Abstract
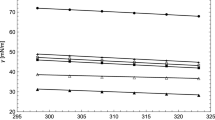
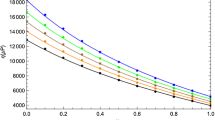
Surface tension and phase equilibria of N,N-dimethylcyclohexylamine (DMCA) mixtures with alcohol (propanol, iso-propanol, butanol, and iso-butanol) were modeled over the whole range of composition and different temperature ranging from (288.15 to 308.15) K. The predictive results of the Peng–Robinson equation of state with quadratic mixing rule indicated that the DMCA + alcohol mixtures are not azeotropic and that the bubble curve is linear, except for the DMCA + ethanol mixture. The surface tension of the binary mixtures was modeled with linear gradient theory, parachor method, Shereshefsky method, and Lamperski method. The linear gradient theory used as adjustment approach improved the results of surface tension prediction and correctly modeled this thermodynamic property for all mixtures, the flexibility of parachor method as an adjustment approach improved the predictive results for some mixtures, Shereshefsky method was able to successfully model the surface tension of the DMCA + ethanol, DMCA + propanol, and DMCA + butanol mixtures, and Lamperski method was able to successfully model the surface tension of the DMCA + propanol and DMCA + butanol mixtures, while the DMCA + ethanol and DMCA + isobutanol mixtures had an acceptable statistical deviation. Furthermore, Lamperski method was the best predicted model to model surface tension of the binary mixtures. Based on Shereshefsky model, the standard Gibbs energy of adsorption and the free energy change in the surface region were calculated. The free energy change was used to obtain the number of molecular layers in the surface region. Also, with Shereshefsky method it was obtained that alcohol is not absorbed at the surface which was also confirmed with Lamperski method. Finally, it is important to note that phase equilibria and surface tension of the DMCA + alcohol mixtures is modeled with theoretical approaches for the first time. On the other hand, for future experimental measurements of phase equilibria, our results could serve as an initial approximation of equilibrium, and the correlations obtained for the binary parameters of the linear gradient theory and parachor method can be used to predict surface tension at other temperatures outside the range 288.15 to 308.15 K.

















Similar content being viewed by others
Abbreviations
- a :
-
Cohesion parameter in the PR-EOS
- b :
-
Covolume parameter in the PR-EOS
- c :
-
Influence parameter
- \(k_{ij}\) :
-
Interaction parameter for the quadratic mixing rule
- \(n_c\) :
-
Number of components of the mixture
- n :
-
Number of points in the linear gradient theory
- N :
-
Number of experimental points
- P :
-
Absolute pressure
- R :
-
Universal gas constant
- T :
-
Absolute temperature
- w :
-
Mole fraction
- x :
-
Liquid mole fraction
- y :
-
Vapor mole fraction
- AAD :
-
Statistical deviation
- z :
-
Position in the interface
- \(f_0\) :
-
Helmholtz energy density
- A :
-
Adjustable parameter of the influence parameter
- B :
-
Adjustable parameter of the influence parameter
- \(\mathbb {P}\) :
-
Parachor parameter of the pure fluid
- \(c_{ij}\) :
-
Cross-influence parameter
- \(\overline{A}_2\) :
-
The molar area of the solute (2)
- \(M_2\) :
-
Molecular weight of solute (2)
- \(N_0\) :
-
Avogadro’s number
- \(n_i\) :
-
Number of moles of the pure ith component
- d :
-
Thickness of monomolecular layer
- \(V_i\) :
-
Molar volume of the ith molecules
- \(r_{12}\) :
-
Quotient between \(n_1\) and \(n_2\)
- \(\mu\) :
-
Chemical potential
- \(\rho\) :
-
Molar density
- \(\gamma\) :
-
Surface tension
- \(\Omega\) :
-
Grand thermodynamic potential
- \(\beta _{12}\) :
-
Symmetric parameter of the linear gradient theory
- \(\omega\) :
-
Acentric factor
- \(\lambda _{12}\) :
-
Adjustable parameter of the parachor method
- \(\Delta G_s\) :
-
Free energy change of replacing one mole of solvent with one mole of solute in the interface
- \(\Delta G ^o\) :
-
Standard Gibbs energy of adsorption
- \(\Delta \gamma _0\) :
-
Difference between the surface tensions of pure solvent and pure solute
- \(\alpha\) :
-
Slope of the Shereshefsky method
- \(\beta\) :
-
Intercept of the Shereshefsky method
- \(\delta _2\) :
-
Number of solute molecular layers in the surface region in respect of the bulk region
- \(\theta _i\) :
-
Surface fraction of the ith component of solution
- \(\nu _i\) :
-
Volume fraction of the ith component of solution
- c :
-
Critical condition
- i, j :
-
Species
- s :
-
Surface
- b :
-
Bulk
- ref :
-
Reference
- 0:
-
Equilibrium condition
- exp :
-
Experimental
- calc :
-
Calculated
- L :
-
Liquid phase
- V :
-
Vapor phase
- o :
-
Standard
References
D. Zhao, Y. Zhuang, C. Fan, F. Yang, Y. Chen, X. Zhang, J. Chem. Thermodyn. 143, 106041 (2020)
X. Liang, M.L. Michelsen, G.M. Kontogeorgis, Fluid Phase Equilib. 428, 153–163 (2016)
U. Brea, D. Gómez-Díaz, J.M. Navaza, A. Rumbo, J. Taiwan Inst. Chem. Eng. 102, 250–258 (2019)
S. Lin, H. Lu, Y. Liu, C. Liu, B. Liang, K. Wu, J. Chem. Thermodyn. 123, 8–16 (2018)
F. Tzirakis, I. Tsivintzelis, A.I. Papadopoulos, P. Seferlis, Chem. Eng. Sci. 199, 20–27 (2019)
D. Papaioannou, C.G. Panayiotou, J. Chem. Eng. Data 39, 457–462 (1994)
D.Y. Peng, D.B. Robinson, Ind. Eng. Chem. Fundam. 15, 59–64 (1976)
T.Y. Kwak, G.A. Mansoori, Chem. Eng. Sci. 41, 1303–1309 (1986)
B.S. Carey, L.E. Scriven, H.T. Davis, AIChE J. 24, 1076–1080 (1978)
B.S. Carey, L.E. Scriven, H.T. Davis, AIChE J. 26, 705–711 (1980)
V. Vinvs, B. Planková, J. Hruby. Int. J. Thermophys. 34, 792–812 (2013)
A. Mejía, H. Segura, Int. J. Thermophys. 25, 1395–1414 (2004)
A. Mejía, H. Segura, J. Wisniak, I. Polishuk, J. Phase Equilibria Diffus. 26, 215–224 (2005)
A. Mejía, H. Segura, L.F. Vega, J. Wisniak, Fluid Phase Equilib. 227, 225–238 (2005)
A. Hernández, M. Cartes, A. Mejía, Fuel 229, 105–115 (2018)
A. Hernández, Chem. Phys. 534, 110747 (2020)
A. Hernández, S. Khosharay, Int. J. Thermophys. 41, 1–22 (2020)
A. Hernández, D. Zabala, Int. J. Thermophys. 42, 1–21 (2021)
Y.-X. Zuo, E.H. Stenby, J. Chem. Eng. Jpn. 29, 159–165 (1996)
Y.-X. Zuo, E.H. Stenby, J. Colloid Interface Sci. 182, 126–132 (1996)
Y.-X. Zuo, E.H. Stenby et al., SPE J. 3, 134–145 (1998)
H. Lin, Y.Y. Duan, J.T. Zhang, Int. J. Thermophys. 29, 423–433 (2008)
A. Hernández, Int. J. Thermophys. 41, 1–18 (2020)
A. Hernández, Int. J. Thermophys. 42, 1–27 (2020)
J.C. Eriksson, Adv. Chem. Phys. 6, 145–174 (1964)
F.B. Sprow, J.M. Prausnitz, Trans. Faraday Soc. 62, 1105–1111 (1966)
J.L. Shereshefsky, J. Colloid Interface Sci. 24, 317–322 (1967)
R. Tahery, H. Modarress, J. Satherley. Chem. Eng. Sci. 60, 4935–4952 (2005)
R. Tahery, J. Solut. Chem. 46, 1152–1164 (2017)
R. Tahery, S. Khosharay, J. Mol. Liq. 247, 354–365 (2017)
R. Tahery, J. Chem. Thermodyn. 106, 95–103 (2017)
R. Tahery, J. Solut. Chem. 47, 278–292 (2018)
S. Lamperski, J. Colloid Interface Sci. 144, 153–158 (1991)
C. Coquelet, A. Chapoy, D. Richon, Int. J. Thermophys. 25, 133–158 (2004)
X. Wu, X. Du, D. Zheng, Int. J. Thermophys. 31, 308–315 (2010)
W. Su, S. Zhou, L. Zhao, N. Zhou, Int. J. Thermophys. 41, 1–24 (2020)
L.A. Román-Ramírez, G.A. Leeke, Int. J. Thermophys. 41, 1–27 (2020)
J.M. Prausnitz, R.N. Lichtenthaler, E.G. De Azevedo, Molecular Thermodynamics of Fluid-Phase Equilibria (Prentice Hall, Upper Saddle River, 1998).
C. Miqueu, B. Mendiboure, A. Graciaa, J. Lachaise, Fluid Phase Equilib. 207, 225–246 (2003)
D.B. Macleod, Trans. Faraday Soc. 19, 38–41 (1923)
J.A. Hugill, A.J. Van Welsenes, Fluid Phase Equilib. 29, 383–390 (1986)
M. Caceres. Alonso, J. Nunez. Delgado, J. Chem. Eng. Data 27, 331–333 (1982)
M. Caceres. Alonso, J. Nunez. Delgado, J. Chem. Eng. Data 28, 61–62 (1983)
P.J. Flory, J. Chem. Phys. 10, 51–61 (1942)
Aspen Plus. V10. 0; Aspen Technology Inc., Burlington (2017)
T.E. Daubert, R.P. Danner, Data Compilation, Tables of Properties of Pure Compounds (DIPPR, New York, 1985)
D. Zhao, Y. Zhuang, C. Fan, X. Zhang, F. Yang, Y. Chen, J. Chem. Eng. Data 65, 1547–1553 (2020)
A.F.H. Ward, Trans. Faraday Soc. 42, 399–407 (1946)
A. Hartkopf, B.L. Karger, Acc. Chem. Res. 6, 209–216 (1973)
Acknowledgements
A.H acknowledges the economic support given by the UCSC.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hernández, A., Tahery, R. Modeling of Phase Equilibria and Surface Tension for N,N-Dimethylcyclohexylamine + Alcohol Mixtures at Different Temperatures. Int J Thermophys 42, 67 (2021). https://doi.org/10.1007/s10765-021-02815-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-021-02815-y