Abstract
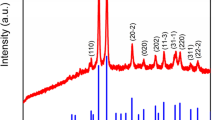
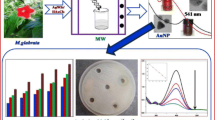
A simple one step green procedure is intended to investigate for the first time by employing ruthenium oxide nanoparticles (RuO2 NPs) derived from Catharanthus roseus (CR) and Moringa oleifera (MO) leaves and evaluate their catalytic—degradation of MB dye, antibacterial screening and in vitro antioxidant assays. The as-synthesized RuO2 NPs were characterized by XRD, HR-TEM, FT-IR, EDX and UV–Visible spectrometer. The HR-TEM image reveals spherical morphology with average particle size 5.6 nm (CR-RuO2 NP) and 4.6 nm (MO-RuO2 NP). The research findings of the current study elucidates the superiority of CR-RuO2 NPs and well exhibited towards scavenging activity, admirable anti-bacterial effect against K. pneumonia (21.6 ± 0.24 mm), and catalytic degradation percentage as 79.02%. The adsorption kinetics indeed suggest more impressive pseudo-second-order kinetic model than Lagergren first-order kinetics at 10 ppm concentration of MB dye. Our results thus provide a platform for further research on the applications of RuO2 NPs as disinfectant and better catalyst.














Similar content being viewed by others
References
K. Singh, D. Kukkara, R. Singh, P. Kukkard, N. Bajaj, J. Singh, M. Rawat, A. Kumar, and K. Kim (2019). J Ind Eng Chem. https://doi.org/10.1016/j.jiec.2019.09.008.
S. Jain and M. M. Singh (2017). J. Sci. Rep. 7, 15867. https://doi.org/10.1038/s41598-017-15724-8.
M. Nasrollahzadeh and S. M. Sajadi (2015). J Colloid Interface Sci. https://doi.org/10.1016/j.jcis.2015.08.020.
I. Fierascu, M. I. Georgiev, A. Ortan, R. C. Fierascu, S. M. Avramescu, D. Ionescu, A. Sutan, A. Brinzan, and L. M. Ditu (2017). J. Sci. Rep. 7, 12428. https://doi.org/10.1038/s41598-017-12804-7.
J. Singh, P. Kukkar, H. Sammi, M. Rawat, G. Singh, and D. Kukkar (2017). Particulate Sci Technol. https://doi.org/10.1080/02726351.2017.1390512.
J. Singh, H. Kaur, D. Kukkar, V. K. Mukamia, S. Kumar, and M. Rawat (2019). Mater. Res. Express. 6, 115007.
N. Saifuddin, C. W. Wong, and A. A. Nur Yasumira (2009). J. Chem. 6, 61–70.
K. Okitsu, Y. Mizukoshi, T. A. Yamamoto, Y. Maeda, and Y. Nagata (2007). Mater Lett. 61, 3429–3431.
M. M. El-Sheekh and H. Y. El-Kassas (2016). Genet. Eng. Biotechnol. 14, 299–310.
N. E. El-Naggar, A. Mohamedin, S. S. Hamza, and A. Sherief (2016). J Nanomate. ID 3257359.
M. Gilaki (2010). J. Biol. Sci. 10, 465–467.
Atta-ul-Haq, M. Saeed, M. A. Jamal, N. Akram, T. H. Bokhari, and U. Afaqet (2018). Z. Phys. Chem. AOP
M. Nasrollahzadeh and S. M. Sajadi (2015). RSC Adv. 5, 46240.
S. M. Sajadi, M. Nasrollahzadeh, and M. Maham (2016). J Colloid Interface Sci. https://doi.org/10.1016/j.jcis.2016.02.009.
K. Singh, D. Kukkar, R. Singh, P. Kukkar, and K. Kim (2018). J. Ind. Eng. Chem. 81, 196–205. https://doi.org/10.1016/j.jiec.2018.07.026.
S. M. Anjum, C. H. Khadri, and K. Riazunnisa (2017). ATMS (Ed. Narayana Reddy) Roshan Publishers, (Chapter 6, pp. 64–69).
S. M. Anjum, M.V. Vani and K. Riazunnisa (2019). Emerging Trends in Advanced Spectroscopy (Ed. Y. Weiman, K.P. Jibin, G.L Praveen, S. Thomas, and N. Kalarikkal) Rivers publishers, (Chapter 6, pp. 69–79).
S. M. Anjum and K. Riazunnisa (2020). Data Brief 29, 105258. https://doi.org/10.1016/j.dib.2020.105258.
S. K. Kannan and M. Sundararajan (2015). Adv. Powder Technol. 26, 1505–1511. https://doi.org/10.1016/j.apt.2015.08.009.
P. K. Gupta and L. Mishra (2020). Nanoscale Adv. 2, 1774.
P. Banerjee and D. Nath (2015). Nano Sci. Technol. 2, 1–14.
K. Gopinath, K. Viswanathan, S. Gowri, S. Venugopal, K. Subramanian, and A. Arumugam (2014). J. Nano Struct. Chem. 4, 83.
T. J. L. Edison and M. G. Sethuraman (2012). J. Process. Biochem. 47, 1351–1357.
S. Lagergren (1989). Handlingar 24, 1–39.
Y. S. Ho and G. McKay (1999). Process. Biochem. 34, 451–465.
S. Gupta, C. Giordano, M. Gradzielski, and S. K. Mehta (2013). J. Colloid Interface Sci. 411, 173–181.
P. S. S. Kumar, A. Manivel, S. Anandan, M. Zhou, F. Grieser, and M. A. Kumar (2010). Colloid Surf. A 356, 140–144.
S. Duman and S. Ozkar (2013). Int. J. Hydrog. Energy 38, 10000–10011.
G. Borah and P. Sharma (2011). Indian J. Chem Sect. B. 50, 41–45.
Y. Wang and N. Herron (1991). J. Phys. Chem. 95, 525–532.
J. W. Racquel, S. K. Lee, I. H. Hyacinth, J. M. Hibbert, M. E. Reid, A. O. Wheatley, and H. N. Asemota (2017). Plants 6, 48. https://doi.org/10.3390/plants6040048.
H. E. Miller, F. Rigelhof, L. Marquart, A. Prakash, and M. Kanter (2000). Nutrition 19, 3125–3195.
K. Jyothi and A. Singh (2016). J. Genet. Eng. Biotechnol. 14, 311–317.
T. Shahwan, S. A. Sirriah, M. Nairat, E. Boyac, A. E. Eroglu, T. B. Scott, and K. R. Hallman (2011). Chem. Eng. J. 172, 258–266.
M. K. Indana, B. R. Gangapuram, R. Dandigala, R. Bandi, and V. Guttena (2016). J. Anal. Sci. Technol. 7, 19.
M. Atarod, M. Nasrollahzadeh, and S. M. Sajadi (2016). J. Colloid Interface Sci. 462, 272–279.
T. V. M. Sreekanth, M. J. Jung, and I. Y. Eom (2016). Appl. Surf. Sci. 361, 102–106.
R. S. Gonzalo, R. T. Neira, C. Alvarado, C. I. Blancheteau, N. Benito, A. G. Rodríguez, R. Marcos, H. Pesenti, and E. R. Carmona (2019). Environ Sci Pollut Res. https://doi.org/10.1007/s11356-019-04934-4.
M. Zahoor, A. Arshad, Y. Khan, et al. (2018). Appl. Nanosci. 8, 1091–1099.
X. Yang, H. Zhong, Y. Zhu, H. Jiang, J. Shen, J. Huang, and C. Li (2014). J. Mater. Chem. A 2, 9040–9047.
W. K. Azira, W. M. Khalir, K. Shameli, S. D. Jazayeri, N. A. Othman, N. W. C. Jusoh, and N. M. Hassan (2020). Nanomaterials 10, 1104. https://doi.org/10.3390/nano10061104.
T. Sowmyya and G. V. Lakshmi (2018). J. Environ. Chem. Eng. 6, 3590–3601.
S. Yihan, L. Mingming, and Z. Guo (2018). J. Colloids Interface Sci. 527, 187–194.
P. S. Rajegaonkar, B. A. Deshpande, M. S. More, S. S. Waghmare, V. V. Sangawe, A. Inamdar, M. D. Shirsat, and N. N. Adhapure (2018). Mater. Sci. Eng. 93, 623–629.
B. Sreedhar, D. K. Devi, and D. Yada (2011). Catal. Commun. 12, 1009.
Z. Wang, C. Xu, G. Gao, and X. Li (2014). RSC Adv. 40, 13644.
P. Zhang, Y. Sui, C. Wang, Y. Wang, G. Cui, and C. Wang (2014). Nanoscale 6, 5343.
L. Ai, C. Zeng, and Q. Wang (2011). Catal. Commun. 14, 68.
A. Hatamifard, M. Nasrollahzadeh, and J. Lipkowski (2015). RSC Adv. https://doi.org/10.1039/C5RA18476B.
K. L. Wu, X. W. Wei, X. M. Zhou, D. H. Wu, X. W. Liu, Y. Ye, et al. (2011). J Phys Chem C. 115, 16268e74.
M. Maryami, M. Nasrollahzadeh, E. Mehdipour, and S. M. Sajadi (2016). Int. J. Hydrogen Energy 41, 21236–21245. https://doi.org/10.1016/j.ijhydene.2016.09.130.
R. Shani, H. Singh, R. Trivedi, and V. Soni (2020). J. Sci. Rep. 10, 9616. https://doi.org/10.1038/S41598-020-66851-8.
S. Kumar, C. M. A. ParleH, M. A. Isaacs, D. V. Jowett, R. E. Douthwaite, M. C. R. Cockett, and A. Lee (2016). Appl. Catal. B 189, 226–232.
W. Zou, Z. Lei, L. Liu, X. Wang, J. Sun, S. Wu, D. Yu, C. Tang, G. Fci, and D. Lin (2016). Appl. Catal. B 181, 495–503.
S. Li, Q. Lin, X. Liu, L. Yang, J. Ding, F. Dong, Y. Li, M. Irfan, and P. Zhang (2018). RSC Adv. 8, 20277–20286.
Acknowledgements
The first author SAM express gratitude towards UGC for the award of MANF research fellowship [Grant No. F1-17.1/2017-18/MANF-2017-18-AND-73354/ (SAIII/Website)] and also thankful to Yogi Vemana University for the facilities provided.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Anjum, S.M., Riazunnisa, K. Fine Ultra-small Ruthenium Oxide Nanoparticle Synthesis by Using Catharanthus roseus and Moringa oleifera Leaf Extracts and Their Efficacy Towards In Vitro Assays, Antimicrobial Activity and Catalytic: Adsorption Kinetic Studies Using Methylene Blue Dye. J Clust Sci 33, 1103–1117 (2022). https://doi.org/10.1007/s10876-021-02037-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-021-02037-0