis, and it is an important prognostic factor regarding various types of cancers, including CRC. Clinical material consisted of biopsy specimens of sixty-seven patients with verified CRC (26 low-grade, 41 high-grade cases). From each patient, we digitized images, at × 50 and × 200 lens magnifications. We designed the hDL-system, employing the VGG16 pre-trained convolution neural network for generating DL-features, the SVM classifier, and the bootstrap evaluation method for assessing the discrimination accuracy between low-grade and high-grade CRC lesions. Furthermore, we compared the hDL-system’s discrimination accuracy with that of a supervised machine learning system (sML-system). We designed the sML-system by (i) generating sixty-nine (69) textural and colour features from each image, (ii) employing the probabilistic neural network (PNN) classifier, and (iii) using the bootstrapping method for evaluating sML-system performance. The system design was enabled by employing the CUDA platform for programming in parallel the multiprocessors of the Nvidia graphics processing unit card. The hDL-system provided the highest discrimination accuracy of 99.1% using the × 200 lens magnification images as compared to the 92.5.% best accuracy achieved by the sML-system, employing both the × 50 and × 200 lens magnification images. Our results showed that the hDL-system was superior to the sML-system (i) in discriminating low-grade from high-grade CRC-lesions and (ii) by requiring fewer images for its best design, only those at the × 200 lens magnification. The sML-system by employing textural and colour features in its design revealed that high-grade CRC lesions are characterized by (i) loss in the definition of structures, (ii) coarser texture in larger structures, (iii) hazy formless texture, (iv) lower AIB1 uptake, (v) lower local correlation and (vi) slower varying image contrast.





Similar content being viewed by others
Abbreviations
- AIB1:
-
Amplified in breast cancer 1
- CART:
-
Classification and Regression Tree
- CNN:
-
Convolutional Neural Network
- CRC:
-
Colorectal cancer
- DL:
-
Deep Learning
- DSS:
-
Decision support system
- H&E:
-
Haematoxylin & Eosin
- HG_CRC:
-
High-grade Colorectal Lesion
- IHC:
-
Immunohistochemistry
- KNN:
-
K-Nearest Neighbour
- LG_CRC:
-
Low-grade Colorectal Cancer
- LDA:
-
Linear Discriminant Analysis
- PNN:
-
Probabilistic Neural Network
- RF:
-
Random Forest
- SVM:
-
Support Vector Machines
- hDL-system:
-
Hybrid Deep Learning System
- sML-system:
-
Supervised Machine Learning System
- CLR MedAD_Ch_a_x50:
-
Median Absolute Deviation of colour channel a
- CLR MedV_Ch_a _x200:
-
Min Value of colour channel a
- CLR MinV_Ch_b_x500:
-
Min Value of colour channel b
- CLR RV_Ch_a_x500:
-
Range Values of colour channel a
- CON _x200:
-
Contrast
- DENT_x200:
-
Difference entropy
- DVAR_x200:
-
Difference variance
- ICM1_x200:
-
Information measure of correlation 1
- IDM _x200:
-
Inverse different moment
- Kurt_x500:
-
Kurtosis
- LRE _x200:
-
Long run emphasis
- RLNU_x50:
-
Run length non-uniformity
- RP_x200:
-
Run percentage
- SAV_x200:
-
Sum average
- Skew_x200:
-
Skewness
- dwt2D_MedV_x50:
-
Median Value of W in diagonal direction (where W is the 2nd level discrete wavelet transform):
- dwt2H_MedV_x200:
-
Median Value of W in horizontal direction (where W is the 2nd level discrete wavelet transform)
References
Siegel, R.L., Miller, K.D., Fedewa, S.A., Ahnen, D.J., Meester, R.G.S., Barzi, A., Jemal, A.: Colorectal cancer statistics, 2017. CA: a cancer journal for clinicians, 67(3):177–193 (2017)
Kunhoth, S., Al Maadeed, S., Bouridane, A.: Medical and computing insights into colorectal tumors. Int. J. Life Sci. Biotech. Pharm. Res. 4(2), 122–126 (2015)
Phillips, R.K.S., Hittinger, R., Blesovsky, L., Fry, J.S., Fielding, L.P.: Large bowel cancer: Surgical pathology and its relationship to survival. Br. J. Surg. 71, 604–610 (1984)
Freedman, L.S., Macaskill, P., Smith, A.N.: Multivariate analysis of prognostic factors for operable rectal cancer. Lancet 2(8405), 733–736 (1984)
Jass, J.R., Atkin, W.S., Cuzick, J., Bussey, H.J., Morson, B.C., Northover, J.M., Todd, I.P.: The grading of rectal cancer: historical perspectives and a multivariate analysis of 447 cases. Histopathology 41(3A), 59–81 (2002)
Thomas, G.D., Dixon, M.F., Smeeton, N.C., Williams, N.S.: Observer variation in the histological grading of rectal carcinoma. J. Clin. Pathol. 36(4), 385–391 (1983)
Andrion, A., Magnani, C., Betta, P.G., Donna, A., Mollo, F., Scelsi, M., Bernardi, P., Botta, M., Terracini, B.: Malignant mesothelioma of the pleura: interobserver variability. J. Clin. Pathol. 48(9), 856–860 (1995)
Rathore, S., Hussain, M., Ali, A., Khan, A.: A recent survey on colon cancer detection techniques. IEEE/ACM Trans. Comput. Biol. Bioinf. 10(3), 545–563 (2013)
Turner, J.K., Williams, G.T., Morgan, M., Wright, M., Dolwani, S.: Interobserver agreement in the reporting of colorectal polyp pathology among bowel cancer screening pathologists in Wales. Histopathology 62(6), 916–924 (2013)
Blenkinsopp, W.K., Stewart-Brown, S., Blesovsky, L., Kearney, G., Fielding, L.P.: Histopathology reporting in large bowel cancer. J. Clin. Pathol. 34(5), 509–513 (1981)
Allen, D.C., Hamilton, P.W., Watt, P.C., Biggart, J.D.: Morphometrical analysis in ulcerative colitis with dysplasia and carcinoma. Histopathology 11(9), 913–926 (1987)
Hamilton, P.W., Allen, D.C., Watt, P.C., Patterson, C.C., Biggart, J.D.: Classification of normal colorectal mucosa and adenocarcinoma by morphometry. Histopathology 11(9), 901–911 (1987)
Allen, D.C., Hamilton, P.W., Watt, P.C., Biggart, J.D.: Architectural morphometry in ulcerative colitis with dysplasia. Histopathology 12(6), 611–621 (1988)
Hamilton, P.W., Allen, D.C., Watt, P.C.: A combination of cytological and architectural morphometry in assessing regenerative hyperplasia and dysplasia in ulcerative colitis. Histopathology 17(1), 59–68 (1990)
Hamilton, P.W., Bartels, P.H., Thompson, D., Anderson, N.H., Montironi, R., Sloan, J.M.: Automated location of dysplastic fields in colorectal histology using image texture analysis. J. Pathol. 182(1), 68–75 (1997)
Esgiar, A.N., Naguib, R.N., Sharif, B.S., Bennett, M.K., Murray, A.: Fractal analysis in the detection of colonic cancer images. IEEE Trans. Inf. Technol. Biomed. Publ. IEEE Eng. Med. Biol. Soc. 6(1), 54–58 (2002)
Ficsor, L., Varga, V.S., Tagscherer, A., Tulassay, Z., Molnar, B.: Automated classification of inflammation in colon histological sections based on digital microscopy and advanced image analysis. Cytometry Part A J. Int. Soc. Anal. Cytol 73(3), 230–237 (2008)
Masood, K., Rajpoot, N.: Texture based classification of hyperspectral colon biopsy samples using CLBP. In: Proceedings of International Symposiumon Biomedical Imaging: From Nano to Macro, Boston 2009. Pp. 1011–1014 (2009)
Kalkan, H., Nap, M., Duin, R. P., Loog, M.: Automated classification of local patches in colon histopathology. In: 21st Int’l Conf. Pattern Recognition, pp. 61–64 (2012).
Jiao, L., Chen, Q., Li, S.: Xu Y Colon cancer detection using whole slide histopathological images. In: World Congress on Medical Physics and Biomedical Engineering, pp. 1283–1286. Springer, Berlin Heidelberg (2013)
Rathore, S., Hussain, M., Aksam Iftikhar, M., Jalil, A.: Ensemble classification of colon biopsy images based on information rich hybrid features. Comput. Biol. Med. 47, 76–92 (2014)
Xu, Y., Mo, T., Feng, Q., Zhong, P., Lai, M., EI-C C Deep learning of feature representation with multiple instance learning for medical image analysis. In: IEEE International Conference on Acoustics, Speech and Signal Processing (ICASSP), pp. 1626–1630 (2014).
Kumar, R., Srivastava, R., Srivastava, S.: Detection and classification of cancer from microscopic biopsy images using clinically significant and biologically interpretable features. J. Med. Eng. 2015, 457906 (2015)
Rathore, S., Hussain, M., Khan, A.: Automated colon cancer detection using hybrid of novel geometric features and some traditional features. Comput. Biol. Med. 65, 279–296 (2015)
Chaddad, A., Desrosiers, C., Bouridane, A., Toews, M., Hassan, L., Tanougast, C.: Multi texture analysis of colorectal cancer continuum using multispectral imagery. PLoS ONE 11(2), 1–17 (2016)
Kather, J.N., Weis, C.A., Bianconi, F., Melchers, S.M., Schad, L.R., Gaiser, T., Marx, A., Zollner, F.G.: Multi-class texture analysis in colorectal cancer histology. Sci. Rep. 6, 27988 (2016)
Chaddad, A., Tanougast, C.: Texture analysis of abnormal cell images for predicting the continuum of colorectal cancer. Anal. Cell. Pathol. 2017, 8428102 (2017)
Awan, R., Sirinukunwattana, K., Epstein, D., Jefferyes, S., Qidwai, U., Aftab, Z., Mujeeb, I., Snead, D., Rajpoot, N.: Glandular morphometrics for objective grading of colorectal adenocarcinoma histology images. Scientific reports 7(1), 16852 (2017)
Peyret, R., Bouridane, A., Khelifi, F., M. Atif Tahir b, Al-Maadeed S, : Automatic classification of colorectal and prostatic histologic tumor images using multiscale multispectral local binary pattern texture features and stacked generalization. Neurocomputing 275, 83–93 (2018)
Pacal, I., Karaboga, D., Basturk, A., Akay, B., Nalbantoglu, U.: A comprehensive review of deep learning in colon cancer. Comput. Biol. Med. 126, 104003 (2020)
Shaban, M., Awan, R., Fraz, M.M., Azam, A., Tsang, Y.W., Snead, D., Rajpoot, N.M.: Context-Aware Convolutional Neural Network for Grading of Colorectal Cancer Histology Images. IEEE Trans. Med. Imaging 39(7), 2395–2405 (2020)
Zhou, Y., Graham, S., Alemi Koohbanani, N., Shaban, M., Heng, P.A., Rajpoot, N.: CGC-net: Cell graph convolutional network for grading of colorectal cancer histology images. In: Proceedings - 2019 International Conference on Computer Vision Workshop, ICCVW 2019, 2019. pp. 388–398.
Anzick, S.L., Kononen, J., Walker, R.L., Azorsa, D.O., Tanner, M.M., Guan, X.Y., Sauter, G., Kallioniemi, O.P., Trent, J.M., Meltzer, P.S.: AIB1, a steroid receptor coactivator amplified in breast and ovarian cancer. Science 277(5328), 965–968 (1997)
Ghadimi, B.M., Schrock, E., Walker, R.L., Wangsa, D., Jauho, A., Meltzer, P.S., Ried, T.: Specific chromosomal aberrations and amplification of the AIB1 nuclear receptor coactivator gene in pancreatic carcinomas. Am. J. Pathol. 154(2), 525–536 (1999)
Sakakura, C., Hagiwara, A., Yasuoka, R., Fujita, Y., Nakanishi, M., Masuda, K., Kimura, A., Nakamura, Y., Inazawa, J., Abe, T., Yamagishi, H.: Amplification and over-expression of the AIB1 nuclear receptor co-activator gene in primary gastric cancers. Int. J. Cancer 89(3), 217–223 (2000)
Wang, Y., Wu, M.C., Sham, J.S., Zhang, W., Wu, W.Q., Guan, X.Y.: Prognostic significance of c-myc and AIB1 amplification in hepatocellular carcinoma. A broad survey using high-throughput tissue microarray. Cancer 95(11), 2346–2352 (2002)
Xie, D., Sham, J.S., Zeng, W.F., Lin, H.L., Bi, J., Che, L.H., Hu, L., Zeng, Y.X., Guan, X.Y.: Correlation of AIB1 overexpression with advanced clinical stage of human colorectal carcinoma. Hum. Pathol. 36(7), 777–783 (2005)
Xu, F.P., Xie, D., Wen, J.M., Wu, H.X., Liu, Y.D., Bi, J., Lv, Z.L., Zeng, Y.X., Guan, X.Y.: SRC-3/AIB1 protein and gene amplification levels in human esophageal squamous cell carcinomas. Cancer Lett. 245(1–2), 69–74 (2007)
Liu, M.Z., Xie, D., Mai, S.J., Tong, Z.T., Shao, J.Y., Fu, Y.S., Xia, W.J., Kung, H.F., Guan, X.Y., Zeng, Y.X.: Overexpression of AIB1 in nasopharyngeal carcinomas correlates closely with advanced tumor stage. Am. J. Clin. Pathol. 129(5), 728–734 (2008)
Tzelepi, V., Grivas, P., Kefalopoulou, Z., Kalofonos, H., Varakis, J.N., Melachrinou, M., Sotiropoulou-Bonikou, G.: Estrogen signaling in colorectal carcinoma microenvironment: expression of ERbeta1, AIB-1, and TIF-2 is upregulated in cancer-associated myofibroblasts and correlates with disease progression. Virchows Archiv Int. J. Pathol. 454(4), 389–399 (2009)
Chen, L., Wang, C., Zhang, X., Gao, K., Liu, R., Shi, B., Hou, P.: AIB1 Genomic Amplification Predicts Poor Clinical Outcomes in Female Glioma Patients. J. Cancer 7(14), 2052–2060 (2016)
Theodosi, A., Glotsos, D., Kostopoulos, S., Kalatzis, I., Tzelepi, V., Ravazoula, P., Asvestas, P., Cavouras, D., Sakellaropoulos, G.: Correlating Changes in the Epithelial Gland Tissue With Advancing Colorectal Cancer Histologic Grade, Using IHC Stained for AIB1 Expression Biopsy Material. Appl Immunohistochem Mol Morphol AIMM (2018)
Greene, F.L.: AJCC Cancer Staging Manual. Springer, New York (2002)
Xu, Y., Jia, Z., Wang, L.B., Ai, Y., Zhang, F., Lai, M., Chang, E.I.: Large scale tissue histopathology image classification, segmentation, and visualization via deep convolutional activation features. BMC Bioinf 18(1), 281 (2017)
Simonyan, K., Zisserman, A.: Very deep convolutional networks for large-scale image recognition. In: 3rd International Conference on Learning Representations, ICLR 2015 - Conference Track Proceedings (2015).
Guyon, I., Weston, J., Barnhill, S., Vapnik, V.: Gene selection for Cancer classification using support vector machines. Mach. Learn. 46(1), 389–422 (2002)
Haralick, R., Shanmugam, K., Dinstein, I.: Textural features for image classification. IEEE Trans. Syst. Man Cybern. 3, 610–621 (1973)
Galloway, M.M.: Texture analysis using grey level run lengths. Comput. Graph. Image Process. 4, 172–179 (1975)
H. Tamura SM, and T. Yamawaki: Textural features corresponding to visual perception. IEEE Trans. Syst. Man Cybern. 8(6), 460–473 (1978)
Specht, D.: Probabilistic neural networks. Neural Netw. 3, 109–118 (1990)
Theodoridis, S., Pikrakis, A., Koutroumbas, K., Cavouras, D.: An Introduction to Pattern Recognition: A Matlab Approach. Academic Press, Oxford (2010)
Theodoridis, S., Koutroumbas, K.: Pattern Recognition, 2nd edn. Elsevier, San Diego (2003)
Foley, D.: Considerations of sample and feature size. IEEE Trans. Inf. Theory 18(5), 618–626 (1972)
Ambroise, C., McLachlan, G.J.: Selection bias in gene extraction on the basis of microarray gene-expression data. Proc. Natl. Acad. Sci. U.S.A. 99(10), 6562–6566 (2002)
Sidiropoulos, K., Glotsos, D., Kostopoulos, S., Ravazoula, P., Kalatzis, I., Cavouras, D., Stonham, J.: Real time decision support system for diagnosis of rare cancers, trained in parallel, on a graphics processing unit. Comput. Biol. Med. 42(4), 376–386 (2012)
Esgiar, A.N., Naguib, R.N., Bennett, M.K., Murray, A.: Automated feature extraction and identification of colon carcinoma. Anal. Quant. Cytol. Histol. 20(4), 297–301 (1998)
Cheng, H.D., Sun, Y.: A Hierarchical Approach to Color Image Segmentation Using Homogeneity. IEEE Trans. Image Process. 9(12), 2071–2082 (2000)
Acknowledgement
The authors would like to thank the Department of Pathology of the University Hospital of Patras, Greece, for supplying the IHC stained for AIB1 expression material.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix
The mathematical formulations of the eight (8) best-features combination of the sML-system design are presented below.
Α.1 Histogram feature
-
(1)
Kurtosis (Kurt)
where g(i,j) is the pixel intensity in position (i,j), N the total number of pixels, m is the mean value of the g, and std is the standard deviation of g.
Α.2 Co-occurrence matrix based features
-
(2)
Sum average (SAV)
where Ng is the number of gray levels in the image, i,j = 1,…,Ng, and p(i,j) is the co-occurrence matrix, and px+y is \(p_{x + y} (k) = \sum\limits_{i = 1}^{{N_{g} }} {\sum\limits_{j = 1}^{{N_{g} }} {p(i,j),i + j = k,k = 2,3,...,2N_{g} } }\).
-
(3)
Difference variance(DVAR)
where px-y is \(p_{x - y} (k) = \sum\limits_{i = 1}^{{N_{g} }} {\sum\limits_{j = 1}^{{N_{g} }} {p(i,j),|i - j| = k,k = 2,3,...,N_{g} - 1} }\).
-
(4)
Difference entropy (DENT)
-
(5)
Information measure of correlation 1 (ICM1)
$$ ICM1 = \frac{HXY - HXY1}{{\max \left\{ {HX,HY} \right\}}} $$
where \(HXY = - \sum\limits_{i = 0}^{{N_{g} - 1}} {\sum\limits_{j = 0}^{{N_{g} - 1}} {p(i,j)\log \left( {p(i,j)} \right)} }\)
Α.4 Wavelet based feature
-
(6)
Median Value of the 2nd level discrete wavelet transform in the horizontal direction (dwt2H_MedV).
The discrete wavelet function of an image \(f\left(x,y\right)\) of size M x N is defined as:
where \(\varphi \left( {x,y} \right)\) is a scaling function, with j = 0,1,2…
\(W_{\varphi }\) are the coefficients define an approximation of image \(f\left( {x,y} \right)\) at level (scale) j0.
and \(W_{\psi }\) are the coefficients that add horizontal, vertical and diagonal details for levels j ≥ j0.
Α.5 Tamura-based features
-
(7)
Tamura coarseness 4
where m, n are region dimensions and
where k is the best scaling for highest neighborhood average.
The particular feature is the value of the 3rd bin histogram of Sbest .
Α.6 Lab colour transform-based features
-
(8)
Median Value of colour channel a* (CLR MedV_Ch_a)
According to the CIE, the coordinates of the Lab colour space are derived by a nonlinear transformation of the three primary colours X, Y and Z. The linear transformation of RGB space to X, Y and Z is defined as [57]
where Ch_x is the *a channel of L*a*b colour transform.
Rights and permissions
About this article
Cite this article
Theodosi, A., Ouzounis, S., Kostopoulos, S. et al. Design of a hybrid deep learning system for discriminating between low- and high-grade colorectal cancer lesions, using microscopy images of IHC stained for AIB1 expression biopsy material. Machine Vision and Applications 32, 58 (2021). https://doi.org/10.1007/s00138-021-01184-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00138-021-01184-8